Key Points
-
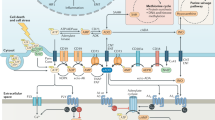
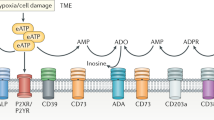
The adenosinergic pathway encompasses ectonucleotidases (CD39 and CD73) and adenosine receptors (A1R, A2AR, A2BR and A3R) that participate in the generation and signalling of adenosine in the tumour microenvironment (TME), respectively. Of the four adenosine receptors, the cyclic AMP (cAMP)-activating receptors A2AR and A2BR predominantly exert immunosuppressive functions in the TME.
-
Molecules of this pathway are regulated by several immunogenic and genetic drivers. Of these, hypoxia and transforming growth factor-β (TGFβ) represent the key drivers of the adenosinergic pathway.
-
Within a tumour niche, adenosinergic molecules are expressed by tumour cells, stromal cells and immune cells, and their critical point of action is not yet fully understood.
-
Adenosine, through activation of cAMP, can directly regulate tumour proliferation, survival, adhesion and migration. In immune cells, adenosine molecules greatly hamper vital immune effector cell functions and may be involved in mediating T cell exhaustion.
-
Novel antibodies and small molecules targeted to members of the adenosinergic pathway are now reaching clinical trials in patients with advanced cancer and may be combined with standard-of-care therapies and novel immunotherapies.
Abstract
Despite the success of anti-programmed cell death protein 1 (PD1), anti-PD1 ligand 1 (PDL1) and anti-cytotoxic T lymphocyte antigen 4 (CTLA4) therapies in advanced cancer, a considerable proportion of patients remain unresponsive to these treatments (known as innate resistance). In addition, one-third of patients relapse after initial response (known as adaptive resistance), which suggests that multiple non-redundant immunosuppressive mechanisms coexist within the tumour microenvironment. A major immunosuppressive mechanism is the adenosinergic pathway, which now represents an attractive new therapeutic target for cancer therapy. Activation of this pathway occurs within hypoxic tumours, where extracellular adenosine exerts local suppression through tumour-intrinsic and host-mediated mechanisms. Preclinical studies in mice with adenosine receptor antagonists and antibodies have reported favourable antitumour immune responses with some definition of the mechanism of action. Currently, agents targeting the adenosinergic pathway are undergoing first-in-human clinical trials as single agents and in combination with anti-PD1 or anti-PDL1 therapies. In this Review, we describe the complex interplay of adenosine and adenosine receptors in the development of primary tumours and metastases and discuss the merits of targeting one or more components that compose the adenosinergic pathway. We also review the early clinical data relating to therapeutic agents inhibiting the adenosinergic pathway.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
22 November 2017
When the article was initially published online, reference 80 was incorrectly listed in the reference list. This has now been corrected in the print and online versions of the article.
References
Hatfield, S. M. et al. Immunological mechanisms of the antitumor effects of supplemental oxygenation. Sci. Transl Med. 7, 277ra30 (2015). This is a landmark paper that elegantly demonstrates that respiratory hyperoxia can provide a therapeutic benefit in cancer by preventing hypoxia and subsequent adenosine production, thus boosting antitumour immune responses.
Hatfield, S. M. et al. Systemic oxygenation weakens the hypoxia and hypoxia inducible factor 1α-dependent and extracellular adenosine-mediated tumor protection. J. Mol. Med. 92, 1283–1292 (2014).
Semenza, G. L. HIF-1 mediates metabolic responses to intratumoral hypoxia and oncogenic mutations. J. Clin. Invest. 123, 3664–3671 (2013).
Thomlinson, R. H. & Gray, L. H. The histological structure of some human lung cancers and the possible implications for radiotherapy. Br. J. Cancer. 9, 539–549 (1955).
Busse, M. & Vaupel, P. Accumulation of purine catabolites in solid tumors exposed to therapeutic hyperthermia. Experientia 52, 469–473 (1996).
Blay, J., White, T. D. & Hoskin, D. W. The extracellular fluid of solid carcinomas contains immunosuppressive concentrations of adenosine. Cancer Res. 57, 2602–2605 (1997).
Stagg, J. & Smyth, M. J. Extracellular adenosine triphosphate and adenosine in cancer. Oncogene 29, 5346–5358 (2010).
Ohta, A. A metabolic immune checkpoint: adenosine in tumor microenvironment. Front. Immunol. 7, 109 (2016).
