Key Points
-
Atherosclerotic plaques are not uniformly distributed throughout the coronary tree, indicating that local mechanical factors might determine plaque development and growth
-
Blood flow and particularly wall shear stress heavily influence endothelial function through diverse mechanisms
-
Biomechanical forces promote adverse changes in plaque composition, promoting a high-risk plaque phenotype that is more prone to rupture, and can result in sudden cardiac events
-
Plaque structural stress is determined by plaque composition, with plaque rupture occurring when plaque structural stress exceeds plaque strength
-
Prospective, observational studies suggest that integrating biomechanical parameters can improve our ability to identify the patients at highest risk of rapid disease progression or plaque rupture
Abstract
Atherosclerosis remains a major cause of morbidity and mortality worldwide, and a thorough understanding of the underlying pathophysiological mechanisms is crucial for the development of new therapeutic strategies. Although atherosclerosis is a systemic inflammatory disease, coronary atherosclerotic plaques are not uniformly distributed in the vascular tree. Experimental and clinical data highlight that biomechanical forces, including wall shear stress (WSS) and plaque structural stress (PSS), have an important role in the natural history of coronary atherosclerosis. Endothelial cell function is heavily influenced by changes in WSS, and longitudinal animal and human studies have shown that coronary regions with low WSS undergo increased plaque growth compared with high WSS regions. Local alterations in WSS might also promote transformation of stable to unstable plaque subtypes. Plaque rupture is determined by the balance between PSS and material strength, with plaque composition having a profound effect on PSS. Prospective clinical studies are required to ascertain whether integrating mechanical parameters with medical imaging can improve our ability to identify patients at highest risk of rapid disease progression or sudden cardiac events.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yeh, R. W. et al. Population trends in the incidence and outcomes of acute myocardial infarction. N. Engl. J. Med. 362, 2155–2165 (2010).
Celermajer, D. S., Chow, C. K., Marijon, E., Anstey, N. M. & Woo, K. S. Cardiovascular disease in the developing world: prevalences, patterns, and the potential of early disease detection. J. Am. Coll. Cardiol. 60, 1207–1216 (2012).
Libby, P., Ridker, P. M. & Hansson, G. K. Progress and challenges in translating the biology of atherosclerosis. Nature 473, 317–325 (2011).
Stary, H. C. et al. A definition of initial, fatty streak, and intermediate lesions of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 89, 2462–2478 (1994).
Kubo, T. et al. The dynamic nature of coronary artery lesion morphology assessed by serial virtual histology intravascular ultrasound tissue characterization. J. Am. Coll. Cardiol. 55, 1590–1597 (2010).
Raber, L. et al. Effect of high-intensity statin therapy on atherosclerosis in non-infarct-related coronary arteries (IBIS-4): a serial intravascular ultrasonography study. Eur. Heart J. 36, 490–500 (2015).
Calvert, P. A. et al. Association between IVUS findings and adverse outcomes in patients with coronary artery disease: the VIVA (VH-IVUS in Vulnerable Atherosclerosis) Study. JACC Cardiovasc. Imaging 4, 894–901 (2011).
Stone, G. W. et al. A prospective natural-history study of coronary atherosclerosis. N. Engl. J. Med. 364, 226–235 (2011).
Cheng, J. M. et al. In vivo detection of high-risk coronary plaques by radiofrequency intravascular ultrasound and cardiovascular outcome: results of the ATHEROREMO-IVUS study. Eur. Heart J. 35, 639–647 (2014).
Pedrigi, R. M. et al. Thin-cap fibroatheroma rupture is associated with a fine interplay of shear and wall stress. Arteriosclerosis Thromb. 34, 2224–2231 (2014).
Hahn, C. & Schwartz, M. A. Mechanotransduction in vascular physiology and atherogenesis. Nat. Rev. Mol. Cell Biol. 10, 53–62 (2009).
Falk, E., Shah, P. K. & Fuster, V. Coronary plaque disruption. Circulation 92, 657–671 (1995).
Chiu, J.-J. & Chien, S. Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol. Rev. 91, 327–387 (2011).
Soulis, J. V., Farmakis, T. M., Giannoglou, G. D. & Louridas, G. E. Wall shear stress in normal left coronary artery tree. J. Biomech. 39, 742–749 (2006).
Zarins, C. K. et al. Carotid bifurcation atherosclerosis. Quantitative correlation of plaque localization with flow velocity profiles and wall shear stress. Circ. Res. 53, 502–514 (1983).
Ku, D. N., Giddens, D. P., Zarins, C. K. & Glagov, S. Pulsatile flow and atherosclerosis in the human carotid bifurcation. Positive correlation between plaque location and low oscillating shear stress. Arteriosclerosis 5, 293–302 (1985).
