Key Points
-
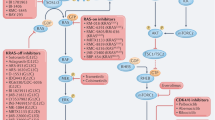
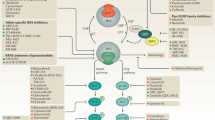
RAS proteins, their regulators and the downstream enzymes that they control are activated in many tumour types by a variety of mechanisms, including oncogenic mutation of RAS genes.
-
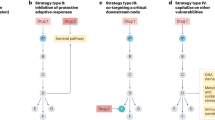
They are crucial mediators of several of the malignant characteristics of transformed cells and are therefore good candidates for tumour therapy.
-
RAS proteins require post-translational modification by farnesylation to be biologically active. Farnesyl transferase inhibitors have some antitumour activity in the clinic, but they seem to act through targets other than RAS.
-
Kinase inhibitors that block either RAF or mitogen-activated protein (MAP) kinase kinase MEK in the RAF/MAP kinase pathway downstream of RAS have been developed and show promise in early clinical trials.
-
Inhibitors acting on epidermal growth factor (EGF) receptor and ERBB2 upstream activators of RAS have been developed. Antibodies directed against ERBB2 have been licensed for the treatment of breast cancer, whereas small-molecule EGF receptor inhibitors show potential against lung cancer in clinical trials.
-
Other RAS-related therapies are in development, including inhibitors of AKT/PKB kinase activity, which is activated by RAS oncogenic mutation and by PTEN tumour-suppressor gene loss.
Abstract
The RAS proteins control signalling pathways that are key regulators of several aspects of normal cell growth and malignant transformation. They are aberrant in most human tumours due to activating mutations in the RAS genes themselves or to alterations in upstream or downstream signalling components. Rational therapies that target the RAS pathways might inhibit tumour growth, survival and spread. Several of these new therapeutic agents are showing promise in the clinic and many more are being developed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Barbacid, M. Ras genes. Annu. Rev. Biochem. 56, 779–827 (1987).
Bos, J. L. Ras oncogenes in human cancer: a review. Cancer Res. 49, 4682–4689 (1989).
Shields, J. M., Pruitt, K., McFall, A., Shaub, A. & Der, C. J. Understanding Ras: 'it ain't over 'til it's over'. Trends Cell Biol. 10, 147–154 (2000).
Campbell, S. L., Khosravi-Far, R., Rossman, K. L., Clark, G. J. & Der, C. J. Increasing complexity of Ras signaling. Oncogene 17, 1395–1413 (1998).
Wittinghofer, A. Signal transduction via Ras. Biol. Chem. 379, 933–937 (1998).
Lowy, D. R. & Willumsen, B. M. Function and regulation of ras. Annu. Rev. Biochem. 62, 851–891 (1993).
Johnson, L. et al. K-ras is an essential gene in the mouse with partial functional overlap with N-ras. Genes Dev. 11, 2468–2481 (1997).
Seabra, M. C. Membrane association and targeting of prenylated Ras-like GTPases. Cell Signal. 10, 167–172 (1998).
Cox, A. D. & Der, C. J. Farnesyltransferase inhibitors and cancer treatment: targeting simply Ras? Biochim. Biophys. Acta 1333, F51–F71 (1997).
Hancock, J. F., Magee, A. I., Childs, J. E. & Marshall, C. J. All ras proteins are polyisoprenylated but only some are palmitoylated. Cell 57, 1167–1177 (1989).
Hancock, J. F., Paterson, H. & Marshall, C. J. A polybasic domain or palmitoylation is required in addition to the CAAX motif to localize p21ras to the plasma membrane. Cell 63, 133–139 (1990). References 10 and 11 describe the original characterization of the modification of RAS by farnesylation and, in the case of HRAS and NRAS, but not KRAS, palmitoylation.
Reuther, G. W. & Der, C. J. The Ras branch of small GTPases: Ras family members don't fall far from the tree. Curr. Opin. Cell Biol. 12, 157–165 (2000).
Takai, Y., Sasaki, T. & Matozaki, T. Small GTP-binding proteins. Physiol. Rev. 81, 153–208 (2001).
