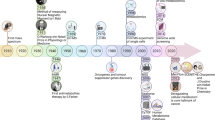
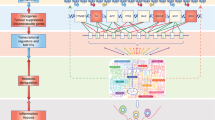
Abstract
Cancer is a prime example of a disease process in which carcinogenic and metabolic changes are intertwined to promote cell survival and growth. One approach to unravel this complex relationship is by studying rare, monogenic disorders caused by mutations in genes encoding metabolic enzymes or regulators. There are hundreds of these diseases, most of which manifest in childhood and are collectively termed 'inborn errors of metabolism' (IEMs). Several IEMs demonstrate the consequences of chronic, systemic loss of a particular metabolic activity that can result in malignancy. In this Opinion article, we present a conceptual categorization of IEMs associated with cancer and discuss how assessment of these rare diseases might inform us about the biological foundations of common types of cancer and opportunities for cancer diagnosis and therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Warburg, O. On the origin of cancer cells. Science 123, 309–314 (1956).
DeBerardinis, R. J., Sayed, N., Ditsworth, D. & Thompson, C. B. Brick by brick: metabolism and tumor cell growth. Curr. Opin. Genet. Dev. 18, 54–61 (2008).
Boroughs, L. K. & DeBerardinis, R. J. Metabolic pathways promoting cancer cell survival and growth. Nat. Cell Biol. 17, 351–359 (2015).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Garrod, A. The Croonian lectures on inborn errors of metabolism, lecture II: alkaptonuria. Lancet 2, 73–79 (1908).
Garrod, A. E. The incidence of alkaptonuria: a study in chemical individuality. 1902. Mol. Med. 2, 274–282 (1996).
Beadle, G. W. & Tatum, E. L. Genetic control of biochemical reactions in Neurospora. Proc. Natl Acad. Sci. USA 27, 499–506 (1941).
Hamosh, A. et al. Online Mendelian Inheritance in Man (OMIM), a knowledgebase of human genes and genetic disorders. Nucleic Acids Res. 30, 52–55 (2002).
Pampols, T. Inherited metabolic rare disease. Adv. Exp. Med. Biol. 686, 397–431 (2010).
Dipple, K. M. & McCabe, E. R. Phenotypes of patients with “simple” Mendelian disorders are complex traits: thresholds, modifiers, and systems dynamics. Am. J. Hum. Genet. 66, 1729–1735 (2000).
Lanpher, B., Brunetti-Pierri, N. & Lee, B. Inborn errors of metabolism: the flux from Mendelian to complex diseases. Nat. Rev. Genet. 7, 449–460 (2006).
Jeffery, C. J. Moonlighting proteins — an update. Mol. Biosyst. 5, 345–350 (2009).
Erez, A., Shchelochkov, O. A., Plon, S. E., Scaglia, F. & Lee, B. Insights into the pathogenesis and treatment of cancer from inborn errors of metabolism. Am. J. Hum. Genet. 88, 402–421 (2011).
Baysal, B. E. et al. Prevalence of SDHB, SDHC, and SDHD germline mutations in clinic patients with head and neck paragangliomas. J. Med. Genet. 39, 178–183 (2002).
Burnichon, N. et al. SDHA is a tumor suppressor gene causing paraganglioma. Hum. Mol. Genet. 19, 3011–3020 (2010).
Guzy, R. D., Sharma, B., Bell, E., Chandel, N. S. & Schumacker, P. T. Loss of the SdhB, but not the SdhA, subunit of complex II triggers reactive oxygen species-dependent hypoxia-inducible factor activation and tumorigenesis. Mol. Cell. Biol. 28, 718–731 (2008).
Kaelin, W. G. Jr. SDH5 mutations and familial paraganglioma: somewhere Warburg is smiling. Cancer Cell 16, 180–182 (2009).
Ricketts, C. J. et al. Tumor risks and genotype–phenotype–proteotype analysis in 358 patients with germline mutations in SDHB and SDHD. Hum. Mutat. 31, 41–51 (2010).
Bayley, J. P., Launonen, V. & Tomlinson, I. P. The FH mutation database: an online database of fumarate hydratase mutations involved in the MCUL (HLRCC) tumor syndrome and congenital fumarase deficiency. BMC Med. Genet. 9, 20 (2008).