Kwak, B. R. et al. Biomechanical factors in atherosclerosis: mechanisms and clinical implications. Eur. Heart J. 35, 3013–3020 (2014).
Taylor, C. A., Fonte, T. A. & Min, J. K. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J. Am. Coll. Cardiol. 61, 2233–2241 (2013).
Mejia, J., Mongrain, R. & Bertrand, O. F. Accurate prediction of wall shear stress in a stented artery: newtonian versus non-newtonian models. J. Biomech. Eng. 133, 074501 (2011).
Timmins, L. H. et al. Focal association between wall shear stress and clinical coronary artery disease progression. Ann. Biomed. Eng. 43, 94–106 (2015).
Morbiducci, U. et al. On the importance of blood rheology for bulk flow in hemodynamic models of the carotid bifurcation. J. Biomech. 44, 2427–2438 (2011).
Li, Y. et al. Impact of side branch modeling on computation of endothelial shear stress in coronary artery disease: coronary tree reconstruction by fusion of 3D angiography and OCT. J. Am. Coll. Cardiol. 66, 125–135 (2015).
Peiffer, V., Sherwin, S. J. & Weinberg, P. D. Does low and oscillatory wall shear stress correlate spatially with early atherosclerosis? A systematic review. Cardiovasc. Res. 99, 242–250 (2013).
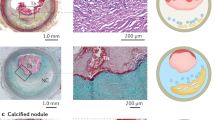
Teng, Z. et al. Coronary plaque structural stress is associated with plaque composition and subtype and higher in acute coronary syndrome: the BEACON I (Biomechanical Evaluation of Atheromatous Coronary Arteries) study. Circ. Cardiovasc. Imaging 7, 461–470 (2014).
Yuan, J. et al. Influence of material property variability on the mechanical behaviour of carotid atherosclerotic plaques: a 3D fluid-structure interaction analysis. Int. J. Numer. Method Biomed. Eng. 13, e02722 (2015).
Sadat, U., Teng, Z. & Gillard, J. H. Biomechanical structural stresses of atherosclerotic plaques. Expert Rev. Cardiovasc. Ther. 8, 1469–1481 (2010).
Fan, R. et al. Human coronary plaque wall thickness correlated positively with flow shear stress and negatively with plaque wall stress: an IVUS-based fluid-structure interaction multi-patient study. Biomed. Eng. Online 13, 13–32 (2014).
Ohayon, J. et al. Necrotic core thickness and positive arterial remodeling index: emergent biomechanical factors for evaluating the risk of plaque rupture. Am. J. Physiol. Heart Circ. Physiol. 295, H717–H727 (2008).
Hoshino, T. et al. Mechanical stress analysis of a rigid inclusion in distensible material: a model of atherosclerotic calcification and plaque vulnerability. Am. J. Physiol. Heart Circ. Physiol. 297, H802–H810 (2009).
Richardson, P. D., Davies, M. J. & Born, G. V. Influence of plaque configuration and stress distribution on fissuring of coronary atherosclerotic plaques. Lancet 2, 941–944 (1989).
Wanjare, M., Agarwal, N. & Gerecht, S. Biomechanical strain induces elastin and collagen production in human pluripotent stem cell derived vascular smooth muscle cells. 309, C271–C281 (2015).
Akyildiz, A. C., Speelman, L. & Gijsen, F. J. H. Mechanical properties of human atherosclerotic intima tissue. J. Biomech. 47, 773–783 (2014).
Holzapfel, G. A., Sommer, G., Gasser, C. T. & Regitnig, P. Determination of layer-specific mechanical properties of human coronary arteries with nonatherosclerotic intimal thickening and related constitutive modeling. Am. J. Physiol. Heart Circ. Physiol. 289, H2048–H2058 (2005).
Teng, Z. et al. Material properties of components in human carotid atherosclerotic plaques: a uniaxial extension study. Acta Biomater. 6, 00379–00371 (2014).
Walsh, M. T. et al. Uniaxial tensile testing approaches for characterisation of atherosclerotic plaques. J. Biomech. 47, 793–804 (2014).
Holzapfel, G. A., Mulvihill, J. J., Cunnane, E. M. & Walsh, M. T. Computational approaches for analyzing the mechanics of atherosclerotic plaques: a review. J. Biomech. 47, 859–869 (2014).
Yang, C. et al. In vivo IVUS-based 3D fluid–structure interaction models with cyclic bending and anisotropic vessel properties for human atherosclerotic coronary plaque mechanical analysis. IEEE Trans. Biomed. Eng. 56, 2420–2428 (2009).