Cullen, P. J. & Lockyer, P. J. Integration of calcium and Ras signalling. Nature Rev. Mol. Cell Biol. 3, 339–348 (2002).
Daub, H., Weiss, F. U., Wallasch, C. & Ullrich, A. Role of transactivation of the EGF receptor in signalling by G-protein-coupled receptors. Nature 379, 557–560 (1996).
Chiu, V. K. et al. Ras signalling on the endoplasmic reticulum and the Golgi. Nature Cell Biol. 4, 343–350 (2002).
Ebinu, J. O. et al. RasGRP, a Ras guanyl nucleotide-releasing protein with calcium- and diacylglycerol-binding motifs. Science 280, 1082–1086 (1998).
Downward, J., Graves, J. D., Warne, P. H., Rayter, S. & Cantrell, D. A. Stimulation of p21ras upon T-cell activation. Nature 346, 719–723 (1990).
Donovan, S., Shannon, K. M. & Bollag, G. GTPase activating proteins: critical regulators of intracellular signaling. Biochim. Biophys. Acta 1602, 23–45 (2002).
Leevers, S. J., Paterson, H. F. & Marshall, C. J. Requirement for Ras in Raf activation is overcome by targeting Raf to the plasma membrane. Nature 369, 411–414 (1994).
Marais, R., Light, Y., Paterson, H. F. & Marshall, C. J. Ras recruits Raf-1 to the plasma membrane for activation by tyrosine phosphorylation. EMBO J. 14, 3136–3145 (1995).
Yordy, J. S. & Muise-Helmericks, R. C. Signal transduction and the Ets family of transcription factors. Oncogene 19, 6503–6513 (2000).
Pruitt, K. & Der, C. J. Ras and Rho regulation of the cell cycle and oncogenesis. Cancer Lett. 171, 1–10 (2001).
Rodriguez-Viciana, P. et al. Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370, 527–532 (1994).
Pacold, M. E. et al. Crystal structure and functional analysis of Ras binding to its effector phosphoinositide 3-kinaseγ. Cell 103, 931–943 (2000). This describes the three-dimensional structure of RAS in complex with one of its effectors, PI3K. The size of the interaction interface shows the difficulty that is likely to face the design of inhibitors of protein–protein interaction.
Datta, S. R., Brunet, A. & Greenberg, M. E. Cellular survival: a play in three Akts. Genes Dev. 13, 2905–2927 (1999).
Khwaja, A., Rodriguez-Viciana, P., Wennstrom, S., Warne, P. H. & Downward, J. Matrix adhesion and Ras transformation both activate a phosphoinositide 3-OH kinase and protein kinase B/Akt cellular survival pathway. EMBO J. 16, 2783–2793 (1997).
Lambert, J. M. et al. Tiam1 mediates Ras activation of Rac by a PI(3)K-independent mechanism. Nature Cell Biol. 4, 621–625 (2002).
Malliri, A. et al. Mice deficient in the Rac activator Tiam1 are resistant to Ras-induced skin tumours. Nature 417, 867–871 (2002).
De Ruiter, N. D., Burgering, B. M. & Bos, J. L. Regulation of the Forkhead transcription factor AFX by Ral-dependent phosphorylation of threonines 447 and 451. Mol. Cell. Biol. 21, 8225–8235 (2001).
Kelley, G. G., Reks, S. E., Ondrako, J. M. & Smrcka, A. V. Phospholipase Cε: a novel Ras effector. EMBO J. 20, 743–754 (2001).
Weiss, B., Bollag, G. & Shannon, K. Hyperactive Ras as a therapeutic target in neurofibromatosis type 1. Am. J. Med. Genet. 89, 14–22 (1999).
Mendelsohn, J. & Baselga, J. The EGF receptor family as targets for cancer therapy. Oncogene 19, 6550–6565 (2000).
Kuan, C. T., Wikstrand, C. J. & Bigner, D. D. EGF mutant receptor vIII as a molecular target in cancer therapy. Endocr. Relat. Cancer 8, 83–96 (2001).
Bellacosa, A. et al. Molecular alterations of the AKT2 oncogene in ovarian and breast carcinomas. Int. J. Cancer 64, 280–285 (1995).