Zheng, L. et al. Fumarate induces redox-dependent senescence by modifying glutathione metabolism. Nat. Commun. 6, 6001 (2015).
Wellen, K. E. & Thompson, C. B. A two-way street: reciprocal regulation of metabolism and signalling. Nat. Rev. Mol. Cell Biol. 13, 270–276 (2012).
Garber, J. E. & Offit, K. Hereditary cancer predisposition syndromes. J. Clin. Oncol. 23, 276–292 (2005).
Samuel, N., Villani, A., Fernandez, C. V. & Malkin, D. Management of familial cancer: sequencing, surveillance and society. Nat. Rev. Clin. Oncol. 11, 723–731 (2014).
Cizmarova, M. et al. Rasopathies — dysmorphic syndromes with short stature and risk of malignancy. Endocr. Regul. 47, 217–222 (2013).
Schiffman, J. D. et al. Update on pediatric cancer predisposition syndromes. Pediatr. Blood Cancer 60, 1247–1252 (2013).
Levy, P. A. An overview of newborn screening. J. Dev. Behav. Pediatr. 31, 622–631 (2010).
Gottlieb, E. & Tomlinson, I. P. Mitochondrial tumour suppressors: a genetic and biochemical update. Nat. Rev. Cancer 5, 857–866 (2005).
Litten, J. B. & Tomlinson, G. E. Liver tumors in children. Oncologist 13, 812–820 (2008).
Nelson, R. L., Davis, F. G., Persky, V. & Becker, E. Risk of neoplastic and other diseases among people with heterozygosity for hereditary hemochromatosis. Cancer 76, 875–879 (1995).
Lithner, F. & Wetterberg, L. Hepatocellular carcinoma in patients with acute intermittent porphyria. Acta Med. Scand. 215, 271–274 (1984).
Savas, N. et al. Hepatocellular carcinoma in Wilson's disease: a rare association in childhood. Pediatr. Transplant. 10, 639–643 (2006).
Yin, P. H. et al. Alteration of the copy number and deletion of mitochondrial DNA in human hepatocellular carcinoma. Br. J. Cancer 90, 2390–2396 (2004).
Cassiman, D. et al. Bilateral renal cell carcinoma development in long-term Fabry disease. J. Inherit. Metab. Dis. 30, 830–831 (2007).
Xu, R. et al. Hepatocellular carcinoma in type 1 Gaucher disease: a case report with review of the literature. Semin. Liver Dis. 25, 226–229 (2005).
Osborne, N. J. et al. HFE C282Y homozygotes are at increased risk of breast and colorectal cancer. Hepatology 51, 1311–1318 (2010).
Harrison, S. A. & Bacon, B. R. Relation of hemochromatosis with hepatocellular carcinoma: epidemiology, natural history, pathophysiology, screening, treatment, and prevention. Med. Clin. North Am. 89, 391–409 (2005).
Bartlett, D. C., Lloyd, C., McKiernan, P. J. & Newsome, P. N. Early nitisinone treatment reduces the need for liver transplantation in children with tyrosinaemia type 1 and improves post-transplant renal function. J. Inherit. Metab. Dis. 37, 745–752 (2014).
Yang, M., Soga, T. & Pollard, P. J. Oncometabolites: linking altered metabolism with cancer. J. Clin. Invest. 123, 3652–3658 (2013).
Selak, M. A. et al. Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-α prolyl hydroxylase. Cancer Cell 7, 77–85 (2005).
Tuppen, H. A., Blakely, E. L., Turnbull, D. M. & Taylor, R. W. Mitochondrial DNA mutations and human disease. Biochim. Biophys. Acta 1797, 113–128 (2010).
Figueroa, M. E. et al. DNA methylation signatures identify biologically distinct subtypes in acute myeloid leukemia. Cancer Cell 17, 13–27 (2010).
Rakheja, D., Medeiros, L. J., Bevan, S. & Chen, W. The emerging role of d-2-hydroxyglutarate as an oncometabolite in hematolymphoid and central nervous system neoplasms. Front. Oncol. 3, 169 (2013).
Letouze, E. et al. SDH mutations establish a hypermethylator phenotype in paraganglioma. Cancer Cell 23, 739–752 (2013).
Sullivan, L. B. et al. The proto-oncometabolite fumarate binds glutathione to amplify ROS-dependent signaling. Mol. Cell 51, 236–248 (2013).
