Key Points
-
Advances in invasive and noninvasive imaging of the coronary arteries have progressed our understanding of the mechanisms underlying the precipitation of acute myocardial infarction, a leading cause of death worldwide
-
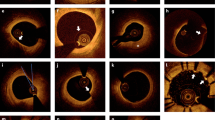
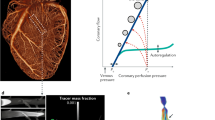
Imaging can provide assessments of luminal stenosis, plaque burden, plaque characteristics, disease activity, and ischaemia, generating complementary and potentially synergistic information about coronary atherosclerosis
-
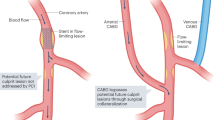
Advances in imaging offer the potential to identify individuals at highest risk of myocardial infarction — the 'vulnerable patient'; these individuals might then be targeted with aggressive therapy to prevent future events
-
The next challenge is to demonstrate that these imaging techniques can have a beneficial and cost-effective effect on cardiovascular outcomes for patients with coronary artery disease
Abstract
Coronary atherosclerosis and the precipitation of acute myocardial infarction are highly complex processes, which makes accurate risk prediction challenging. Rapid developments in invasive and noninvasive imaging technologies now provide us with detailed, exquisite images of the coronary vasculature that allow direct investigation of a wide range of these processes. These modalities include sophisticated assessments of luminal stenoses and myocardial perfusion, complemented by novel measures of the atherosclerotic plaque burden, adverse plaque characteristics, and disease activity. Together, they can provide comprehensive, individualized assessments of coronary atherosclerosis as it occurs in patients. Not only can this information provide important pathological insights, but it can also potentially be used to guide personalized treatment decisions. In this Review, we describe the latest advances in both established and emerging imaging techniques, focusing on the strengths and weakness of each approach. Moreover, we discuss how these technological advances might be translated from attractive images into novel imaging strategies and definite improvements in clinical risk prediction and patient outcomes. This process will not be easy, and the many potential barriers and difficulties are also reviewed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Yahagi, K. et al. Pathophysiology of native coronary, vein graft, and in-stent atherosclerosis. Nat. Rev. Cardiol. 13, 79–98 (2016).
Libby, P. Mechanisms of acute coronary syndromes and their implications for therapy. N. Engl. J. Med. 368, 2004–2013 (2013).
Brown, A. J. et al. Role of biomechanical forces in the natural history of coronary atherosclerosis. Nat. Rev. Cardiol. 13, 210–220 (2016).
Stary, H. C. et al. A definition of initial, fatty streak, and intermediate lesions of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 89, 2462–2478 (1994).
Bentzon, J. F., Otsuka, F., Virmani, R. & Falk, E. Mechanisms of plaque formation and rupture. Circ. Res. 114, 1852–1866 (2014).
Aikawa, E. et al. Osteogenesis associates with inflammation in early-stage atherosclerosis evaluated by molecular imaging in vivo. Circulation 116, 2841–2850 (2007).
Proudfoot, D. et al. Apoptosis regulates human vascular calcification in vitro: evidence for initiation of vascular calcification by apoptotic bodies. Circ. Res. 87, 1055–1062 (2000).
Hutcheson, J. D. et al. Genesis and growth of extracellular-vesicle-derived microcalcification in atherosclerotic plaques. Nat. Mater. 15, 335–343 (2016).
Farb, A. et al. Coronary plaque erosion without rupture into a lipid core. A frequent cause of coronary thrombosis in sudden coronary death. Circulation 93, 1354–1363 (1996).
Sugiyama, S. et al. Hypochlorous acid, a macrophage product, induces endothelial apoptosis and tissue factor expression: involvement of myeloperoxidase-mediated oxidant in plaque erosion and thrombogenesis. Arterioscler. Thromb. Vasc. Biol. 24, 1309–1314 (2004).