Huang, Y. et al. The influence of computational strategy on prediction of mechanical stress in carotid atherosclerotic plaques: comparison of 2D structure-only, 3D structure-only, one-way and fully coupled fluid-structure interaction analyses. J. Biomech. 47, 1465–1471 (2014).
Cheruvu, P. K. et al. Frequency and distribution of thin-cap fibroatheroma and ruptured plaques in human coronary arteries: a pathologic study. J. Am. Coll. Cardiol. 50, 940–949 (2007).
Wykrzykowska, J. J. et al. Longitudinal distribution of plaque burden and necrotic core-rich plaques in nonculprit lesions of patients presenting with acute coronary syndromes. JACC Cardiovasc. Imaging 5, 006 (2012).
Suo, J., Oshinski, J. N. & Giddens, D. P. Blood flow patterns in the proximal human coronary arteries: relationship to atherosclerotic plaque occurrence. Mol. Cell. Biomech. 5, 9–18 (2008).
Caro, C. G., Fitz-Gerald, J. M. & Schroter, R. C. Arterial wall shear and distribution of early atheroma in man. Nature 223, 1159–1161 (1969).
White, S. J. et al. Characterization of the differential response of endothelial cells exposed to normal and elevated laminar shear stress. J. Cell. Physiol. 226, 2841–2848 (2011).
Cheng, C. et al. Shear stress-induced changes in atherosclerotic plaque composition are modulated by chemokines. J. Clin. Invest. 117, 616–626 (2007).
Cheng, C. et al. Atherosclerotic lesion size and vulnerability are determined by patterns of fluid shear stress. Circulation 113, 2744–2753 (2006).
Nam, D. et al. Partial carotid ligation is a model of acutely induced disturbed flow, leading to rapid endothelial dysfunction and atherosclerosis. Am. J. Physiol. Heart Circ. Physiol. 297, H1535–H1543 (2009).
Chen, Y. C. et al. A novel mouse model of atherosclerotic plaque instability for drug testing and mechanistic/therapeutic discoveries using gene and microRNA expression profiling. Circ. Res. 113, 252–265 (2013).
Gijsen, F., van der Giessen, A., van der Steen, A. & Wentzel, J. Shear stress and advanced atherosclerosis in human coronary arteries. J. Biomech. 46, 240–247 (2013).
Davies, P. F., Civelek, M., Fang, Y. & Fleming, I. The atherosusceptible endothelium: endothelial phenotypes in complex haemodynamic shear stress regions in vivo. Cardiovasc. Res. 99, 315–327 (2013).
Chaudhury, H. et al. c-Jun N-terminal kinase primes endothelial cells at atheroprone sites for apoptosis. Arterioscler. Thromb. Vasc. Biol. 30, 546–553 (2010).
Heo, K.-S. et al. PKCζ mediates disturbed flow-induced endothelial apoptosis via p53 SUMOylation. J. Cell Biol. 193, 867–884 (2011).
Magid, R. & Davies, P. F. Endothelial protein kinase C isoform identity and differential activity of PKCζ in an athero-susceptible region of porcine aorta. Circ. Res. 97, 443–449 (2005).
Civelek, M., Manduchi, E., Riley, R. J., Stoeckert, C. J. Jr & Davies, P. F. Chronic endoplasmic reticulum stress activates unfolded protein response in arterial endothelium in regions of susceptibility to atherosclerosis. Circ. Res. 105, 453–461 (2009).
Lee, D.-Y. et al. Role of histone deacetylases in transcription factor regulation and cell cycle modulation in endothelial cells in response to disturbed flow. Proc. Natl Acad. Sci. USA 109, 1967–1972 (2012).
Schober, A., Nazari-Jahantigh, M. & Weber, C. MicroRNA-mediated mechanisms of the cellular stress response in atherosclerosis. Nat. Rev. Cardiol. 12, 361–374 (2015).
Warboys, C. M. et al. Disturbed flow promotes endothelial senescence via a p53-dependent pathway. Arterioscler. Thromb. Vasc. Biol. 34, 985–995 (2014).
Ley, K., Laudanna, C., Cybulsky, M. I. & Nourshargh, S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat. Rev. Immunol. 7, 678–689 (2007).
Chiu, J. J. et al. Shear stress increases ICAM-1 and decreases VCAM-1 and E-selectin expressions induced by tumor necrosis factor-α in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 24, 73–79 (2004).
Nagel, T., Resnick, N., Atkinson, W. J., Dewey, C. F. Jr & Gimbrone, M. A. Jr. Shear stress selectively upregulates intercellular adhesion molecule-1 expression in cultured human vascular endothelial cells. J. Clin. Invest. 94, 885–891 (1994).