Ooi, A. et al. An antioxidant response phenotype shared between hereditary and sporadic type 2 papillary renal cell carcinoma. Cancer Cell 20, 511–523 (2011).
Adam, J. et al. Renal cyst formation in Fh1-deficient mice is independent of the Hif/Phd pathway: roles for fumarate in KEAP1 succination and Nrf2 signaling. Cancer Cell 20, 524–537 (2011).
Singh, A. K., Pandey, P., Tewari, M., Pandey, H. P. & Shukla, H. S. Human mitochondrial genome flaws and risk of cancer. Mitochondrial DNA 25, 329–334 (2014).
Carew, J. S. & Huang, P. Mitochondrial defects in cancer. Mol. Cancer 1, 9 (2002).
Scheers, I., Bachy, V., Stephenne, X. & Sokal, E. M. Risk of hepatocellular carcinoma in liver mitochondrial respiratory chain disorders. J. Pediatr. 146, 414–417 (2005).
Kunz, B. A. Mutagenesis and deoxyribonucleotide pool imbalance. Mutat. Res. 200, 133–147 (1988).
Kulawiec, M. et al. Tumorigenic transformation of human breast epithelial cells induced by mitochondrial DNA depletion. Cancer Biol. Ther. 7, 1732–1743 (2008).
Dimmock, D. et al. Citrin deficiency, a perplexing global disorder. Mol. Genet. Metab. 96, 44–49 (2009).
Nagasaka, H. et al. Sustaining hypercitrullinemia, hypercholesterolemia and augmented oxidative stress in Japanese children with aspartate/glutamate carrier isoform 2-citrin-deficiency even during the silent period. Mol. Genet. Metab. 97, 21–26 (2009).
Kimura, A. et al. Histological findings in the livers of patients with neonatal intrahepatic cholestasis caused by citrin deficiency. Hepatol. Res. 40, 295–303 (2010).
Fukuhara, N., Tokiguchi, S., Shirakawa, K. & Tsubaki, T. Myoclonus epilepsy associated with ragged-red fibres (mitochondrial abnormalities): disease entity or a syndrome? Light-and electron-microscopic studies of two cases and review of literature. J. Neurol. Sci. 47, 117–133 (1980).
Holme, E. et al. Multiple symmetric lipomas with high levels of mtDNA with the tRNA(Lys) A-->G(8344) mutation as the only manifestation of disease in a carrier of myoclonus epilepsy and ragged-red fibers (MERRF) syndrome. Am. J. Hum. Genet. 52, 551–556 (1993).
Bourgeron, T. et al. Mutation of a nuclear succinate dehydrogenase gene results in mitochondrial respiratory chain deficiency. Nat. Genet. 11, 144–149 (1995).
Kerrigan, J. F., Aleck, K. A., Tarby, T. J., Bird, C. R. & Heidenreich, R. A. Fumaric aciduria: clinical and imaging features. Ann. Neurol. 47, 583–588 (2000).
Gimenez-Roqueplo, A. P. New advances in the genetics of pheochromocytoma and paraganglioma syndromes. Ann. NY Acad. Sci. 1073, 112–121 (2006).
Tomlinson, I. P. et al. Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat. Genet. 30, 406–410 (2002).
Stratakis, C. A. & Carney, J. A. The triad of paragangliomas, gastric stromal tumours and pulmonary chondromas (Carney triad), and the dyad of paragangliomas and gastric stromal sarcomas (Carney-Stratakis syndrome): molecular genetics and clinical implications. J. Intern. Med. 266, 43–52 (2009).
Pollard, P. J. et al. Accumulation of Krebs cycle intermediates and over-expression of HIF1α in tumours which result from germline FH and SDH mutations. Hum. Mol. Genet. 14, 2231–2239 (2005).
Adam, J. et al. A role for cytosolic fumarate hydratase in urea cycle metabolism and renal neoplasia. Cell Rep. 3, 1440–1448 (2013).
Zheng, L. et al. Reversed argininosuccinate lyase activity in fumarate hydratase-deficient cancer cells. Cancer Metab. 1, 12 (2013).
Alam, N. A. et al. Genetic and functional analyses of FH mutations in multiple cutaneous and uterine leiomyomatosis, hereditary leiomyomatosis and renal cancer, and fumarate hydratase deficiency. Hum. Mol. Genet. 12, 1241–1252 (2003).