Hachamovitch, R. et al. Incremental prognostic value of myocardial perfusion single photon emission computed tomography for the prediction of cardiac death: differential stratification for risk of cardiac death and myocardial infarction. Circulation 97, 535–543 (1998).
Hachamovitch, R., Hayes, S. W., Friedman, J. D., Cohen, I. & Berman, D. S. Comparison of the short-term survival benefit associated with revascularization compared with medical therapy in patients with no prior coronary artery disease undergoing stress myocardial perfusion single photon emission computed tomography. Circulation 107, 2900–2907 (2003).
Mock, M. B. et al. Survival of medically treated patients in the coronary artery surgery study (CASS) registry. Circulation 66, 562–568 (1982).
Emond, M. et al. Long-term survival of medically treated patients in the Coronary Artery Surgery Study (CASS) Registry. Circulation 90, 2645–2657 (1994).
Tonino, P. A. L. et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N. Engl. J. Med. 360, 213–224 (2009).
Kern, M. J. & Samady, H. Current concepts of integrated coronary physiology in the catheterization laboratory. J. Am. Coll. Cardiol. 55, 173–185 (2010).
US National Library of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT02328820 (2014).
Miller, J. M. et al. Diagnostic performance of coronary angiography by 64-row CT. N. Engl. J. Med. 359, 2324–2336 (2008).
Vanhoenacker, P. K. et al. Diagnostic performance of multidetector CT angiography for assessment of coronary artery disease: meta-analysis. Radiology 244, 419–428 (2007).
Min, J. K. et al. Prognostic value of multidetector coronary computed tomographic angiography for prediction of all-cause mortality. J. Am. Coll. Cardiol. 50, 1161–1170 (2007).
Pundziute, G. et al. Prognostic value of multislice computed tomography coronary angiography in patients with known or suspected coronary artery disease. J. Am. Coll. Cardiol. 49, 62–70 (2007).
Gilard, M. et al. Midterm prognosis of patients with suspected coronary artery disease and normal multislice computed tomographic findings: a prospective management outcome study. Arch. Intern. Med. 167, 1686–1689 (2007).
Hadamitzky, M. et al. Prognostic value of coronary computed tomography angiography during 5 years of follow-up in patients with suspected coronary artery disease. Eur. Heart J. 34, 3277–3285 (2013).
Ostrom, M. P. et al. Mortality incidence and the severity of coronary atherosclerosis assessed by computed tomography angiography. J. Am. Coll. Cardiol. 52, 1335–1343 (2008).
Danad, I., Fayad, Z. A., Willemink, M. J. & Min, J. K. New applications of cardiac computed tomography: dual-energy, spectral, and molecular CT imaging. JACC Cardiovasc. Imaging 8, 710–723 (2015).
Andreini, D. et al. Diagnostic accuracy of rapid kilovolt peak-switching dual-energy CT coronary angiography in patients with a high calcium score. JACC Cardiovasc. Imaging 8, 746–748 (2015).
Budoff, M. J. et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) trial. J. Am. Coll. Cardiol. 52, 1724–1732 (2008).
Meijboom, W. B. et al. Diagnostic accuracy of 64-slice computed tomography coronary angiography: a prospective, multicenter, multivendor study. J. Am. Coll. Cardiol. 52, 2135–2144 (2008).
Hulten, E. et al. Outcomes after coronary computed tomography angiography in the emergency department: a systematic review and meta-analysis of randomized, controlled trials. J. Am. Coll. Cardiol. 61, 880–892 (2013).
Douglas, P. S. et al. Outcomes of anatomical versus functional testing for coronary artery disease. N. Engl. J. Med. 372, 1291–1300 (2015).
SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet 385, 2383–2391 (2015).
Williams, M. C. et al. Use of coronary computed tomographic angiography to guide management of patients with coronary disease. J. Am. Coll. Cardiol. 67, 1759–1768 (2016).
