Key Points
-
An ultra-rare variant in an inherited cardiac condition-related gene that might have contributed to an arrhythmogenic sudden death of the infant is identified in 14% of sudden infant death syndrome (SIDS) cases
-
Current data indicate that the diagnostic yield of nonsynonymous, ultra-rare variants in inherited cardiac condition-related genes in SIDS cohorts is not higher than in the general population
-
This 'reality check' highlights the importance of understanding that causation of SIDS is complex and multifactorial, and that highly-penetrant, monogenic cardiac disorders contribute to only a small number of SIDS cases
-
Simultaneous genetic evaluation of the deceased infant and the parents might offer the only ready way to interpret ultra-rare and novel variants for clinical use
-
When a gene mutation is identified by molecular autopsy, initial familial genetic testing should focus on the parents to determine whether the mutation is inherited or arose de novo in the deceased
-
Specialized, multidisciplinary, cardiac genetic teams have a pivotal role in management of the surviving family; cardiological investigation of surviving relatives might identify a genetic risk, although limited evidence supports this assertion
Abstract
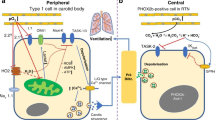
Sudden infant death syndrome (SIDS) is the leading cause of post-neonatal infant mortality in developed countries, characterized by the death of infants for no obvious reason and without prior warning. The complex interaction of multiple factors in the pathogenesis of SIDS is illustrated by the 'triple risk hypothesis', which proposed that SIDS results from a convergence of three overlapping risk factors: a critical developmental period, an exogenous stressor, and underlying genetic and/or nongenetic vulnerability in the infant. Rare variants in genes associated with inherited arrhythmia syndromes and cardiomyopathies have been proposed as the substrate for an infant's critical vulnerability in a small subset of SIDS cases. Given the potential risk of inherited cardiac disease, current guidelines recommend post-mortem genetic testing (molecular autopsy) and cardiological investigation of the surviving family, complemented by targeted genetic testing if appropriate. In this Review, we highlight the latest developments in understanding the spectrum and prevalence of cardiac-mediated SIDS, and discuss the clinical implications of SIDS in the surviving family and the general population.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Goldstein, R. D., Kinney, H. C. & Willinger, M. Sudden unexpected death in fetal life through early childhood. Pediatrics 137, e20154661 (2016).
Moon, R. Y., Horne, R. S., & Hauck, F. R. Sudden infant death syndrome. Lancet 370, 1578–1587 (2007).
Kinney, H. C. & Thach, B. T. The sudden infant death syndrome. N. Engl. J. Med. 361, 795–805 (2009).
Goldstein, R. D., Trachtenberg, F. L., Sens, M. A., Harty, B. J. & Kinney, H. C. Overall postneonatal mortality and rates of SIDS. Pediatrics 137, e20152298 (2016).
Willinger, M. et al. Factors associated with the transition to nonprone sleep positions of infants in the United States: the national infant sleep position study. JAMA 280, 329–335 (1998).
Shapiro-Mendoza, C. K., Tomashek, K. M., Anderson, R. N. & Wingo, J. Recent national trends in sudden, unexpected infant deaths: more evidence supporting a change in classification or reporting. Am. J. Epidemiol. 163, 762–769 (2006).
Matthews, T. J., MacDorman, M. F. & Thoma, M. E. Infant mortality statistics from the 2013 period linked birth/infant death data set. Natl Vital Stat. Rep. 64, 1–30 (2015).
Guntheroth, W. G. & Spiers, P. S. The triple risk hypotheses in sudden infant death syndrome. Pediatrics 110, e64 (2002).
Filiano, J. J. & Kinney, H. C. A perspective on neuropathologic findings in victims of the sudden infant death syndrome: the triple-risk model. Biol. Neonate 65, 194–197 (1994).
Carlin, R. F. & Moon, R. Y. Risk factors, protective factors, and current recommendations to reduce sudden infant death syndrome: a review. JAMA Pediatr. 171, 175–180 (2017).
Weinberg, S. B. & Purdy, B. A. Postmortem leukocyte culture studies in sudden infant death. Nature 226, 1264–1265 (1970).