Losman, J. A. & Kaelin, W. G. Jr. What a difference a hydroxyl makes: mutant IDH, (R)-2-hydroxyglutarate, and cancer. Genes Dev. 27, 836–852 (2013).
Rogers, R. E. et al. Wilms tumor in a child with L-2-hydroxyglutaric aciduria. Pediatr. Dev. Pathol. 13, 408–411 (2010).
Moroni, I. et al. L-2-hydroxyglutaric aciduria and brain malignant tumors: a predisposing condition? Neurology 62, 1882–1884 (2004).
Van Schaftingen, E., Rzem, R. & Veiga-da-Cunha, M. L-2-Hydroxyglutaric aciduria, a disorder of metabolite repair. J. Inherit. Metab. Dis. 32, 135–142 (2009).
Kranendijk, M., Struys, E. A., Salomons, G. S., Van der Knaap, M. S. & Jakobs, C. Progress in understanding 2-hydroxyglutaric acidurias. J. Inherit. Metab. Dis. 35, 571–587 (2012).
Kranendijk, M. et al. IDH2 mutations in patients with d-2-hydroxyglutaric aciduria. Science 330, 336 (2010).
Nota, B. et al. Deficiency in SLC25A1, encoding the mitochondrial citrate carrier, causes combined d-2- and L-2-hydroxyglutaric aciduria. Am. J. Hum. Genet. 92, 627–631 (2013).
Koivunen, P. et al. Transformation by the (R)-enantiomer of 2-hydroxyglutarate linked to EGLN activation. Nature 483, 484–488 (2012).
Lei, K. J., Shelly, L. L., Pan, C. J., Sidbury, J. B. & Chou, J. Y. Mutations in the glucose-6-phosphatase gene that cause glycogen storage disease type 1a. Science 262, 580–583 (1993).
Rake, J. P. et al. Glycogen storage disease type I: diagnosis, management, clinical course and outcome. Results of the European Study on Glycogen Storage Disease Type I (ESGSD I). Eur. J. Pediatr. 161, S20–S34 (2002).
Chou, J. Y. & Mansfield, B. C. Mutations in the glucose-6-phosphatase-alpha (G6PC) gene that cause type Ia glycogen storage disease. Hum. Mutat. 29, 921–930 (2008).
Di Rocco, M. et al. Hepatocellular adenoma and metabolic balance in patients with type Ia glycogen storage disease. Mol. Genet. Metab. 93, 398–402 (2008).
Park, S. K., Haase, V. H. & Johnson, R. S. von Hippel Lindau tumor suppressor regulates hepatic glucose metabolism by controlling expression of glucose transporter 2 and glucose 6-phosphatase. Int. J. Oncol. 30, 341–348 (2007).
Nicholson, L. J. et al. Epigenetic silencing of argininosuccinate synthetase confers resistance to platinum-induced cell death but collateral sensitivity to arginine auxotrophy in ovarian cancer. Int. J. Cancer 125, 1454–1463 (2009).
Dillon, B. J. et al. Incidence and distribution of argininosuccinate synthetase deficiency in human cancers: a method for identifying cancers sensitive to arginine deprivation. Cancer 100, 826–833 (2004).
Delage, B. et al. Arginine deprivation and argininosuccinate synthetase expression in the treatment of cancer. Int. J. Cancer 126, 2762–2772 (2010).
Yang, H., Zhai, G., Ji, X., Su, J. & Lin, M. Reduced expression of argininosuccinate lyase is closely associated with postresectional survival in hepatocellular carcinoma: an immunohistochemistry study of 61 cases. Appl. Immunohistochem. Mol. Morphol. 20, 602–606 (2012).
Kobayashi, E. et al. Reduced argininosuccinate synthetase is a predictive biomarker for the development of pulmonary metastasis in patients with osteosarcoma. Mol. Cancer Ther. 9, 535–544 (2010).
Syed, N. et al. Epigenetic status of argininosuccinate synthetase and argininosuccinate lyase modulates autophagy and cell death in glioblastoma. Cell Death Dis. 4, e458 (2013).
Lincet, H. & Icard, P. How do glycolytic enzymes favour cancer cell proliferation by nonmetabolic functions? Oncogene http://dx.doi.org/10.1038/onc.2014.320 (2014).