Li, S. et al. The diagnostic performance of CT-derived fractional flow reserve for evaluation of myocardial ischaemia confirmed by invasive fractional flow reserve: a meta-analysis. Clin. Radiol. 70, 476–486 (2015).
Douglas, P. S. et al. Clinical outcomes of fractional flow reserve by computed tomographic angiography-guided diagnostic strategies versus usual care in patients with suspected coronary artery disease: the prospective longitudinal trial of FFRct: outcome and resource impacts study. Eur. Heart J. 36, 3359–3367 (2015).
Kim, K.-H. et al. A novel noninvasive technology for treatment planning using virtual coronary stenting and computed tomography-derived computed fractional flow reserve. JACC Cardiovasc. Interv. 7, 72–78 (2014).
Dweck, M. R., Puntman, V., Vesey, A. T., Fayad, Z. A. & Nagel, E. MR imaging of coronary arteries and plaques. JACC. Cardiovasc. Imaging 9, 306–316 (2016).
Yonezawa, M. et al. Quantitative analysis of 1.5-T whole-heart coronary MR angiograms obtained with 32-channel cardiac coils: a comparison with conventional quantitative coronary angiography. Radiology 271, 356–364 (2014).
American College of Cardiology Foundation Task Force on Expert Consensus Documents et al. ACCF/ACR/AHA/NASCI/SCMR 2010 expert consensus document on cardiovascular magnetic resonance: a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents. J. Am. Coll. Cardiol. 55, 2614–2662 (2010).
Flotats, A. et al. Hybrid cardiac imaging: SPECT/CT and PET/CT. A joint position statement by the European Association of Nuclear Medicine (EANM), the European Society of Cardiac Radiology (ESCR) and the European Council of Nuclear Cardiology (ECNC). Eur. J. Nucl. Med. Mol. Imaging 38, 201–212 (2011).
Santana, C. A. et al. Diagnostic performance of fusion of myocardial perfusion imaging (MPI) and computed tomography coronary angiography. J. Nucl. Cardiol. 16, 201–211 (2009).
Sato, A. et al. Incremental value of combining 64-slice computed tomography angiography with stress nuclear myocardial perfusion imaging to improve noninvasive detection of coronary artery disease. J. Nucl. Cardiol. 17, 19–26 (2010).
Gaemperli, O., Bengel, F. M. & Kaufmann, P. A. Cardiac hybrid imaging. Eur. Heart J. 32, 2100–2108 (2011).
van Werkhoven, J. M. et al. Prognostic value of multislice computed tomography and gated single-photon emission computed tomography in patients with suspected coronary artery disease. J. Am. Coll. Cardiol. 53, 623–632 (2009).
Greenwood, J. P. et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): a prospective trial. Lancet 379, 453–460 (2012).
Topol, E. J. & Nissen, S. E. Our preoccupation with coronary luminology. The dissociation between clinical and angiographic findings in ischemic heart disease. Circulation 92, 2333–2342 (1995).
Ambrose, J. A. et al. Angiographic progression of coronary artery disease and the development of myocardial infarction. J. Am. Coll. Cardiol. 12, 56–62 (1988).
Falk, E., Shah, P. K. & Fuster, V. Coronary plaque disruption. Circulation 92, 657–671 (1995).
Alderman, E. L. et al. Five-year angiographic follow-up of factors associated with progression of coronary artery disease in the Coronary Artery Surgery Study (CASS). CASS Participating Investigators and Staff. J. Am. Coll. Cardiol. 22, 1141–1154 (1993).
Boden, W. E. et al. Optimal medical therapy with or without PCI for stable coronary disease. N. Engl. J. Med. 356, 1503–1516 (2007).
BARI 2D Study Group. A randomized trial of therapies for type 2 diabetes and coronary artery disease. N. Engl. J. Med. 360, 2503–2515 (2009).
De Bruyne, B. et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N. Engl. J. Med. 367, 991–1001 (2012).
De Bruyne, B. et al. Fractional flow reserve-guided PCI for stable coronary artery disease. N. Engl. J. Med. 371, 1208–1217 (2014).
