Abstract
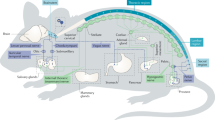
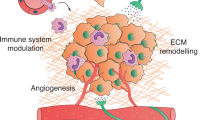
The peripheral autonomic nervous system (ANS) is known to regulate gene expression in primary tumours and their surrounding microenvironment. Activation of the sympathetic division of the ANS in particular modulates gene expression programmes that promote metastasis of solid tumours by stimulating macrophage infiltration, inflammation, angiogenesis, epithelial–mesenchymal transition and tumour invasion, and by inhibiting cellular immune responses and programmed cell death. Haematological cancers are modulated by sympathetic nervous system (SNS) regulation of stem cell biology and haematopoietic differentiation programmes. In addition to identifying a molecular basis for physiologic stress effects on cancer, these findings have also identified new pharmacological strategies to inhibit cancer progression in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Weiner, H. Perturbing the Organism: The Biology of Stressful Experience (Univ. of Chicago Press, 1992).
Sapolsky, R. M. Why Zebras Don't Get Ulcers: A Guide To Stress, Stress-Related Diseases, And Coping (Freeman, 1994).
Sherwood, L. Human Physiology: From Cells to Systems (Cengage Learning, 2015).
Katayama, Y. et al. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 124, 407–421 (2006).
Sloan, E. K. et al. Social stress enhances sympathetic innervation of primate lymph nodes: mechanisms and implications for viral pathogenesis. J. Neurosci. 27, 8857–8865 (2007).
Lutgendorf, S. K. et al. Depression, social support, and β-adrenergic transcription control in human ovarian cancer. Brain Behav. Immun. 23, 176–183 (2009).
Powell, N. D. et al. Social stress up-regulates inflammatory gene expression in the leukocyte transcriptome via β-adrenergic induction of myelopoiesis. Proc. Natl Acad. Sci. USA 110, 16574–16579 (2013).
Scheiermann, C., Kunisaki, Y. & Frenette, P. S. Circadian control of the immune system. Nat. Rev. Immunol. 13, 190–198 (2013).
Cole, S. W. Social regulation of human gene expression: mechanisms and implications for public health. Am. J. Publ. Health 103 (Suppl. 1), S84–S92 (2013).
Cole, S. W. Human social genomics. PLoS Genet. 10, e1004601 (2014).
Hanoun, M., Maryanovich, M., Arnal-Estape, A. & Frenette, P. S. Neural regulation of hematopoiesis, inflammation, and cancer. Neuron 86, 360–373 (2015).
Irwin, M. R. & Cole, S. W. Reciprocal regulation of the neural and innate immune systems. Nat. Rev. Immunol. 11, 625–632 (2011).
Antoni, M. H. et al. The influence of bio-behavioural factors on tumour biology: pathways and mechanisms. Nat. Rev. Cancer. 6, 240–248 (2006).
Cole, S. W. & Sood, A. K. Molecular pathways: β-adrenergic signaling in cancer. Clin. Cancer Res. 18, 1201–1206 (2012).
Armaiz-Pena, G. N., Cole, S. W., Lutgendorf, S. K. & Sood, A. K. Neuroendocrine influences on cancer progression. Brain Behav. Immun. 30, S19–S25 (2013).
Cole, S. W. Nervous system regulation of the cancer genome. Brain Behav. Immun. 30 (Suppl.), S10–S18 (2013).
Powe, D. G. & Entschladen, F. Targeted therapies: using β-blockers to inhibit breast cancer progression. Nat. Rev. Clin. Oncol. 8, 511–512 (2011).
Richter, S. D. et al. Time kinetics of the endocrine response to acute psychological stress. J. Clin. Endocrinol. Metab. 81, 1956–1960 (1996).
Schommer, N. C., Hellhammer, D. H. & Kirschbaum, C. Dissociation between reactivity of the hypothalamus–pituitary–adrenal axis and the sympathetic–adrenal–medullary system to repeated psychosocial stress. Psychosom. Med. 65, 450–460 (2003).
Wingenfeld, K., Whooley, M. A., Neylan, T. C., Otte, C. & Cohen, B. E. Effect of current and lifetime posttraumatic stress disorder on 24-h urinary catecholamines and cortisol: results from the Mind Your Heart Study. Psychoneuroendocrinology 52, 83–91 (2015).