Sheikh, S., Rainger, G. E., Gale, Z., Rahman, M. & Nash, G. B. Exposure to fluid shear stress modulates the ability of endothelial cells to recruit neutrophils in response to tumor necrosis factor-α: a basis for local variations in vascular sensitivity to inflammation. Blood 102, 2828–2834 (2003).
Matharu, N. M. et al. Inflammatory responses of endothelial cells experiencing reduction in flow after conditioning by shear stress. J. Cell. Physiol. 216, 732–741 (2008).
Chappell, D. C., Varner, S. E., Nerem, R. M., Medford, R. M. & Alexander, R. W. Oscillatory shear stress stimulates adhesion molecule expression in cultured human endothelium. Circ. Res. 82, 532–539 (1998).
Schmitt, M. M. N. et al. Endothelial junctional adhesion molecule-A guides monocytes into flow-dependent predilection sites of atherosclerosis. Circulation 129, 66–76 (2014).
Hajra, L., Zhu, S. N. & Cybulsky, M. I. Modulation of NF-kappa B signal transduction in different regions of the arterial tree. FASEB J. 15, A247–A247 (2001).
Cuhlmann, S. et al. Disturbeces RelA Expression via c-Jun N-terminal kinase 1: a novel mode of NF-κB regulation that promotes arterial inflammation. Circ. Res. 108, 950–959 (2011).
Bryan, M. T. et al. Mechanoresponsive networks controlling vascular inflammation. Arterioscler. Thromb. Vasc. Biol. 34, 2199–2205 (2014).
Ruland, J. Return to homeostasis: downregulation of NF-κB responses. Nat. Immunol. 12, 709–714 (2011).
Fang, Y., Shi, C., Manduchi, E., Civelek, M. & Davies, P. F. MicroRNA-10a regulation of proinflammatory phenotype in athero-susceptible endothelium in vivo and in vitro. Proc. Natl Acad. Sci. USA 107, 13450–13455 (2010).
SenBanerjee, S. et al. KLF2 is a novel transcriptional regulator of endothelial proinflammatory activation. J. Exp. Med. 199, 1305–1315 (2004).
Yurdagul, A. Jr et al. Altered nitric oxide production mediates matrix-specific PAK2 and NF-κB activation by flow. Mol. Biol. Cell 24, 398–408 (2013).
Partridge, J. et al. Laminar shear stress acts as a switch to regulate divergent functions of NF-κB in endothelial cells. FASEB J. 21, 3553–3561 (2007).
Boon, R. A. et al. KLF2 suppresses TGF-β signaling in endothelium through induction of Smad7 and inhibition of AP-1. Arterioscler. Thromb. Vasc. Biol. 27, 532–539 (2007).
Fledderus, J. O. et al. Prolonged shear stress and KLF2 suppress constitutive proinflammatory transcription through inhibition of ATF2. Blood 109, 4249–4257 (2007).
Boon, R. A. et al. KLF2-induced actin shear fibers control both alignment to flow and JNK signaling in vascular endothelium. Blood 115, 2533–2542 (2010).
Dai, G. et al. Biomechanical forces in atherosclerosis-resistant vascular regions regulate endothelial redox balance via phosphoinositol 3-kinase/Akt-dependent activation of Nrf2. Circ. Res. 101, 723–733 (2007).
Fledderus, J. O. et al. KLF2 primes the antioxidant transcription factor Nrf2 for activation in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 28, 1339–1346 (2008).
Zakkar, M. et al. Activation of Nrf2 in endothelial cells protects arteries from exhibiting a proinflammatory state. Arterioscler. Thromb. Vasc. Biol. 29, 1851–1857 (2009).
Zakkar, M. et al. Increased endothelial mitogen-activated protein kinase phosphatase-1 expression suppresses proinflammatory activation at sites that are resistant to atherosclerosis. Circ. Res. 103, 726–732 (2008).
Saitoh, M. et al. Mammalian thioredoxin is a direct inhibitor of apoptosis signal-regulating kinase (ASK) 1. EMBO J. 17, 2596–2606 (1998).
Castier, Y., Brandes, R. P., Leseche, G., Tedgui, A. & Lehoux, S. p47phox-dependent NADPH oxidase regulates flow-induced vascular remodeling. Circ. Res. 97, 533–540 (2005).
Wang, J. C., Normand, S.-L. T., Mauri, L. & Kuntz, R. E. Coronary artery spatial distribution of acute myocardial infarction occlusions. Circulation 110, 278–284 (2004).
McDaniel, M. C. et al. Localization of culprit lesions in coronary arteries of patients with ST-segment elevation myocardial infarctions: relation to bifurcations and curvatures. Am. Heart J. 161, 508–515 (2011).
Phinikaridou, A., Hua, N., Pham, T. & Hamilton, J. A. Regions of low endothelial shear stress colocalize with positive vascular remodeling and atherosclerotic plaque disruption: an in vivo magnetic resonance imaging study. Circ. Cardiovasc. Imaging 6, 302–310 (2013).