Froggatt, P., Lynas, M. A. & MacKenzie, G. Epidemiology of sudden unexpected death in infants ('cot death') in Northern Ireland. Br. J. Prev. Soc. Med. 25, 119–134 (1971).
Carpenter, R. G. et al. Repeat sudden unexpected and unexplained infant deaths: natural or unnatural? Lancet 365, 29–35 (2005).
Smialek, J. E. Simultaneous sudden infant death syndrome in twins. Pediatrics 77, 816–821 (1986).
Getahun, D., Demissie, K., Lu, S.-E. & Rhoads, G. G. Sudden infant death syndrome among twin births: United States, 1995–1998. J. Perinatol. 24, 544–551 (2004).
Van Norstrand, D. W & Ackerman, M. J. Genomic risk factors in sudden infant death syndrome. Genome Med. 2, 86 (2010).
Schwartz, P. J. Cardiac sympathetic innervation and the sudden infant death syndrome. A possible pathogenetic link. Am. J. Med. 60, 167–172 (1976).
Schwartz, P. J. et al. The QT interval throughout the first 6 months of life: a prospective study. Circulation 66, 496–501 (1982).
Schwartz, P. J. The quest for the mechanisms of the sudden infant death syndrome: doubts and progress. Circulation 75, 677–683 (1987).
Maron, B. J., Clark, C. E., Goldstein, R. E. & Epstein, S. E. Potential role of QT interval prolongation in sudden infant death syndrome. Circulation 54, 423–430 (1976).
Schwartz, P. J. et al. Prolongation of the QT interval and the sudden infant death syndrome. N. Engl. J. Med. 338, 1709–1714 (1998).
Yiallourou, S. R., Sands, S. A., Walker, A. M. & Horne, R. S. C. Maturation of heart rate and blood pressure variability during sleep in term-born infants. Sleep 35, 177–186 (2012).
Jean-Louis, M. et al. Power spectral analysis of heart rate in relation to sleep position. Biol. Neonate 86, 81–84 (2004).
Schwartz, P. J. et al. A molecular link between the sudden infant death syndrome and the long-QT syndrome. N. Engl. J. Med. 343, 262–267 (2000).
Schwartz, P. J. et al. Molecular diagnosis in a child with sudden infant death syndrome. Lancet 358, 1342–1343 (2001).
Wedekind, H. et al. De novo mutation in the SCN5A gene associated with early onset of sudden infant death. Circulation 104, 1158–1164 (2001).
Ackerman, M. J. et al. Postmortem molecular analysis of SCN5A defects in sudden infant death syndrome. JAMA 286, 2264–2269 (2001).
Priori, S. G. et al. HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart Rhythm 10, 1932–1963 (2013).
Brunklaus, A., Ellis, R., Reavey, E., Semsarian, C. & Zuberi, S. M. Genotype phenotype associations across the voltage-gated sodium channel family. J. Med. Genet. 51, 650–658 (2014).
Gray, B. & Behr, E. R. New insights into the genetic basis of inherited arrhythmia syndromes. Circ. Cardiovasc. Genet. 9, 569–577 (2016).
Schwartz, P. J. et al. Prevalence of the congenital long-QT syndrome. Circulation 120, 1761–1767 (2009).
Goldenberg, I. et al. Risk factors for aborted cardiac arrest and sudden cardiac death in children with the congenital long-QT syndrome. Circulation 117, 2184–2191 (2008).
Priori, S. G. et al. Risk stratification in the long-QT syndrome. N. Engl. J. Med. 348, 1866–1874 (2003).
Antzelevitch, C. et al. J-Wave syndromes expert consensus conference report: emerging concepts and gaps in knowledge. Heart Rhythm 13, e295–e324 (2016).
Priori, S. G., Napolitano, C., Giordano, U., Collisani, G. & Memmi, M. Brugada syndrome and sudden cardiac death in children. Lancet 355, 808–809 (2000).
Andorin, A. et al. Impact of clinical and genetic findings on the management of young patients with Brugada syndrome. Heart Rhythm 13, 1274–1282 (2016).
Tester, D. J. et al. A mechanism for sudden infant death syndrome (SIDS): stress-induced leak via ryanodine receptors. Heart Rhythm 4, 733–739 (2007).