Sloan, E. K., Capitanio, J. P., Tarara, R. P. & Cole, S. W. Social temperament and lymph node innervation. Brain Behav. Immun. 22, 717–726 (2008).
Schofl, C., Becker, C., Prank, K., von zur Muhlen, A. & Brabant, G. Twenty-four-hour rhythms of plasma catecholamines and their relation to cardiovascular parameters in healthy young men. Eur. J. Endocrinol. 137, 675–683 (1997).
Dimitrov, S. et al. Cortisol and epinephrine control opposing circadian rhythms in T cell subsets. Blood 113, 5134–5143 (2009).
Eng, J. W. et al. Housing temperature-induced stress drives therapeutic resistance in murine tumour models through β2-adrenergic receptor activation. Nat. Commun. 6, 6426 (2015).
Mendez-Ferrer, S., Lucas, D., Battista, M. & Frenette, P. S. Haematopoietic stem cell release is regulated by circadian oscillations. Nature 452, 442–447 (2008).
Dutta, P. et al. Myocardial infarction accelerates atherosclerosis. Nature 487, 325–329 (2012).
Heidt, T. et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 20, 754–758 (2014).
Nakai, A., Hayano, Y., Furuta, F., Noda, M. & Suzuki, K. Control of lymphocyte egress from lymph nodes through β2-adrenergic receptors. J. Exp. Med. 211, 2583–2598 (2014).
Sloan, E. K., Capitanio, J. P. & Cole, S. W. Stress-induced remodeling of lymphoid innervation. Brain Behav. Immun. 22, 15–21 (2008).
Elenkov, I. J., Wilder, R. L., Chrousos, G. P. & Vizi, E. S. The sympathetic nerve — an integrative interface between two supersystems: the brain and the immune system. Pharmacol. Rev. 52, 595–638 (2000).
Kohm, A. P. & Sanders, V. M. Norepinephrine and β 2-adrenergic receptor stimulation regulate CD4+ T and B lymphocyte function in vitro and in vivo. Pharmacol. Rev. 53, 487–525 (2001).
Cole, S. et al. Computational identification of gene-social environment interaction at the human IL6 locus. Proc. Natl Acad. Sci. USA 107, 5681–5686 (2010).
Hori, Y. et al. Naftopidil, a selective α1-adrenoceptor antagonist, suppresses human prostate tumor growth by altering interactions between tumor cells and stroma. Cancer Prev. Res. 4, 87–96 (2011).
Calvani, M. et al. Norepinephrine promotes tumor microenvironment reactivity through β3-adrenoreceptors during melanoma progression. Oncotarget 6, 4615–4632 (2015).
Thaker, P. H. et al. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat. Med. 12, 939–944 (2006).
Dal Monte, M. et al. Functional involvement of β3-adrenergic receptors in melanoma growth and vascularization. J. Mol. Med. 91, 1407–1419 (2013).
Sterling, P. in Allostasis, Homeostasis, and the Costs of Physiological Adaptation (ed. Schulkin, J.) 17–64 (Cambridge Univ. Press, 2004).
Chida, Y., Hamer, M., Wardle, J. & Steptoe, A. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nat. Clin. Pract. Oncol. 5, 466–475 (2008).
Powe, D. G. et al. β-blocker drug therapy reduces secondary cancer formation in breast cancer and improves cancer specific survival. Oncotarget 1, 628–638 (2010).
Barron, T. I., Connolly, R. M., Sharp, L., Bennett, K. & Visvanathan, K. β blockers and breast cancer mortality: a population-based study. J. Clin. Oncol. 29, 2635–2644 (2011).
Melhem-Bertrandt, A. et al. β-blocker use is associated with improved relapse-free survival in patients with triple-negative breast cancer. J. Clin. Oncol. 29, 2645–2652 (2011).
De Giorgi, V. et al. Treatment with β-blockers and reduced disease progression in patients with thick melanoma. Arch. Intern. Med. 171, 779–781 (2011).
Lemeshow, S. et al. β-blockers and survival among Danish patients with malignant melanoma: a population-based cohort study. Cancer Epidemiol. Biomarkers Prev. 20, 2273–2279 (2011).
Aydiner, A., Ciftci, R., Karabulut, S. & Kilic, L. Does β-blocker therapy improve the survival of patients with metastatic non-small cell lung cancer? Asian Pac. J. Cancer Prev. 14, 6109–6114 (2013).