Koskinas, K. C. et al. Natural history of experimental coronary atherosclerosis and vascular remodeling in relation to endothelial shear stress: a serial, in vivo intravascular ultrasound study. Circulation 121, 2092–2101 (2010).
Chatzizisis, Y. S. et al. Role of endothelial shear stress in the natural history of coronary atherosclerosis and vascular remodeling: molecular, cellular, and vascular behavior. J. Am. Coll. Cardiol. 49, 2379–2393 (2007).
Krams, R. et al. Evaluation of endothelial shear stress and 3D geometry as factors determining the development of atherosclerosis and remodeling in human coronary arteries in vivo: combining 3D reconstruction from angiography and IVUS (ANGUS) with computational fluid dynamics. Arterioscler. Thromb. Vasc. Biol. 17, 2061–2065 (1997).
Stone, P. H. et al. Effect of endothelial shear stress on the progression of coronary artery disease, vascular remodeling, and in-stent restenosis in humans: in vivo 6-month follow-up study. Circulation 108, 438–444 (2003).
Stone, P. H. et al. Regions of low endothelial shear stress are the sites where coronary plaque progresses and vascular remodelling occurs in humans: an in vivo serial study. Eur. Heart J. 28, 705–710 (2007).
Papafaklis, M. I. et al. Effect of the local hemodynamic environment on the de novo development and progression of eccentric coronary atherosclerosis in humans: insights from PREDICTION. Atherosclerosis 240, 205–211 (2015).
Samady, H. et al. Coronary artery wall shear stress is associated with progression and transformation of atherosclerotic plaque and arterial remodeling in patients with coronary artery disease. Circulation 124, 779–788 (2011).
Corban, M. T. et al. Combination of plaque burden, wall shear stress, and plaque phenotype has incremental value for prediction of coronary atherosclerotic plaque progression and vulnerability. Atherosclerosis 232, 271–276 (2014).
Stone, P. H. et al. Prediction of progression of coronary artery disease and clinical outcomes using vascular profiling of endothelial shear stress and arterial plaque characteristics: the PREDICTION Study. Circulation 126, 172–181 (2012).
Maurovich-Horvat, P., Ferencik, M., Voros, S., Merkely, B. & Hoffmann, U. Comprehensive plaque assessment by coronary CT angiography. Nat. Rev. Cardiol. 11, 390–402 (2014).
Hetterich, H. et al. Coronary computed tomography angiography based assessment of endothelial shear stress and its association with atherosclerotic plaque distribution in-vivo. PLoS ONE 10, e0115408 (2015).
Burke, A. P. et al. Coronary risk factors and plaque morphology in men with coronary disease who died suddenly. N. Engl. J. Med. 336, 1276–1282 (1997).
Davies, M. J. & Thomas, A. Thrombosis and acute coronary-artery lesions in sudden cardiac ischemic death. N. Engl. J. Med. 310, 1137–1140 (1984).
Virmani, R., Kolodgie, F. D., Burke, A. P., Farb, A. & Schwartz, S. M. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler. Thromb. Vasc. Biol. 20, 1262–1275 (2000).
Fujii, K. et al. OCT assessment of thin-cap fibroatheroma distribution in native coronary arteries. JACC Cardiovasc. Imaging 3, 168–175 (2010).
Zhao, Z. et al. Dynamic nature of nonculprit coronary artery lesion morphology in STEMI: a serial IVUS analysis from the HORIZONS-AMI trial. JACC Cardiovasc. Imaging 6, 86–95 (2013).
Chatzizisis, Y. S. et al. Prediction of the localization of high-risk coronary atherosclerotic plaques on the basis of low endothelial shear stress: an intravascular ultrasound and histopathology natural history study. Circulation 117, 993–1002 (2008).
Traub, O. & Berk, B. C. Laminar shear stress: mechanisms by which endothelial cells transduce an atheroprotective force. Arterioscler. Thromb. Vasc. Biol. 18, 677–685 (1998).
Tarbell, J. M. Shear stress and the endothelial transport barrier. Cardiovasc. Res. 87, 320–330 (2010).
Mehta, J. L., Chen, J., Hermonat, P. L., Romeo, F. & Novelli, G. Lectin-like, oxidized low-density lipoprotein receptor-1 (LOX-1): a critical player in the development of atherosclerosis and related disorders. Cardiovasc. Res. 69, 36–45 (2006).
Murase, T. et al. Fluid shear stress transcriptionally induces lectin-like oxidized LDL receptor-1 in vascular endothelial cells. Circ. Res. 83, 328–333 (1998).