Neubauer, J. et al. Post-mortem whole-exome analysis in a large sudden infant death syndrome cohort with a focus on cardiovascular and metabolic genetic diseases. Eur. J. Hum. Genet. 25, 404–409 (2017).
Brugada, R. et al. Sudden death associated with short-QT syndrome linked to mutations in HERG. Circulation 109, 30–35 (2004).
Arnestad, M. et al. Prevalence of long-QT syndrome gene variants in sudden infant death syndrome. Circulation 115, 361–367 (2007).
Wang, D. W. et al. Cardiac sodium channel dysfunction in sudden infant death syndrome. Circulation 115, 368–376 (2007).
Cronk, L. B. et al. Novel mechanism for sudden infant death syndrome: persistent late sodium current secondary to mutations in caveolin-3. Heart Rhythm 4, 161–166 (2007).
Van Norstrand, D. W. et al. Molecular and functional characterization of novel glycerol-3-phosphate dehydrogenase 1 like gene (GPD1-L) mutations in sudden infant death syndrome. Circulation 116, 2253–2259 (2007).
Tan, B.-H. et al. Sudden infant death syndrome-associated mutations in the sodium channel beta subunits. Heart Rhythm 7, 771–778 (2010).
Cheng, J. et al. Alpha1-syntrophin mutations identified in sudden infant death syndrome cause an increase in late cardiac sodium current. Circ. Arrhythm. Electrophysiol. 2, 667–676 (2009).
Hu, D. et al. A novel rare variant in SCN1Bb linked to Brugada syndrome and SIDS by combined modulation of Na(v)1.5 and K(v)4.3 channel currents. Heart Rhythm 9, 760–769 (2012).
Behr, E. R. et al. Role of common and rare variants in SCN10A: results from the Brugada syndrome QRS locus gene discovery collaborative study. Cardiovasc. Res. 106, 520–529 (2015).
Tester, D. J. & Ackerman, M. J. Sudden infant death syndrome: how significant are the cardiac channelopathies? Cardiovasc. Res. 67, 388–396 (2005).
Tester, D. J. et al. Loss-of-function mutations in the KCNJ8-encoded Kir6.1 K(ATP) channel and sudden infant death syndrome. Circ. Cardiovasc. Genet. 4, 510–515 (2011).
Giudicessi, J. R. et al. Novel mutations in the KCND3-encoded Kv4.3 K+ channel associated with autopsy-negative sudden unexplained death. Hum. Mutat. 33, 989–997 (2012).
Hertz, C. L. et al. Genetic investigations of sudden unexpected deaths in infancy using next-generation sequencing of 100 genes associated with cardiac diseases. Eur. J. Hum. Genet. 24, 817–822 (2016).
Crotti, L. et al. Calmodulin mutations associated with recurrent cardiac arrest in infants. Circulation 127, 1009–1017 (2013).
Van Norstrand, D. W. et al. Connexin43 mutation causes heterogeneous gap junction loss and sudden infant death. Circulation 125, 474–481 (2012).
Evans, A., Bagnall, R. D., Duflou, J. & Semsarian, C. Postmortem review and genetic analysis in sudden infant death syndrome: an 11-year review. Hum. Pathol. 44, 1730–1736 (2013).
Plant, L. D. et al. A common cardiac sodium channel variant associated with sudden infant death in African Americans, SCN5A S1103Y. J. Clin. Invest. 116, 430–435 (2006).
Brion, M. et al. Sarcomeric gene mutations in sudden infant death syndrome (SIDS). Forensic Sci. Int. 219, 278–281 (2012).
Wedekind, H. et al. Sudden infant death syndrome and long QT syndrome: an epidemiological and genetic study. Int. J. Legal Med. 120, 129–137 (2006).
Otagiri, T. et al. Cardiac ion channel gene mutations in sudden infant death syndrome. Pediatr. Res. 64, 482–487 (2008).
Millat, G. et al. Contribution of long-QT syndrome genetic variants in sudden infant death syndrome. Pediatr. Cardiol. 30, 502–509 (2009).
Winkel, B. G. et al. The role of the sodium current complex in a nonreferred nationwide cohort of sudden infant death syndrome. Heart Rhythm 12, 1241–1249 (2015).