Botteri, E. et al. Therapeutic effect of β-blockers in triple-negative breast cancer postmenopausal women. Breast Cancer Res. Treat. 140, 567–575 (2013).
De Giorgi, V. et al. Effect of β-blockers and other antihypertensive drugs on the risk of melanoma recurrence and death. Mayo Clin. Proc. 88, 1196–1203 (2013).
Grytli, H. H., Fagerland, M. W., Fossa, S. D., Tasken, K. A. & Haheim, L. L. Use of β-blockers is associated with prostate cancer-specific survival in prostate cancer patients on androgen deprivation therapy. Prostate 73, 250–260 (2013).
Grytli, H. H., Fagerland, M. W., Fossa, S. D. & Tasken, K. A. Association between use of β-blockers and prostate cancer-specific survival: a cohort study of 3561 prostate cancer patients with high-risk or metastatic disease. Eur. Urol. 65, 635–641 (2014).
Sloan, E. K. et al. The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res. 70, 7042–7052 (2010).
Madden, K. S., Szpunar, M. J. & Brown, E. B. β-adrenergic receptors (β-AR) regulate VEGF and IL-6 production by divergent pathways in high β-AR-expressing breast cancer cell lines. Breast Cancer Res. Treat. 130, 747–758 (2011).
Palm, D. et al. The norepinephrine-driven metastasis development of PC-3 human prostate cancer cells in BALB/c nude mice is inhibited by β-blockers. Int. J. Cancer. 118, 2744–2749 (2006).
Hassan, S. et al. Behavioral stress accelerates prostate cancer development in mice. J. Clin. Invest. 123, 874–886 (2013).
Pasquier, E. et al. β-blockers increase response to chemotherapy via direct antitumour and anti-angiogenic mechanisms in neuroblastoma. Br. J. Cancer 108, 2485–2494 (2013).
Wolter, J. K. et al. Anti-tumor activity of the β-adrenergic receptor antagonist propranolol in neuroblastoma. Oncotarget 5, 161–172 (2014).
Hasegawa, H. & Saiki, I. Psychosocial stress augments tumor development through β-adrenergic activation in mice. Jpn J. Cancer Res. 93, 729–735 (2002).
Goldfarb, Y. et al. Improving postoperative immune status and resistance to cancer metastasis: a combined perioperative approach of immunostimulation and prevention of excessive surgical stress responses. Ann. Surg. 253, 798–810 (2011).
Kim-Fuchs, C. et al. Chronic stress accelerates pancreatic cancer growth and invasion: a critical role for β-adrenergic signaling in the pancreatic microenvironment. Brain Behav. Immun. 40, 40–47 (2014).
Lamkin, D. M. et al. Chronic stress enhances progression of acute lymphoblastic leukemia via β-adrenergic signaling. Brain Behav. Immun. 26, 635–641 (2012).
Inbar, S. et al. Do stress responses promote leukemia progression? An animal study suggesting a role for epinephrine and prostaglandin-E2 through reduced NK activity. PLoS ONE 6, e19246 (2011).
Hara, M. R. et al. A stress response pathway regulates DNA damage through β2-adrenoreceptors and β-arrestin-1. Nature 477, 349–353 (2011).
Hara, M. R., Sachs, B. D., Caron, M. G. & Lefkowitz, R. J. Pharmacological blockade of a β2AR-β-arrestin-1 signaling cascade prevents the accumulation of DNA damage in a behavioral stress model. Cell Cycle 12, 219–224 (2013).
Reeder, A. et al. Stress hormones reduce the efficacy of paclitaxel in triple negative breast cancer through induction of DNA damage. Br. J. Cancer 112, 1461–1470 (2015).
Armaiz-Pena, G. N. et al. Src activation by β-adrenoreceptors is a key switch for tumour metastasis. Nat. Commun. 4, 1403 (2013).
Shi, M. et al. The β2-adrenergic receptor and Her2 comprise a positive feedback loop in human breast cancer cells. Breast Cancer Res. Treat. 125, 351–362 (2011).
Gu, L., Lau, S. K., Loera, S., Somlo, G. & Kane, S. E. Protein kinase A activation confers resistance to trastuzumab in human breast cancer cell lines. Clin. Cancer Res. 15, 7196–7206 (2009).