Park, H.-B. et al. Atherosclerotic plaque characteristics by CT angiography identify coronary lesions that cause ischemia: a direct comparison to fractional flow reserve. JACC. Cardiovasc. Imaging 8, 1–10 (2015).
Dey, D. et al. Relationship between quantitative adverse plaque features from coronary computed tomography angiography and downstream impaired myocardial flow reserve by 13N-ammonia positron emission tomography: a pilot study. Circ. Cardiovasc. Imaging 8, e003255 (2015).
Hermiller, J. B. et al. Unrecognized left main coronary artery disease in patients undergoing interventional procedures. Am. J. Cardiol. 71, 173–176 (1993).
Nicholls, S. J. et al. Intravascular ultrasound-derived measures of coronary atherosclerotic plaque burden and clinical outcome. J. Am. Coll. Cardiol. 55, 2399–2407 (2010).
Stone, P. H. et al. Prediction of progression of coronary artery disease and clinical outcomes using vascular profiling of endothelial shear stress and arterial plaque characteristics: the PREDICTION Study. Circulation 126, 172–181 (2012).
Nissen, S. E. et al. Effect of intensive compared with moderate lipid-lowering therapy on progression of coronary atherosclerosis: a randomized controlled trial. JAMA 291, 1071–1080 (2004).
Erbel, R. et al. Coronary risk stratification, discrimination, and reclassification improvement based on quantification of subclinical coronary atherosclerosis: the Heinz Nixdorf Recall study. J. Am. Coll. Cardiol. 56, 1397–1406 (2010).
Yeboah, J. et al. Comparison of novel risk markers for improvement in cardiovascular risk assessment in intermediate-risk individuals. JAMA 308, 788–795 (2012).
Nasir, K. et al. Implications of coronary artery calcium testing among statin candidates according to American College of Cardiology/American Heart Association Cholesterol Management Guidelines: MESA (Multi-Ethnic Study of Atherosclerosis). J. Am. Coll. Cardiol. 66, 1657–1668 (2015).
Berman, D. S., Arnson, Y. & Rozanski, A. Coronary artery calcium scanning: the Agatston score and beyond. JACC. Cardiovasc. Imaging (in the press).
Baber, U. et al. Prevalence, impact, and predictive value of detecting subclinical coronary and carotid atherosclerosis in asymptomatic adults: the BioImage study. J. Am. Coll. Cardiol. 65, 1065–1074 (2015).
García-García, H. M., Gogas, B. D., Serruys, P. W. & Bruining, N. IVUS-based imaging modalities for tissue characterization: similarities and differences. Int. J. Cardiovasc. Imaging 27, 215–224 (2011).
Obaid, D. R. et al. Atherosclerotic plaque composition and classification identified by coronary computed tomography. Circ. Cardiovasc. Imaging 6, 655–664 (2013).
Stone, G. W. et al. A prospective natural-history study of coronary atherosclerosis. N. Engl. J. Med. 364, 226–235 (2011).
Calvert, P. A. et al. Association between IVUS findings and adverse outcomes in patients with coronary artery disease the VIVA (VH-IVUS in Vulnerable Atherosclerosis) Study. JACC Cardiovasc. Imaging 4, 894–901 (2011).
Cheng, J. M. et al. In vivo detection of high-risk coronary plaques by radiofrequency intravascular ultrasound and cardiovascular outcome: results of the ATHEROREMO-IVUS study. Eur. Heart J. 35, 639–647 (2014).
Bezerra, H. G., Costa, M. A., Guagliumi, G., Rollins, A. M. & Simon, D. I. Intracoronary optical coherence tomography: a comprehensive review clinical and research applications. JACC Cardiovasc. Interv. 2, 1035–1046 (2009).
Suter, M. J. et al. Intravascular optical imaging technology for investigating the coronary artery. JACC Cardiovasc. Imaging 4, 1022–1039 (2011).