Simpson, L. & Parsons, R. PTEN: life as a tumor suppressor. Exp. Cell Res. 264, 29–41 (2001).
Sebti, S. M. & Hamilton, A. D. Farnesyltransferase and geranylgeranyltransferase I inhibitors and cancer therapy: lessons from mechanism and bench-to-bedside translational studies. Oncogene 19, 6584–6593 (2000).
Cox, A. D. & Der, C. J. Farnesyltransferase inhibitors: promises and realities. Curr. Opin. Pharmacol. 2, 388–393 (2002).
Kohl, N. E. et al. Inhibition of farnesyltransferase induces regression of mammary and salivary carcinomas in ras transgenic mice. Nature Med. 1, 792–797 (1995). The study that launched massive interest in farnesyl-transferase inhibitors as tumour therapeutic drugs aimed at RAS. Unfortunately, the activity turned out to be selective for HRAS, an isoform that is only very rarely activated in human tumours.
Lobell, R. B. et al. Evaluation of farnesyl:protein transferase and geranylgeranyl:protein transferase inhibitor combinations in preclinical models. Cancer Res. 61, 8758–8768 (2001).
Prendergast, G. C. Actin' up: RhoB in cancer and apoptosis. Nature Rev. Cancer 1, 162–168 (2001).
Ohkanda, J., Knowles, D. B., Blaskovich, M. A., Sebti, S. M. & Hamilton, A. D. Inhibitors of protein farnesyltransferase as novel anticancer agents. Curr. Top. Med. Chem. 2, 303–323 (2002).
Singh, S. B. & Lingham, R. B. Current progress on farnesyl protein transferase inhibitors. Curr. Opin. Drug. Discov. Devel. 5, 225–244 (2002).
Cortes, J. E. et al. Efficacy of the farnesyl transferase inhibitor, ZARNESTRATM (R115777), in chronic myeloid leukemia and other other hematological malignancies. Blood 31, 31 Oct 31 2002 [epub ahead of print].
Crooke, S. T. Potential roles of antisense technology in cancer chemotherapy. Oncogene 19, 6651–6659 (2000).
Mukhopadhyay, T., Tainsky, M., Cavender, A. C. & Roth, J. A. Specific inhibition of K-ras expression and tumorigenicity of lung cancer cells by antisense RNA. Cancer Res. 51, 1744–1748 (1991).
Chen, G., Oh, S., Monia, B. P. & Stacey, D. W. Antisense oligonucleotides demonstrate a dominant role of c-Ki-RAS proteins in regulating the proliferation of diploid human fibroblasts. J. Biol. Chem. 271, 28259–28265 (1996).
Monia, B. P., Johnston, J. F., Geiger, T., Muller, M. & Fabbro, D. Antitumor activity of a phosphorothioate antisense oligodeoxynucleotide targeted against C-raf kinase. Nature Med. 2, 668–675 (1996).
HŸser, M. et al. MEK kinase activity is not necessary for Raf-1 function. EMBO J. 20, 1940–1951 (2001).
Coudert, B. et al. Phase II trial with ISIS 5132 in patients with small-cell (SCLC) and non-small cell (NSCLC) lung cancer. A European Organization for Research and Treatment of Cancer (EORTC) Early Clinical Studies Group report. Eur. J. Cancer 37, 2194–2198 (2001).
Sawyers, C. L. Rational therapeutic intervention in cancer: kinases as drug targets. Curr. Opin. Genet. Dev. 12, 111–115 (2002).
Hoshino, R. et al. Constitutive activation of the 41-/43-kDa mitogen-activated protein kinase signaling pathway in human tumors. Oncogene 18, 813–822 (1999).
Dudley, D. T., Pang, L., Decker, S. J., Bridges, A. J. & Saltiel, A. R. A synthetic inhibitor of the mitogen-activated protein kinase cascade. Proc. Natl Acad. Sci. USA 92, 7686–7689 (1995).
Favata, M. F. et al. Identification of a novel inhibitor of mitogen-activated protein kinase kinase. J. Biol. Chem. 273, 18623–18632 (1998).