Chang, M. et al. β-adrenoreceptors reactivate Kaposi's sarcoma-associated herpesvirus lytic replication via PKA-dependent control of viral RTA. J. Virol. 79, 13538–13547 (2005).
zur Hausen, H. Infections Causing Human Cancer, (Wiley-VCH, 2008).
Nilsson, M. B. et al. Stress hormones regulate interleukin-6 expression by human ovarian carcinoma cells through a Src-dependent mechanism. J. Biol. Chem. 282, 29919–29926 (2007).
Shahzad, M. M. et al. Stress effects on FosB- and interleukin-8 (IL8)-driven ovarian cancer growth and metastasis. J. Biol. Chem. 285, 35462–35470 (2010).
Yang, R., Lin, Q., Gao, H. B. & Zhang, P. Stress-related hormone norepinephrine induces interleukin-6 expression in GES-1 cells. Braz. J. Med. Biol. Res. 47, 101–109 (2014).
Cakir, Y., Plummer, H. K., 3rd, Tithof, P. K. & Schuller, H. M. β-adrenergic and arachidonic acid-mediated growth regulation of human breast cancer cell lines. Int. J. Oncol. 21, 153–157 (2002).
Armaiz-Pena, G. N. et al. Adrenergic regulation of monocyte chemotactic protein 1 leads to enhanced macrophage recruitment and ovarian carcinoma growth. Oncotarget 6, 4266–4273 (2015).
Collado-Hidalgo, A., Sung, C. & Cole, S. Adrenergic inhibition of innate anti-viral response: PKA blockade of type I interferon gene transcription mediates catecholamine support for HIV-1 replication. Brain Behav. Immun. 20, 552–563 (2006).
Cole, S. W., Korin, Y. D., Fahey, J. L. & Zack, J. A. Norepinephrine accelerates HIV replication via protein kinase A-dependent effects on cytokine production. J. Immunol. 161, 610–616 (1998).
Glasner, A. et al. Improving survival rates in two models of spontaneous postoperative metastasis in mice by combined administration of a β-adrenergic antagonist and a cyclooxygenase-2 inhibitor. J. Immunol. 184, 2449–2457 (2010).
Lee, J. W. et al. Surgical stress promotes tumor growth in ovarian carcinoma. Clin. Cancer Res. 15, 2695–2702 (2009).
Magnon, C. et al. Autonomic nerve development contributes to prostate cancer progression. Science 341, 1236361 (2013).
Bruzzone, A. et al. α2-adrenoceptors enhance cell proliferation and mammary tumor growth acting through both the stroma and the tumor cells. Curr. Cancer Drug Targets 11, 763–774 (2011).
Flint, M. S. et al. Chronic exposure to stress hormones promotes transformation and tumorigenicity of 3T3 mouse fibroblasts. Stress 16, 114–121 (2013).
Cao, L. et al. Environmental and genetic activation of a brain–adipocyte BDNF/leptin axis causes cancer remission and inhibition. Cell 142, 52–64 (2010).
Cao, L. & During, M. J. What is the brain–cancer connection? Annu. Rev. Neurosci. 35, 331–345 (2012).
Hanoun, M. et al. Acute myelogenous leukemia-induced sympathetic neuropathy promotes malignancy in an altered hematopoietic stem cell niche. Cell Stem Cell 15, 365–375 (2014).
Lang, K. et al. Induction of a metastatogenic tumor cell type by neurotransmitters and its pharmacological inhibition by established drugs. Int. J. Cancer 112, 231–238 (2004).
Drell, T. L. t. et al. Effects of neurotransmitters on the chemokinesis and chemotaxis of MDA-MB-468 human breast carcinoma cells. Breast Cancer Res. Treat. 80, 63–70 (2003).
Landen, C. N. Jr et al. Neuroendocrine modulation of signal transducer and activator of transcription-3 in ovarian cancer. Cancer Res. 67, 10389–10396 (2007).
Sood, A. K. et al. Stress hormone-mediated invasion of ovarian cancer cells. Clin. Cancer Res. 12, 369–375 (2006).
Yang, E. V. et al. Norepinephrine up-regulates the expression of vascular endothelial growth factor, matrix metalloproteinase (MMP)-2, and MMP-9 in nasopharyngeal carcinoma tumor cells. Cancer Res. 66, 10357–10364 (2006).