Koskinas, K. C. et al. Synergistic effect of local endothelial shear stress and systemic hypercholesterolemia on coronary atherosclerotic plaque progression and composition in pigs. Int. J. Cardiol. 169, 394–401 (2013).
Zhou, J. et al. Regulation of vascular smooth muscle cell turnover by endothelial cell-secreted microRNA-126: role of shear stress. Circ. Res. 113, 40–51 (2013).
Qi, Y. X. et al. Rho–GDP dissociation inhibitor alpha downregulated by low shear stress promotes vascular smooth muscle cell migration and apoptosis: a proteomic analysis. Cardiovasc. Res. 80, 114–122 (2008).
Koskinas, K. C. et al. Thin-capped atheromata with reduced collagen content in pigs develop in coronary arterial regions exposed to persistently low endothelial shear stress. Arterioscler. Thromb. Vasc. Biol. 33, 1494–1504 (2013).
Chatzizisis, Y. S. et al. Augmented expression and activity of extracellular matrix-degrading enzymes in regions of low endothelial shear stress colocalize with coronary atheromata with thin fibrous caps in pigs. Circulation 123, 621–630 (2011).
Newby, A. C. Metalloproteinase expression in monocytes and macrophages and its relationship to atherosclerotic plaque instability. Arterioscler. Thromb. Vasc. Biol. 28, 2108–2114 (2008).
Pedrigi, R. M. et al. Inducing persistent flow disturbances accelerates atherogenesis and promotes thin cap fibroatheroma development in D374Y-PCSK9 hypercholesterolemic minipigs. Circulation 132, 1003–1012 (2015).
Obaid, D. R. et al. Atherosclerotic plaque composition and classification identified by coronary computed tomography: assessment of computed tomography-generated plaque maps compared with virtual histology intravascular ultrasound and histology. Circ. Cardiovasc. Imaging 6, 655–664 (2013).
Voros, S. et al. Coronary atherosclerosis imaging by coronary CT angiography: current status, correlation with intravascular interrogation and meta-analysis. JACC Cardiovasc. Imaging 4, 537–548 (2011).
Pundziute, G. et al. Head-to-head comparison of coronary plaque evaluation between multislice computed tomography and intravascular ultrasound radiofrequency data analysis. JACC Cardiovasc. Interv. 1, 176–182 (2008).
Nair, A., Margolis, M. P., Kuban, B. D. & Vince, D. G. Automated coronary plaque characterisation with intravascular ultrasound backscatter: ex vivo validation. EuroIntervention 3, 113–120 (2007).
Yabushita, H. et al. Characterization of human atherosclerosis by optical coherence tomography. Circulation 106, 1640–1645 (2002).
Eshtehardi, P. et al. Association of coronary wall shear stress with atherosclerotic plaque burden, composition, and distribution in patients with coronary artery disease. J. Am. Heart Assoc. 1, e002543 (2012).
Wentzel, J. J. et al. In vivo assessment of the relationship between shear stress and necrotic core in early and advanced coronary artery disease. EuroIntervention 9, 989–995 (2013).
Rodriguez-Granillo, G. A. et al. Plaque composition and its relationship with acknowledged shear stress patterns in coronary arteries. J. Am. Coll. Cardiol. 47, 884–885 (2006).
Vergallo, R. et al. Endothelial shear stress and coronary plaque characteristics in humans: a combined frequency-domain optical coherence tomography and computational fluid dynamics study. Circ. Cardiovasc. Imaging 7, 905–911 (2014).
Falk, E. Why do plaques rupture? Circulation 86 (Suppl. 6), III30–III42 (1992).
Burke, A. P. et al. Healed plaque ruptures and sudden coronary death: evidence that subclinical rupture has a role in plaque progression. Circulation 103, 934–940 (2001).
Loree, H. M., Kamm, R. D., Stringfellow, R. G. & Lee, R. T. Effects of fibrous cap thickness on peak circumferential stress in model atherosclerotic vessels. Circ. Res. 71, 850–858 (1992).
Teng, Z. et al. Arterial luminal curvature and fibrous-cap thickness affect critical stress conditions within atherosclerotic plaque: an in vivo MRI-based 2D finite-element study. Ann. Biomed. Eng. 38, 3096–3101 (2010).
Tang, D., Yang, C., Kobayashi, S. & Ku, D. N. Effect of a lipid pool on stress/strain distributions in stenotic arteries: 3D fluid-structure interactions (FSI) models. J. Biomech. Eng. 126, 363–370 (2004).
Gao, H. & Long, Q. Effects of varied lipid core volume and fibrous cap thickness on stress distribution in carotid arterial plaques. J. Biomech. 41, 3053–3059 (2008).