Erez, A. Argininosuccinic aciduria: from a monogenic to a complex disorder. Genet. Med. 15, 251–257 (2013).
Majewski, N. et al. Hexokinase–mitochondria interaction mediated by Akt is required to inhibit apoptosis in the presence or absence of Bax and Bak. Mol. Cell 16, 819–830 (2004).
Danial, N. N. et al. BAD and glucokinase reside in a mitochondrial complex that integrates glycolysis and apoptosis. Nature 424, 952–956 (2003).
Yang, J., Li, J. H., Wang, J. & Zhang, C. Y. Molecular modeling of BAD complex resided in a mitochondrion integrating glycolysis and apoptosis. J. Theor. Biol. 266, 231–241 (2010).
Niinaka, Y., Paku, S., Haga, A., Watanabe, H. & Raz, A. Expression and secretion of neuroleukin/phosphohexose isomerase/maturation factor as autocrine motility factor by tumor cells. Cancer Res. 58, 2667–2674 (1998).
Fruman, D. A. & Rommel, C. PI3K and cancer: lessons, challenges and opportunities. Nat. Rev. Drug Discov. 13, 140–156 (2014).
Guo, C., Liu, S. & Sun, M. Z. Novel insight into the role of GAPDH playing in tumor. Clin. Transl. Oncol. 15, 167–172 (2013).
Tristan, C., Shahani, N., Sedlak, T. W. & Sawa, A. The diverse functions of GAPDH: views from different subcellular compartments. Cell Signal. 23, 317–323 (2011).
Iqbal, M. A., Gupta, V., Gopinath, P., Mazurek, S. & Bamezai, R. N. Pyruvate kinase M2 and cancer: an updated assessment. FEBS Lett. 588, 2685–2692 (2014).
Li, B. et al. Fructose-1,6-bisphosphatase opposes renal carcinoma progression. Nature 513, 251–255 (2014).
Giovannini, M., Biasucci, G., Luotti, D., Fiori, L. & Riva, E. Nutrition in children affected by inherited metabolic diseases. Ann. Ist. Super. Sanita 31, 489–502 (1995).
Seyfried, T. N., Flores, R., Poff, A. M., D'Agostino, D. P. & Mukherjee, P. Metabolic therapy: a new paradigm for managing malignant brain cancer. Cancer Lett. 356, 289–300 (2015).
Klement, R. J. Restricting carbohydrates to fight head and neck cancer — is this realistic? Cancer Biol. Med. 11, 145–161 (2014).
Yang, Y. et al. Increased intake of vegetables, but not fruit, reduces risk for hepatocellular carcinoma: a meta-analysis. Gastroenterology 147, 1031–1042 (2014).
Schwingshackl, L. & Hoffmann, G. Adherence to Mediterranean diet and risk of cancer: a systematic review and meta-analysis of observational studies. Int. J. Cancer 135, 1884–1897 (2014).
Shils, M. E. Principles of nutritional therapy. Cancer 43, 2093–2102 (1979).
Wilcken, B. An introduction to nutritional treatment in inborn errors of metabolism — different disorders, different approaches. Southeast Asian J. Trop. Med. Publ. Health 34 (Suppl. 3), 198–201 (2003).
Nagamani, S. C. et al. A randomized controlled trial to evaluate the effects of high-dose versus low-dose of arginine therapy on hepatic function tests in argininosuccinic aciduria. Mol. Genet. Metab. 107, 315–321 (2012).
Walker, V. Ammonia toxicity and its prevention in inherited defects of the urea cycle. Diabetes Obes. Metab. 11, 823–835 (2009).
Locasale, J. W. et al. Phosphoglycerate dehydrogenase diverts glycolytic flux and contributes to oncogenesis. Nat. Genet. 43, 869–874 (2011).
Johnson, S. C. et al. mTOR inhibition alleviates mitochondrial disease in a mouse model of Leigh syndrome. Science 342, 1524–1528 (2013).
Folling, I. The discovery of phenylketonuria. Acta Paediatr. Suppl. 407, 4–10 (1994).
Guthrie, R. & Susi, A. A. Simple phenylalanine method for detecting phenylketonuria in large populations of newborn infants. Pediatrics 32, 338–343 (1963).
Woo, S. L., Lidsky, A. S., Guttler, F., Chandra, T. & Robson, K. J. Cloned human phenylalanine hydroxylase gene allows prenatal diagnosis and carrier detection of classical phenylketonuria. Nature 306, 151–155 (1983).