Sebolt-Leopold, J. S. Development of anticancer drugs targeting the MAP kinase pathway. Oncogene 19, 6594–6599 (2000).
Sebolt-Leopold, J. S. et al. Blockade of the MAP kinase pathway suppresses growth of colon tumors in vivo. Nature Med. 5, 810–816 (1999).
Lyons, J. F., Wilhelm, S., Hibner, B. & Bollag, G. Discovery of a novel Raf kinase inhibitor. Endocr. Relat. Cancer. 8, 219–225 (2001).
Davies, H. et al. Mutations of the BRAF gene in human cancer. Nature 417, 949–954 (2002).
Normanno, N., Bianco, C., De Luca, A. & Salomon, D. S. The role of EGF-related peptides in tumor growth. Front. Biosci. 6, D685–D707 (2001).
Sibilia, M. et al. The EGF receptor provides an essential survival signal for SOS-dependent skin tumor development. Cell 102, 211–220 (2000). An elegant transgenic mouse study showing that the ability of activated endogenous RAS protein to promote tumour formation is dependent on the activity of EGFR, indicating the importance of autocrine EGF signalling in RAS transformation.
Fabbro, D., Parkinson, D. & Matter, A. Protein tyrosine kinase inhibitors: new treatment modalities? Curr. Opin. Pharmacol. 2, 374–381 (2002).
Wakeling, A. E. Epidermal growth factor receptor tyrosine kinase inhibitors. Curr. Opin. Pharmacol. 2, 382–387 (2002).
de Bono, J. S. & Rowinsky, E. K. The ErbB receptor family: a therapeutic target for cancer. Trends Mol. Med. 8, S19–S26 (2002).
Herbst, R. S. ZD1839: targeting the epidermal growth factor receptor in cancer therapy. Expert Opin. Investig. Drugs 11, 837–849 (2002).
Hidalgo, M. et al. Phase I and pharmacologic study of OSI-774, an epidermal growth factor receptor tyrosine kinase inhibitor, in patients with advanced solid malignancies. J. Clin. Oncol. 19, 3267–3279 (2001).
Schulze, A., Lehmann, K., Jefferies, H. B., McMahon, M. & Downward, J. Analysis of the transcriptional program induced by Raf in epithelial cells. Genes Dev. 15, 981–994 (2001).
Herbst, R. S. & Shin, D. M. Monoclonal antibodies to target epidermal growth factor receptor-positive tumors: a new paradigm for cancer therapy. Cancer 94, 1593–1611 (2002).
Robert, F. et al. Phase I study of anti-epidermal growth factor receptor antibody cetuximab in combination with radiation therapy in patients with advanced head and neck cancer. J. Clin. Oncol. 19, 3234–3243 (2001).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001). The results of the Phase III trials with trastuzumab (Herceptin) that showed it to be an effective treatment for metastatic breast cancer with amplification of ERBB2.
Xie, Y., Li, K. & Hung, M. C. Tyrosine phosphorylation of Shc proteins and formation of Shc/Grb2 complex correlate to the transformation of NIH3T3 cells mediated by the point-mutation activated neu. Oncogene 10, 2409–2413 (1995).
Keshamouni, V. G., Mattingly, R. R. & Reddy, K. B. Mechanism of 17-β-estradiol-induced Erk1/2 activation in breast cancer cells: A role for HER2 and PKC-δ. J. Biol. Chem. 277, 22558–22565 (2002).
Hunger, R. E. et al. Successful induction of immune responses against mutant ras in melanoma patients using intradermal injection of peptides and GM-CSF as adjuvant. Exp. Dermatol. 10, 161–167 (2001).
Reuveni, H. et al. Toward a PKB inhibitor: modification of a selective PKA inhibitor by rational design. Biochemistry 41, 10304–10314 (2002).
Bergo, M. O. et al. Absence of CAAX endoprotease Rce 1: effects on cell growth and transformation. Mol. Cell. Biol. 22, 171–181 (2002).
Kloog, Y., Cox, A. D. & Sinensky, M. Concepts in Ras-directed therapy. Exp. Opin. Investig. Drugs 8, 2121–2140 (1999).