Tearney, G. J. et al. Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: a report from the International Working Group for Intravascular Optical Coherence Tomography Standardization and Validation. J. Am. Coll. Cardiol. 59, 1058–1072 (2012).
Kubo, T. et al. Assessment of culprit lesion morphology in acute myocardial infarction: ability of optical coherence tomography compared with intravascular ultrasound and coronary angioscopy. J. Am. Coll. Cardiol. 50, 933–939 (2007).
Prati, F. et al. OCT-based diagnosis and management of STEMI associated with intact fibrous cap. JACC Cardiovasc. Imaging 6, 283–287 (2013).
Kim, B.-K. et al. Optical coherence tomography-based evaluation of malapposed strut coverage after drug-eluting stent implantation. Int. J. Cardiovasc. Imaging 28, 1887–1894 (2012).
Hattori, K. et al. Impact of statin therapy on plaque characteristics as assessed by serial OCT, grayscale and integrated backscatter-IVUS. JACC Cardiovasc. Imaging 5, 169–177 (2012).
Vergallo, R. et al. Endothelial shear stress and coronary plaque characteristics in humans: combined frequency-domain optical coherence tomography and computational fluid dynamics study. Circ. Cardiovasc. Imaging 7, 905–911 (2014).
Tearney, G. J. et al. Quantification of macrophage content in atherosclerotic plaques by optical coherence tomography. Circulation 107, 113–119 (2003).
Caplan, J. D., Waxman, S., Nesto, R. W. & Muller, J. E. Near-infrared spectroscopy for the detection of vulnerable coronary artery plaques. J. Am. Coll. Cardiol. 47, C92–96 (2006).
Waxman, S., Ishibashi, F. & Caplan, J. D. Rationale and use of near-infrared spectroscopy for detection of lipid-rich and vulnerable plaques. J. Nucl. Cardiol. 14, 719–728 (2007).
Waxman, S. et al. In vivo validation of a catheter-based near-infrared spectroscopy system for detection of lipid core coronary plaques: initial results of the SPECTACL study. JACC Cardiovasc. Imaging 2, 858–868 (2009).
Gardner, C. M. et al. Detection of lipid core coronary plaques in autopsy specimens with a novel catheter-based near-infrared spectroscopy system. JACC Cardiovasc. Imaging 1, 638–648 (2008).
Puri, R. et al. Near-infrared spectroscopy enhances intravascular ultrasound assessment of vulnerable coronary plaque. Arterioscler. Thromb. Vasc. Biol. 35, 2423–2431 (2015).
Madder, R. D. et al. Detection by near-infrared spectroscopy of large lipid core plaques at culprit sites in patients with acute ST-segment elevation myocardial infarction. JACC Cardiovasc. Interv. 6, 838–846 (2013).
US National Library of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT02033694 (2016).
Leber, A. W. et al. Accuracy of 64-slice computed tomography to classify and quantify plaque volumes in the proximal coronary system: a comparative study using intravascular ultrasound. J. Am. Coll. Cardiol. 47, 672–677 (2006).
Øvrehus, K. A. et al. Reproducibility of semi-automatic coronary plaque quantification in coronary CT angiography with sub-mSv radiation dose. J. Cardiovasc. Comput. Tomogr. 10, 115–120 (2016).
Motoyama, S. et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J. Am. Coll. Cardiol. 50, 319–326 (2007).
Pundziute, G. et al. Evaluation of plaque characteristics in acute coronary syndromes: non-invasive assessment with multi-slice computed tomography and invasive evaluation with intravascular ultrasound radiofrequency data analysis. Eur. Heart J. 29, 2373–2381 (2008).
Hoffmann, U. et al. Noninvasive assessment of plaque morphology and composition in culprit and stable lesions in acute coronary syndrome and stable lesions in stable angina by multidetector computed tomography. J. Am. Coll. Cardiol. 47, 1655–1662 (2006).