Pearson, P. et al. The status of online Mendelian inheritance in man (OMIM) medio 1994. Nucleic Acids Res. 22, 3470–3473 (1994).
Flier, J. S., Mueckler, M. M., Usher, P. & Lodish, H. F. Elevated levels of glucose transport and transporter messenger RNA are induced by ras or src oncogenes. Science 235, 1492–1495 (1987).
DiLella, A. G., Huang, W. M. & Woo, S. L. Screening for phenylketonuria mutations by DNA amplification with the polymerase chain reaction. Lancet 1, 497–499 (1988).
Goldwater, R. S. Gaucher disease, enzyme replacement therapy, and the Patient Assistance Program. J. Intraven Nurs. 19, 83–88 (1996).
Afaq, A., Syed, R. & Bomanji, J. PET/MRI: a new technology in the field of molecular imaging. Br. Med. Bull. 108, 159–171 (2013).
Gimm, O., Armanios, M., Dziema, H., Neumann, H. P. & Eng, C. Somatic and occult germ-line mutations in SDHD, a mitochondrial complex II gene, in nonfamilial pheochromocytoma. Cancer Res. 60, 6822–6825 (2000).
Desnick, R. J. Enzyme replacement therapy for Fabry disease: lessons from two α-galactosidase A orphan products and one FDA approval. Expert Opin. Biol. Ther. 4, 1167–1176 (2004).
Matoba, S. et al. p53 regulates mitochondrial respiration. Science 312, 1650–1653 (2006).
Rivkees, S. A. “The Newborn Screening Saves Lives Act” — four million calls for support! J. Pediatr. Endocrinol. Metab. 20, 457–458 (2007).
Parsons, D. W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, 1807–1812 (2008).
Dang, L. et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462, 739–744 (2009).
Yan, H. et al. IDH1 and IDH2 mutations in gliomas. N. Engl. J. Med. 360, 765–773 (2009).
Wang, F. et al. Targeted inhibition of mutant IDH2 in leukemia cells induces cellular differentiation. Science 340, 622–626 (2013).
Acknowledgements
The authors thank D. Valle and B. Lee for sharing their insights on the mechanisms by which IEMs lead to tissue dysfunction. R.J.D. is the Joel B. Steinberg Chair of Pediatrics at the University of Texas Southwestern Medical Center and is supported by grants from the US National Institutes of Health (CA157996) and the Cancer Prevention and Research Institute of Texas (RP130272). He is on the scientific advisory boards of Agios Pharmaceuticals and Peloton Therapeutics. A.E. is the incumbent Leah Omenn Career Development Chair and is supported by research grants from the European research program (CIG618113, ERC614204), the Israel Science Foundation (1343/13; 1952/13) and the Minerva grant award (711730). A.E. received additional support from the Adelis Foundation, the Henry S. and Anne S. Reich Research Fund, the Dukler Fund for Cancer Research, the Paul Sparr Foundation, the Saul and Theresa Esman Foundation, Joseph Piko Baruch and the estate of Fannie Sherr.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
R.J.D. is a member of the Scientific Advisory Board of Agios Pharmaceuticals and Peloton Therapeutics, two companies interested in cancer metabolism. A.E. declares no competing interests.
Related links
DATABASES
Rights and permissions
About this article
Cite this article
Erez, A., DeBerardinis, R. Metabolic dysregulation in monogenic disorders and cancer — finding method in madness. Nat Rev Cancer 15, 440–448 (2015). https://doi.org/10.1038/nrc3949
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3949
This article is cited by
-
Participation of protein metabolism in cancer progression and its potential targeting for the management of cancer
Amino Acids (2023)
-
ASS1 and ASL suppress growth in clear cell renal cell carcinoma via altered nitrogen metabolism
Cancer & Metabolism (2021)
-
The role of metabolic ecosystem in cancer progression — metabolic plasticity and mTOR hyperactivity in tumor tissues
Cancer and Metastasis Reviews (2021)
-
ASL expression in ALDH1A1+ neurons in the substantia nigra metabolically contributes to neurodegenerative phenotype
Human Genetics (2021)
-
Late-onset multiple acyl-CoA dehydrogenase deficiency with breast cancer
Egyptian Journal of Medical Human Genetics (2020)