Kimoto, M., Sakamoto, K., Shirouzu, M., Mirao, I. & Yokoyama, S. RNA aptamers that specifically bind to the Ras-binding domain of c-Raf-1. FEBS Lett. 441, 322–326 (1998).
Kato-Stankiewicz, J. et al. Inhibitors of Ras/Raf-1 interaction identified by two-hybrid screening revert Ras-dependent transformation phenotypes in human cancer cells. Proc. Natl Acad. Sci. USA 99, 14398–14403 (2002).
Ahmadian, M. R. et al. Guanosine triphopshatase stimulation of oncogenic Ras mutants. Proc. Natl Acad. Sci. USA 96, 7065–7070 (1999).
Hidalgo, M. & Rowinsky, E. K. The rapamycin-sensitive signal transduction pathway as a target for cancer therapy. Oncogene 19, 6680–6686 (2000).
Neckers, L. Hsp90 inhibitors as novel cancer chemotherapeutic agents. Trends Mol. Med. 8, S55–S61 (2002).
Zucker, S., Cao, J. & Chen, W. T. Critical appraisal of the use of matrix metalloproteinase inhibitors in cancer treatment. Oncogene 19, 6642–6650 (2000).
Rosen, L. S. Angiogenesis inhibition in solid tumors. Cancer J. 7 (Suppl. 3), S120–S128 (2001).
Kirn, D., Niculescu-Duvaz, I., Hallden, G. & Springer, C. J. The emerging fields of suicide gene therapy and virotherapy. Trends Mol. Med. 8, S68–S73 (2002).
Biederer, C., Ries, S., Brandts, C. H. & McCormick, F. Replication-selective viruses for cancer therapy. J. Mol. Med. 80, 163–175 (2002).
Raff, M. C. Social controls on cell survival and cell death. Nature 356, 397–400 (1992).
End, D. W. et al. Characterization of the antitumor effects of the selective farnesyl protein transferase inhibitor R115777 in vivo and in vitro. Cancer Res. 61, 131–137 (2001).
Rose, W. C. et al. Preclinical antitumor activity of BMS-214662, a highly apoptotic and novel farnesyltransferase inhibitor. Cancer Res. 61, 7507–7517 (2001). References 86 and 87 describe preclinical data on two of the leading farnesyltransferase inhibitors. The significant effects that are seen in these model systems are probably not due to targeting RAS, and it remains unclear whether they can be replicated in the clinic.
Author information
Authors and Affiliations
Related links
Related links
DATABASES
Cancer.gov
LocusLink
Glossary
- SH2 DOMAIN
-
Src homology 2 domain. A protein domain capable of binding tyrosine phosphorylated sites.
- SH3 DOMAIN
-
Src homology 3 domain. A protein domain capable of binding proline-rich motifs.
- AKT/PKB
-
A family of three closely related serine/threonine protein kinases containing amino-terminal pleckstrin homology domains that bind to the lipid products of phosphatidylinositol 3-kinase (PI3K). They are activated on PI3K stimulation and provide anti-apoptotic signals to the cell.
- FORKHEAD
-
A large superfamily of transcription factors, of which one family, FoxO, is phosphorylated and inhibited by AKT/PKB.
- BIM
-
A member of the BCL-2 family of apoptosis regulatory proteins. BIM induces apoptosis by acting at mitochondria to promote the release of cytochrome c into the cytosol.
- FAS LIGAND
-
An extracellular protein that binds and activates the death receptor FAS, also known as CD95 and APO1, leading to initiation of apoptosis.
- p110α
-
One isoform of the catalytic subunit of phosphoinositide 3-kinase (PI3K). Type I PI3Ks are made up of either α, β, γ or δ forms of p110, together with a regulatory p85 subunit (for p110α, β and δ) or p101 subunit (for p110γ).
- IC50
-
The concentration of a drug giving a 50% inhibition of the activity of a target enzyme.
- RNA APTAMER
-
A synthetic RNA molecule selected for ability to interact with a target protein.
Rights and permissions
About this article
Cite this article
Downward, J. Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer 3, 11–22 (2003). https://doi.org/10.1038/nrc969
Issue Date:
DOI: https://doi.org/10.1038/nrc969