Anderson, J. H., Tester, D. J., Will, M. L. & Ackerman, M. J. Whole-exome molecular autopsy after exertion-related sudden unexplained death in the young. Circ. Cardiovasc. Genet. 9, 259–265 (2016).
Ackerman, M. J. Genetic purgatory and the cardiac channelopathies: exposing the variants of uncertain/unknown significance issue. Heart Rhythm 12, 2325–2331 (2015).
Ackerman, M. J. et al. HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies this document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Heart Rhythm 8, 1308–1339 (2011).
Wong, L. C. H. & Behr, E. R. Sudden unexplained death in infants and children: the role of undiagnosed inherited cardiac conditions. Europace 16, 1706–1713 (2014).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Probst, V. et al. Clinical aspects and prognosis of Brugada syndrome in children. Circulation 115, 2042–2048 (2007).
Skinner, J. R. et al. Brugada syndrome masquerading as febrile seizures. Pediatrics 119, e1206–e1211 (2007).
Behr, E. R. & Roden, D. Drug-induced arrhythmia: pharmacogenomic prescribing? Eur. Heart J. 34, 89–95 (2013).
Skinner, J. R. & Van Hare, G. F. Routine ECG screening in infancy and early childhood should not be performed. Heart Rhythm 11, 2322–2327 (2014).
Saul, J. P., Schwartz, P. J., Ackerman, M. J. & Triedman, J. K. Rationale and objectives for ECG screening in infancy. Heart Rhythm 11, 2316–2321 (2014).
Saul, J. P., Schwartz, P. J., Ackerman, M. J. & Triedman, J. K. Neonatal ECG screening: opinions and facts. Heart Rhythm 12, 610–611 (2015).
Schwartz, P. J. & Dagradi, F. Management of survivors of cardiac arrest — the importance of genetic investigation. Nat. Rev. Cardiol. 13, 560–566 (2016).
Petrikin, J. E., Willig, L. K., Smith, L. D. & Kingsmore, S. F. Rapid whole genome sequencing and precision neonatology. Semin. Perinatol. 39, 623–631 (2015).
Christiansen, M. et al. Mutations in the HERG K+-ion channel: a novel link between long QT syndrome and sudden infant death syndrome. Am. J. Cardiol. 95, 433–434 (2005).
Nof, E. et al. A common single nucleotide polymorphism can exacerbate long-QT type 2 syndrome leading to sudden infant death. Circ. Cardiovasc. Genet. 3, 199–206 (2010).
Kato, K. et al. Cardiac channelopathies associated with infantile fatal ventricular arrhythmias: from the cradle to the bench. J. Cardiovasc. Electrophysiol. 25, 66–73 (2014).
Sauer, C. W. & Marc-Aurele, K. L. A. Neonate with susceptibility to long QT syndrome type 6 who presented with ventricular fibrillation and sudden unexpected infant death. Am. J. Case Rep. 17, 544–548 (2016).
Yang, Y. et al. Identification of a Kir3.4 mutation in congenital long QT syndrome. Am. J. Hum. Genet. 86, 872–880 (2010).
Skinner, J. R. et al. Near-miss SIDS due to Brugada syndrome. Arch. Dis. Child. 90, 528–529 (2005).
Turillazzi, E. et al. Heterozygous nonsense SCN5A mutation W822X explains a simultaneous sudden infant death syndrome. Virchows Arch. 453, 209–216 (2008).
Huang, H. et al. Biophysical characterization of a new SCN5A mutation S1333Y in a SIDS infant linked to long QT syndrome. FEBS Lett. 583, 890–896 (2009).
Wang, D. W. et al. Malignant perinatal variant of long-QT syndrome caused by a profoundly dysfunctional cardiac sodium channel. Circ. Arrhythm. Electrophysiol. 1, 370–378 (2008).
Bankston, J. R. et al. A novel and lethal de novo LQT-3 mutation in a newborn with distinct molecular pharmacology and therapeutic response. PLoS ONE 2, e1258 (2007).
Acknowledgements
A.-E.B.'s research work is supported by a research grant of the ESC and by research funds from Cardiac Risk in the Young.