Motoyama, S. et al. Plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up. J. Am. Coll. Cardiol. 66, 337–346 (2015).
Nakazato, R. et al. Quantification and characterisation of coronary artery plaque volume and adverse plaque features by coronary computed tomographic angiography: a direct comparison to intravascular ultrasound. Eur. Radiol. 23, 2109–2117 (2013).
Dey, D. et al. Automated three-dimensional quantification of noncalcified coronary plaque from coronary CT angiography: comparison with intravascular US. Radiology 257, 516–522 (2010).
Boogers, M. J. et al. Automated quantification of coronary plaque with computed tomography: comparison with intravascular ultrasound using a dedicated registration algorithm for fusion-based quantification. Eur. Heart J. 33, 1007–1016 (2012).
Fayad, Z. A. et al. Noninvasive in vivo human coronary artery lumen and wall imaging using black-blood magnetic resonance imaging. Circulation 102, 506–510 (2000).
Ehara, S. et al. Hyperintense plaque identified by magnetic resonance imaging relates to intracoronary thrombus as detected by optical coherence tomography in patients with angina pectoris. Eur. Heart J. Cardiovasc. Imaging 13, 394–399 (2012).
Jansen, C. H. P. et al. Detection of intracoronary thrombus by magnetic resonance imaging in patients with acute myocardial infarction. Circulation 124, 416–424 (2011).
Noguchi, T. et al. High-intensity signals in coronary plaques on noncontrast T1-weighted magnetic resonance imaging as a novel determinant of coronary events. J. Am. Coll. Cardiol. 63, 989–999 (2014).
Davies, M. J., Bland, J. M., Hangartner, J. R., Angelini, A. & Thomas, A. C. Factors influencing the presence or absence of acute coronary artery thrombi in sudden ischaemic death. Eur. Heart J. 10, 203–208 (1989).
Mann, J. & Davies, M. J. Mechanisms of progression in native coronary artery disease: role of healed plaque disruption. Heart 82, 265–268 (1999).
Arbab-Zadeh, A. & Fuster, V. The myth of the 'vulnerable plaque': transitioning from a focus on individual lesions to atherosclerotic disease burden for coronary artery disease risk assessment. J. Am. Coll. Cardiol. 65, 846–855 (2015).
Vergallo, R. et al. Pancoronary plaque vulnerability in patients with acute coronary syndrome and ruptured culprit plaque: a 3-vessel optical coherence tomography study. Am. Heart J. 167, 59–67 (2014).
Kubo, T. et al. The dynamic nature of coronary artery lesion morphology assessed by serial virtual histology intravascular ultrasound tissue characterization. J. Am. Coll. Cardiol. 55, 1590–1597 (2010).
Jaffer, F. A., Libby, P. & Weissleder, R. Optical and multimodality molecular imaging: insights into atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 29, 1017–1024 (2009).
Vinegoni, C. et al. Indocyanine green enables near-infrared fluorescence imaging of lipid-rich, inflamed atherosclerotic plaques. Sci. Transl. Med. 3, 84ra45 (2011).
Thukkani, A. K. & Jaffer, F. A. Intravascular near-infrared fluorescence molecular imaging of atherosclerosis. Am. J. Nucl. Med. Mol. Imaging 3, 217–231 (2013).
Jaffer, F. A. et al. Real-time catheter molecular sensing of inflammation in proteolytically active atherosclerosis. Circulation 118, 1802–1809 (2008).
Jaffer, F. A. et al. Two-dimensional intravascular near-infrared fluorescence molecular imaging of inflammation in atherosclerosis and stent-induced vascular injury. J. Am. Coll. Cardiol. 57, 2516–2526 (2011).
Hara, T. et al. Intravascular fibrin molecular imaging improves the detection of unhealed stents assessed by optical coherence tomography in vivo. Eur. Heart J. http://dx.doi.org/10.1093/eurheartj/ehv677 (2015).