Finet, G., Ohayon, J. & Rioufol, G. Biomechanical interaction between cap thickness, lipid core composition and blood pressure in vulnerable coronary plaque: impact on stability or instability. Coron. Artery Dis. 15, 13–20 (2004).
Li, Z. Y. et al. Does calcium deposition play a role in the stability of atheroma? Location may be the key. Cerebrovasc. Dis. 24, 452–459 (2007).
Bluestein, D. et al. Influence of microcalcifications on vulnerable plaque mechanics using FSI modeling. J. Biomech. 41, 1111–1118 (2008).
Imoto, K. et al. Longitudinal structural determinants of atherosclerotic plaque vulnerability: a computational analysis of stress distribution using vessel models and three-dimensional intravascular ultrasound imaging. J. Am. Coll. Cardiol. 46, 1507–1515 (2005).
Vengrenyuk, Y. et al. A hypothesis for vulnerable plaque rupture due to stress-induced debonding around cellular microcalcifications in thin fibrous caps. Proc. Natl Acad. Sci. USA 103, 14678–14683 (2006).
Kelly-Arnold, A. et al. Revised microcalcification hypothesis for fibrous cap rupture in human coronary arteries. Proc. Natl Acad. Sci. USA 110, 10741–10746 (2013).
Irkle, A. et al. Identifying active vascular microcalcification by 18F-sodium fluoride positron emission tomography. Nat. Commun. 6, 7495 (2015).
Tarkin, J. M., Joshi, F. R. & Rudd, J. H. PET imaging of inflammation in atherosclerosis. Nat. Rev. Cardiol. 11, 443–457 (2014).
Joshi, N. V. et al. F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: a prospective clinical trial. Lancet 383, 705–703 (2014).
Cheng, G. C., Loree, H. M., Kamm, R. D., Fishbein, M. C. & Lee, R. T. Distribution of circumferential stress in ruptured and stable atherosclerotic lesions. A structural analysis with histopathological correlation. Circulation 87, 1179–1187 (1993).
Lee, R. T. et al. Computational structural analysis based on intravascular ultrasound imaging before in vitro angioplasty: prediction of plaque fracture locations. J. Am. Coll. Cardiol. 21, 777–782 (1993).
Huang, H. et al. The impact of calcification on the biomechanical stability of atherosclerotic plaques. Circulation 103, 1051–1056 (2001).
Lee, R. T., Schoen, F. J., Loree, H. M., Lark, M. W. & Libby, P. Circumferential stress and matrix metalloproteinase 1 in human coronary atherosclerosis: implications for plaque rupture. Arterioscler. Thromb. Vasc. Biol. 16, 1070–1073 (1996).
Hallow, K. M., Taylor, W. R., Rachev, A. & Vito, R. P. Markers of inflammation collocate with increased wall stress in human coronary arterial plaque. Biomech. Model. Mechanobiol. 8, 473–486 (2009).
Qiu, J. et al. Biomechanical regulation of vascular smooth muscle cell functions: from in vitro to in vivo understanding. J. R. Soc. Interface 11, 20130852 (2014).
Rodriguez, A. I. et al. MEF2B-Nox1 signaling is critical for stretch-induced phenotypic modulation of vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 35, 430–438 (2015).
Cattaruzza, M., Dimigen, C., Ehrenreich, H. & Hecker, M. Stretch-induced endothelin B receptor-mediated apoptosis in vascular smooth muscle cells. FASEB J. 14, 991–998 (2000).
Teng, Z. et al. Critical mechanical conditions around neovessels in carotid atherosclerotic plaque may promote intraplaque hemorrhage. Atherosclerosis 223, 321–326 (2012).
Tang, D. et al. 3D MRI-based multicomponent FSI models for atherosclerotic plaques. Ann. Biomed. Eng. 32, 947–960 (2004).
Li, Z. Y. et al. Stress analysis of carotid plaque rupture based on in vivo high resolution MRI. J. Biomech. 39, 2611–2622 (2006).
Li, Z. Y. et al. Structural analysis and magnetic resonance imaging predict plaque vulnerability: a study comparing symptomatic and asymptomatic individuals. J. Vasc. Surg. 45, 768–775 (2007).
Sadat, U. et al. Association between biomechanical structural stresses of atherosclerotic carotid plaques and subsequent ischaemic cerebrovascular events — a longitudinal in vivo magnetic resonance imaging-based finite element study. Eur. J. Vasc. Endovasc. Surg. 40, 485–491 (2010).
Ohayon, J., Teppaz, P., Finet, G. & Rioufol, G. In-vivo prediction of human coronary plaque rupture location using intravascular ultrasound and the finite element method. Coron. Artery Dis. 12, 655–663 (2001).