Chakroborty, D., Sarkar, C., Basu, B., Dasgupta, P. S. & Basu, S. Catecholamines regulate tumor angiogenesis. Cancer Res. 69, 3727–3730 (2009).
Yang, E. V. et al. Norepinephrine upregulates VEGF, IL-8, and IL-6 expression in human melanoma tumor cell lines: implications for stress-related enhancement of tumor progression. Brain Behav. Immun. 23, 267–275 (2009).
Moretti, S. et al. β-adrenoceptors are upregulated in human melanoma and their activation releases pro-tumorigenic cytokines and metalloproteases in melanoma cell lines. Lab Invest. 93, 279–290 (2013).
Liu, J. et al. The effect of chronic stress on anti-angiogenesis of sunitinib in colorectal cancer models. Psychoneuroendocrinology 52, 130–142 (2015).
Sood, A. K. et al. Adrenergic modulation of focal adhesion kinase protects human ovarian cancer cells from anoikis. J. Clin. Invest. 120, 1515–1523 (2010).
Sastry, K. S. et al. Epinephrine protects cancer cells from apoptosis via activation of cAMP-dependent protein kinase and BAD phosphorylation. J. Biol. Chem. 282, 14094–14100 (2007).
Deng, G. H. et al. Exogenous norepinephrine attenuates the efficacy of sunitinib in a mouse cancer model. J. Exp. Clin. Cancer Res. 33, 21 (2014).
Dar, A. et al. Rapid mobilization of hematopoietic progenitors by AMD3100 and catecholamines is mediated by CXCR4-dependent SDF-1 release from bone marrow stromal cells. Leukemia 25, 1286–1296 (2011).
Lucas, D. et al. Chemotherapy-induced bone marrow nerve injury impairs hematopoietic regeneration. Nat. Med. 19, 695–703 (2013).
Lutgendorf, S. K. et al. Social isolation is associated with elevated tumor norepinephrine in ovarian carcinoma patients. Brain Behav. Immun. 25, 250–255 (2011).
Ayala, G. E. et al. Cancer-related axonogenesis and neurogenesis in prostate cancer. Clin. Cancer Res. 14, 7593–7603 (2008).
Voss, M. J. & Entschladen, F. Tumor interactions with soluble factors and the nervous system. Cell Commun. Signal 8, 21 (2010).
Guo, K. et al. Interaction of the sympathetic nerve with pancreatic cancer cells promotes perineural invasion through the activation of STAT3 signaling. Mol. Cancer Ther. 12, 264–273 (2013).
Xu, Q. et al. Stromal-derived factor-1α/CXCL12–CXCR4 chemotactic pathway promotes perineural invasion in pancreatic cancer. Oncotarget 6, 4717–4732 (2015).
Flierl, M. A. et al. Phagocyte-derived catecholamines enhance acute inflammatory injury. Nature. 449, 721–725 (2007).
Campbell, J. P. et al. Stimulation of host bone marrow stromal cells by sympathetic nerves promotes breast cancer bone metastasis in mice. PLoS Biol. 10, e1001363 (2012).
Lu, H. et al. Impact of β-blockers on prostate cancer mortality: a meta-analysis of 16,825 patients. Onco Targets Ther. 8, 985–990 (2015).
Wang, H. M. et al. Improved survival outcomes with the incidental use of β-blockers among patients with non-small-cell lung cancer treated with definitive radiation therapy. Ann. Oncol. 24, 1312–1319 (2013).
Diaz, E. S., Karlan, B. Y. & Li, A. J. Impact of β blockers on epithelial ovarian cancer survival. Gynecol. Oncol. 127, 375–378 (2012).
Watkins, J. L. et al. Clinical impact of selective and non-selective β-blockers on survival in ovarian cancer patients. Cancer (in the press).
Schuller, H. M., Porter, B. & Riechert, A. β-adrenergic modulation of NNK-induced lung carcinogenesis in hamsters. J. Cancer Res. Clin. Oncol. 126, 624–630 (2000).
Pasquier, E. et al. Propranolol potentiates the anti-angiogenic effects and anti-tumor efficacy of chemotherapy agents: implication in breast cancer treatment. Oncotarget 2, 797–809 (2011).