Jenkins, W. S. A. et al. Valvular 18F-fluoride and 18F-fluorodeoxyglucose uptake predict disease progression and clinical outcome in patients with aortic stenosis. J. Am. Coll. Cardiol. 66, 1200–1201 (2015).
Dweck, M. R. et al. Aortic stenosis, atherosclerosis, and skeletal bone: is there a common link with calcification and inflammation? Eur. Heart J. 34, 1567–1574 (2013).
Adamson, P. D., Newby, D. E. & Dweck, M. R. Translational coronary atherosclerosis imaging with PET. Cardiol. Clin. 34, 179–186 (2016).
Adamson, P. D., Dweck, M. R. & Newby, D. E. The vulnerable atherosclerotic plaque: in vivo identification and potential therapeutic avenues. Heart 101, 1755–1766 (2015).
Rudd, J. H. F. et al. Imaging atherosclerotic plaque inflammation by fluorodeoxyglucose with positron emission tomography: ready for prime time? J. Am. Coll. Cardiol. 55, 2527–2535 (2010).
Folco, E. J. et al. Hypoxia but not inflammation augments glucose uptake in human macrophages. J. Am. Coll. Cardiol. 58, 603–614 (2011).
Joshi, N. V. et al. Systemic atherosclerotic inflammation following acute myocardial infarction: myocardial infarction begets myocardial infarction. J. Am. Heart Assoc. 4, e001956 (2015).
Tahara, N. et al. Simvastatin attenuates plaque inflammation: evaluation by fluorodeoxyglucose positron emission tomography. J. Am. Coll. Cardiol. 48, 1825–1831 (2006).
Fayad, Z. A. et al. Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial. Lancet 378, 1547–1559 (2011).
Tawakol, A. et al. Effect of treatment for 12 weeks with rilapladib, a lipoprotein-associated phospholipase A2 inhibitor, on arterial inflammation as assessed with 18F-fluorodeoxyglucose-positron emission tomography imaging. J. Am. Coll. Cardiol. 63, 86–88 (2014).
Figueroa, A. L. et al. Measurement of arterial activity on routine FDG PET/CT images improves prediction of risk of future CV events. JACC Cardiovasc. Imaging 6, 1250–1259 (2013).
Moon, S.-H. et al. Carotid FDG uptake improves prediction of future cardiovascular events in asymptomatic individuals. JACC Cardiovasc. Imaging 8, 949–956 (2015).
US National Library of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT00738725 (2015).
Rogers, I. S. et al. Feasibility of FDG imaging of the coronary arteries: comparison between acute coronary syndrome and stable angina. JACC Cardiovasc. Imaging 3, 388–397 (2010).
Cheng, V. Y. et al. Coronary arterial 18F-FDG uptake by fusion of PET and coronary CT angiography at sites of percutaneous stenting for acute myocardial infarction and stable coronary artery disease. J. Nucl. Med. 53, 575–583 (2012).
Joshi, N. V. et al. 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: a prospective clinical trial. Lancet 383, 705–713 (2014).
Irkle, A. et al. Identifying active vascular microcalcification by 18F-sodium fluoride positron emission tomography. Nat. Commun. 6, 1–11 (2015).
Dweck, M. R. et al. 18F-sodium fluoride uptake is a marker of active calcification and disease progression in patients with aortic stenosis. Circ. Cardiovasc. Imaging 7, 371–378 (2014).
Dweck, M. R. et al. Coronary arterial 18F-sodium fluoride uptake. J. Am. Coll. Cardiol. 59, 1539–1548 (2012).
Dweck, M. R., Joshi, F. R., Newby, D. E. & Rudd, J. H. F. Noninvasive imaging in cardiovascular therapy: the promise of coronary arterial 18F-sodium fluoride uptake as a marker of plaque biology. Expert Rev. Cardiovasc. Ther. 10, 1075–1077 (2012).