Dirksen, M. T., van der Wal, A. C., van den Berg, F. M., van der Loos, C. M. & Becker, A. E. Distribution of inflammatory cells in atherosclerotic plaques relates to the direction of flow. Circulation 98, 2000–2003 (1998).
Wentzel, J. J. et al. Endothelial shear stress in the evolution of coronary atherosclerotic plaque and vascular remodelling: current understanding and remaining questions. Cardiovasc. Res. 96, 234–243 (2012).
Waxman, S., Ishibashi, F. & Muller, J. E. Detection and treatment of vulnerable plaques and vulnerable patients: novel approaches to prevention of coronary events. Circulation 114, 2390–2411 (2006).
Sameshima, N. et al. The values of wall shear stress, turbulence kinetic energy and blood pressure gradient are associated with atherosclerotic plaque erosion in rabbits. J. Atheroscler. Thromb. 21, 831–838 (2014).
Maalej, N., Holden, J. & Folts, J. Effect of shear stress on acute platelet thrombus formation in canine stenosed carotid arteries: an in vivo quantitative study. J. Thromb. Thrombolysis 5, 231–238 (1998).
Bark, D. L. Jr, Para, A. N. & Ku, D. N. Correlation of thrombosis growth rate to pathological wall shear rate during platelet accumulation. Biotechnol. Bioeng. 109, 2642–2650 (2012).
Farb, A. et al. Coronary plaque erosion without rupture into a lipid core: a frequent cause of coronary thrombosis in sudden coronary death. Circulation 93, 1354–1363 (1996).
Arbustini, E. et al. Plaque erosion is a major substrate for coronary thrombosis in acute myocardial infarction. Heart 82, 269–272 (1999).
Virmani, R., Burke, A. P. & Farb, A. Plaque rupture and plaque erosion. Thromb. Haemost. 1, 1–3 (1999).
Fukumoto, Y. et al. Localized elevation of shear stress is related to coronary plaque rupture: a 3-dimensional intravascular ultrasound study with in-vivo color mapping of shear stress distribution. J. Am. Coll. Cardiol. 51, 645–650 (2008).
Gijsen, F. J. et al. Strain distribution over plaques in human coronary arteries relates to shear stress. Am. J. Physiol. Heart Circ. Physiol. 295, H1608–H1614 (2008).
Bank, A. J., Versluis, A., Dodge, S. M. & Douglas, W. H. Atherosclerotic plaque rupture: a fatigue process? Med. Hypotheses 55, 480–484 (2000).
Versluis, A., Bank, A. J. & Douglas, W. H. Fatigue and plaque rupture in myocardial infarction. J. Biomech. 39, 339–347 (2006).
Huang, Y. et al. In vivo MRI-based simulation of fatigue process: a possible trigger for human carotid atherosclerotic plaque rupture. Biomed. Eng. Online 12, 36 (2013).
Pei, X., Wu, B., Tang, T. Y., Gillard, J. H. & Li, Z.-Y. Fatigue crack growth under pulsatile pressure and plaque rupture. JACC Cardiovasc. Imaging 7, 738–740 (2014).
Motoyama, S. et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J. Am. Coll. Cardiol. 50, 319–326 (2007).
Motoyama, S. et al. Plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up. J. Am. Coll. Cardiol. 66, 337–346 (2015).
Acknowledgements
The authors were supported by the British Heart Foundation (FS/13/33/30168), Heart Research UK (RG2638/14/16), the Cambridge NIHR Biomedical Research Centre, and the BHF Cambridge Centre for Research Excellence. The authors would also like to thank Shuo Wang (Department of Radiology, University of Cambridge, UK) for the preparation of the images for publication.
Author information
Authors and Affiliations
Contributions
A.J.B., Z.T., and P.C. researched data for the article, and wrote and edited the manuscript. J.H.G., H.S., and M.R.B. discussed the content, and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Brown, A., Teng, Z., Evans, P. et al. Role of biomechanical forces in the natural history of coronary atherosclerosis. Nat Rev Cardiol 13, 210–220 (2016). https://doi.org/10.1038/nrcardio.2015.203
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2015.203
This article is cited by
-
Macrophages in cardiac remodelling after myocardial infarction
Nature Reviews Cardiology (2023)
-
Matrix stiffness, endothelial dysfunction and atherosclerosis
Molecular Biology Reports (2023)
-
Purinergic receptors mediate endothelial dysfunction and participate in atherosclerosis
Purinergic Signalling (2023)
-
Atherosclerosis: nexus of vascular dynamics and cellular cross talks
Molecular and Cellular Biochemistry (2022)
-
Lipid-rich Plaques Detected by Near-infrared Spectroscopy Are More Frequently Exposed to High Shear Stress
Journal of Cardiovascular Translational Research (2021)