Author information
Authors and Affiliations
Contributions
A.-E.B. researched data for the article and wrote the manuscript. M.J.A. and E.R.B. conceptualized and designed the article. All authors provided substantial contribution to the discussion of contents and critically reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.J.A. is a consultant for Boston Scientific, Gilead Sciences, Invitae, Medtronic, MyoKardia, and St. Jude Medical; M.J.A. and Mayo Clinic received royalties from Transgenomic for their FAMILION-LQTS and FAMILION-CPVT genetic tests, but none of these entities was involved in this article. The other authors declare no competing interests.
Related links
Supplementary information
Supplementary information S1 (table)
Variants in arrhythmia syndrome-related genes reported in SIDS cases: Na+ channel (PDF 151 kb)
Supplementary information S2 (table)
Variants in arrhythmia syndrome-related genes reported in SIDS cases: K+ channels (PDF 142 kb)
Supplementary information S3 (table)
Variants in arrhythmia syndrome-related genes reported in SIDS cases: Ca2+-handling and other ion channels (PDF 134 kb)
Supplementary information S4 (table)
Variants in inherited cardiomyopathy-related genes reported in SIDS cohort studies (PDF 134 kb)
Supplementary information S5 (box)
Analytical methods for Table 3 (PDF 96 kb)
Glossary
- De novo genetic variants
-
A genetic variant that is present for the first time in one family member as a result of a genetic alteration in a germ cell of one of the parents, or a variant that arises in the fertilized egg itself during early embryogenesis.
- Loss-of-function mutations
-
A mutation that results in reduced or abolished protein function.
- Gain-of-function mutations
-
A mutation that results in an increased protein function.
- Whole-exome sequencing
-
Sequencing of the protein-coding (exonic) regions in the genome and related sequences, which represents approximately 1% of the DNA sequence.
- Next-generation sequencing
-
A high-throughput method used to determine a portion of the nucleotide sequence of an individual's genome; utilizes sequencing technologies capable of processing multiple DNA sequences in parallel.
- Sanger sequencing
-
A low-throughput method used to determine a portion of the nucleotide sequence of an individual's genome; involves polymerase chain reaction (PCR) amplification of genetic regions of interest followed by sequencing of PCR products.
- Diagnostic yield
-
The likelihood that a test or procedure will provide the information needed to establish a diagnosis.
- Linkage studies
-
Gene-hunting technique that traces patterns of disease in families at high risk to locate a disease-causing gene by identifying genetic markers of known chromosomal location that are co-inherited with the trait of interest.
- Minor allele frequency
-
Frequency at which the second most common allele occurs in a given population.
- Mendelian inherited disorders
-
Disorders caused by gene mutations that are transmitted from parents to children according to Mendel's laws.
- Segregation studies
-
Process of fitting formal genetic models to data on expressed disease characteristics (phenotype) in biological family members to determine the most probable mode of inheritance for the trait or disease.
- Proband
-
The individual through whom a family with a genetic disorder is ascertained; also called a propositus.
- Penetrance
-
A characteristic of a genotype; refers to the likelihood that a clinical condition will occur when a particular genotype is present.
- Trio analyses
-
Genetic analysis of a case and two parents to determine whether a genetic variant is transmitted or occurred de novo.
Rights and permissions
About this article
Cite this article
Baruteau, AE., Tester, D., Kapplinger, J. et al. Sudden infant death syndrome and inherited cardiac conditions. Nat Rev Cardiol 14, 715–726 (2017). https://doi.org/10.1038/nrcardio.2017.129
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2017.129
This article is cited by
-
Sudden cardiac arrest in infants and children: proposal for a diagnostic workup to identify the etiology. An 18-year multicenter evaluation in the Netherlands
European Journal of Pediatrics (2023)
-
Genetic innovations and our understanding of stillbirth
Human Genetics (2020)
-
A standardized postmortem protocol to assess the real burden of sudden infant death syndrome
Virchows Archiv (2020)
-
Evidence for an association of interferon gene variants with sudden infant death syndrome
International Journal of Legal Medicine (2019)
-
Peri-mortem evaluation of infants who die without a diagnosis: focus on advances in genomic technology
Journal of Perinatology (2018)