Joshi, N. V., Vesey, A., Newby, D. E. & Dweck, M. R. Will 18F-sodium fluoride PET-CT imaging be the magic bullet for identifying vulnerable coronary atherosclerotic plaques? Curr. Cardiol. Rep. 16, 521–527 (2014).
US National Library of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT02278211 (2015).
Rubeaux, M. et al. Motion correction of 18F-sodium fluoride PET for imaging coronary atherosclerotic plaques. J. Nucl. Med. 57, 54–59 (2016).
Rominger, A. et al. In vivo imaging of macrophage activity in the coronary arteries using 68Ga-DOTATATE PET/CT: correlation with coronary calcium burden and risk factors. J. Nucl. Med. 51, 193–197 (2010).
Tahara, N. et al. 2-deoxy-2-[18F]fluoro-d-mannose positron emission tomography imaging in atherosclerosis. Nat. Med. 20, 215–219 (2014).
Vesey, A. T., Dweck, M. R. & Fayad, Z. A. Utility of combining PET and MR imaging of carotid plaque. Neuroimaging Clin. N. Am. 26, 55–68 (2016).
Achenbach, S. et al. Coronary computed tomography angiography with a consistent dose below 1 mSv using prospectively electrocardiogram-triggered high-pitch spiral acquisition. Eur. Heart J. 31, 340–346 (2010).
Acknowledgements
We thank Michelle Williams (Centre for Cardiovascular Science, University of Edinburgh, UK) and Charles Taylor (HeartFlow Inc., Redwood, California, USA) for providing images used as part of Fig. 1. M.R.D. and D.E.N. are supported by the British Heart Foundation (SS/CH/ 09/002/26360, FS/13/77/30488, SS/CH/09/002/2636, FS/14/78/31020, CH/09/002). M.R.D. is the recipient of the Sir Jules Thorn Award for Biomedical Research 2015. M.M. is supported by the Dowager Countess Eleanor Peel Trust (Rothwell-Jackson Travelling Fellowship, UK) and the Dickinson Trust Scholarship (UK). P.S. is supported by NIH/NHLBI R01 HL089765. D.D. is supported by research grants from the American Heart Association. D.D. and D.S.B. are supported by research grants from the American Heart Association and Adelson Family Foundation. Z.A.F. is supported by NIH/NHLBI R01 HL071021, NIH/NHLBI R01 HL128056, and NIH/NBIB R01 EB009638. D.E.N. is the recipient of a Wellcome Trust Senior Investigator Award (WT103782AIA).
Author information
Authors and Affiliations
Contributions
M.R.D. and D.B. discussed the content of the article, and M.R.D. and M.K.D. wrote the manuscript. All the authors reviewed/edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
Cedars-Sinai Medical Center (M.M., P.S., D.D., D.B.) licenses nuclear cardiology software to several instrumentation vendors. P.S. has received grant support from Siemens. D.E.N. is the chief investigator of the PREFFIR study (NCT02278211). The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Dweck, M., Doris, M., Motwani, M. et al. Imaging of coronary atherosclerosis — evolution towards new treatment strategies. Nat Rev Cardiol 13, 533–548 (2016). https://doi.org/10.1038/nrcardio.2016.79
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2016.79
This article is cited by
-
Clinical quantitative coronary artery stenosis and coronary atherosclerosis imaging: a Consensus Statement from the Quantitative Cardiovascular Imaging Study Group
Nature Reviews Cardiology (2023)
-
Imaging coronary and aortic microcalcification activity with 18F-sodium fluoride
Journal of Nuclear Cardiology (2022)
-
Let’s embrace optical imaging: a growing branch on the clinical molecular imaging tree
European Journal of Nuclear Medicine and Molecular Imaging (2021)
-
Hiding beyond plain sight: Textural analysis of positron emission tomography to identify high-risk plaques in carotid atherosclerosis
Journal of Nuclear Cardiology (2021)
-
A novel fluorescein-bisphosphonate based diagnostic tool for the detection of hydroxyapatite in both cell and tissue models
Scientific Reports (2018)