Lin, Q. et al. Effect of chronic restraint stress on human colorectal carcinoma growth in mice. PLoS ONE 8, e61435 (2013).
Ganz, P. A. & Cole, S. W. Expanding our therapeutic options: β blockers for breast cancer? J. Clin. Oncol. 29, 2612–2616 (2011).
Neeman, E., Zmora, O. & Ben-Eliyahu, S. A new approach to reducing postsurgical cancer recurrence: perioperative targeting of catecholamines and prostaglandins. Clin. Cancer Res. 18, 4895–4902 (2012).
Horowitz, M., Neeman, E., Sharon, E. & Ben-Eliyahu, S. Exploiting the critical perioperative period to improve long-term cancer outcomes. Nat. Rev. Clin. Oncol. 12, 213–226 (2015).
Boucek, R. J. Jr., Kirsh, A. L., Majesky, M. W. & Perkins, J. A. Propranolol responsiveness in vascular tumors is not determined by qualitative differences in adrenergic receptors. Otolaryngol. Head Neck Surg. 149, 772–776 (2013).
Mendez-Ferrer, S., Battista, M. & Frenette, P. S. Cooperation of β2- and β3-adrenergic receptors in hematopoietic progenitor cell mobilization. Ann. NY Acad. Sci. 1192, 139–144 (2010).
Magnon, C., Lucas, D. & Frenette, P. S. Trafficking of stem cells. Methods Mol. Biol. 750, 3–24 (2011).
Szpunar, M. J., Burke, K. A., Dawes, R. P., Brown, E. B. & Madden, K. S. The antidepressant desipramine and α2-adrenergic receptor activation promote breast tumor progression in association with altered collagen structure. Cancer Prev. Res. 6, 1262–1272 (2013).
Lamkin, D. M. et al. α2-adrenergic blockade mimics the enhancing effect of chronic stress on breast cancer progression. Psychoneuroendocrinology 51, 262–270 (2015).
Friedman, G. D., Udaltsova, N. & Habel, L. A. Norepinephrine antagonists and cancer risk. Int. J. Cancer 128, 737–738; author reply 739 (2011).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19, 1423–1437 (2013).
Shan, T. et al. β2-adrenoceptor blocker synergizes with gemcitabine to inhibit the proliferation of pancreatic cancer cells via apoptosis induction. Eur. J. Pharmacol. 665, 1–7 (2011).
Obeid, E. I. & Conzen, S. D. The role of adrenergic signaling in breast cancer biology. Cancer Biomark. 13, 161–169 (2013).
Zhao, C. M. et al. Denervation suppresses gastric tumorigenesis. Sci. Transl Med. 6,250ra115 (2014).
Rosas-Ballina, M. et al. Splenic nerve is required for cholinergic antiinflammatory pathway control of TNF in endotoxemia. Proc. Natl Acad. Sci. USA 105, 11008–11013 (2008).
Villanueva, M. T. Therapeutics: gastric cancer gets a red carpet treatment. Nat. Rev. Cancer 14, 648–649 (2014).
Acknowledgements
This work was supported by grants from the United States National Institutes of Health (CA083639, CA098258, CA104825, CA109298, CA116778, CA140933, CA151668, CA177909, AG017265, AG033590) and Department of Defense (OC120547, OC093416), the Betty Ann Asche Murray Distinguished Professorship, the Cancer Prevention and Research Institute of Texas (CPRIT RP140106), and the Breast Cancer Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Cole, S., Nagaraja, A., Lutgendorf, S. et al. Sympathetic nervous system regulation of the tumour microenvironment. Nat Rev Cancer 15, 563–572 (2015). https://doi.org/10.1038/nrc3978
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3978
This article is cited by
-
Perineural invasion in colorectal cancer: mechanisms of action and clinical relevance
Cellular Oncology (2024)
-
Elucidation of the mechanisms underlying tumor aggravation by the activation of stress-related neurons in the paraventricular nucleus of the hypothalamus
Molecular Brain (2023)
-
Chronic stress in solid tumor development: from mechanisms to interventions
Journal of Biomedical Science (2023)
-
Environmental signals perceived by the brain abate pro-metastatic monocytes by dampening glucocorticoids receptor signaling
Cancer Cell International (2023)
-
Association between pretreatment emotional distress and neoadjuvant immune checkpoint blockade response in melanoma
Nature Medicine (2023)