Ruiz, M. L., Lim, Y.-H. & Zheng, J. Adenosine A2A receptors as drug discovery target. J. Med. Chem. 57, 3623–3650 (2014).
Ohta, A. & Sitkovsky, M. Role of G-protein-coupled adenosine receptors in downregulation of inflammation and protection from tissue damage. Nature 414, 916–920 (2001). This is the first study demonstrating the role of adenosine, A2AR and cAMP in the immune responses to autoimmunity and viral hepatitis and, importantly, paved the way for future investigations into the role of A2AR as a potential cancer immunotherapeutic target.
Ohta, A. et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc. Natl Acad. Sci. USA 103, 13132–13137 (2006). This is the first study to provide genetic and pharmacological evidence of how A2AR expression in T cells regulates tumour immunity.
Vecchio, E. A. et al. Ligand-independent adenosine A2B receptor constitutive activity as a promoter of prostate cancer cell proliferation. J. Pharmacol. Exp. Ther. 357, 36–44 (2016).
Cronstein, B. N. et al. Neutrophil adherence to endothelium is enhanced via adenosine A1 receptors and inhibited via adenosine A2 receptors. J. Immunol. 148, 2201–2206 (1992).
Butler, M. et al. Impairment of adenosine A3 receptor activity disrupts neutrophil migratory capacity and impacts innate immune function in vivo. Eur. J. Immunol. 42, 3358–3368 (2012).
Allard, D., Allard, B., Gaudreau, P. O., Chrobak, P. & Stagg, J. CD73-adenosine: a next-generation target in immuno-oncology. Immunotherapy 8, 145–163 (2016).
Allard, B., Longhi, M. S., Robson, S. C. & Stagg, J. The ectonucleotidases CD39 and CD73: novel checkpoint inhibitor targets. Immunol. Rev. 276, 121–144 (2017).
Young, A., Mittal, D., Stagg, J. & Smyth, M. J. Targeting cancer-derived adenosine: new therapeutic approaches. Cancer Discov. 4, 879–888 (2014).
Beavis, P. A. et al. Blockade of A2A receptors potently suppresses the metastasis of CD73+ tumors. Proc. Natl Acad. Sci. USA 110, 14711–14716 (2013).
Cekic, C., Day, Y. J., Sag, D. & Linden, J. Myeloid expression of adenosine A2A receptor suppresses T and NK cell responses in the solid tumor microenvironment. Cancer Res. 74, 7250–7259 (2014). This study shows that tumour-associated myeloid cells all express immunosuppressive A2AR, which is a potential target of adenosine receptor inhibitors to improve immune control of tumours.
Young, A. et al. Co-inhibition of CD73 and A2AR adenosine signaling improves anti-tumor immune responses. Cancer Cell. 30, 391–403 (2016). The non-redundant nature of adenosinergic molecules is tested in this study for the first time and highlights the potential to co-target CD73 and A2AR molecules in cancer treatment.
Mittal, D. et al. Adenosine 2B receptor expression on cancer cells promotes metastasis. Cancer Res. 76, 4372–4382 (2016). This is a critical paper that thoroughly delineates host and tumour-intrinsic roles of A2BR in the progression of metastasis through the use of A2BR- deficient mice and A2BR-knockdown tumour cells.
Ryzhov, S. et al. Host A2B adenosine receptors promote carcinoma growth. Neoplasia 10, 987–995 (2008).
Stagg, J. et al. CD73-deficient mice have increased antitumor immunity and are resistant to experimental metastasis. Cancer Res. 71, 2892–2900 (2011).
Bastid, J. et al. Inhibition of CD39 enzymatic function at the surface of tumor cells alleviates their immunosuppressive activity. Cancer Immunol. Res. 3, 254–265 (2015). This is an insightful study that thoroughly characterizes the expression of CD39 across several human tumours and cancer cell lines.
Borsellino, G. et al. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood 110, 1225–1232 (2007).
Figueiro, F. et al. Phenotypic and functional characteristics of CD39high human regulatory B cells (Breg). Oncoimmunology 5, e1082703 (2016).
Ryzhov, S. V. et al. Role of TGF-β signaling in generation of CD39+CD73+ myeloid cells in tumors. J. Immunol. 193, 3155–3164 (2014).
Mascanfroni, I. D. et al. IL-27 acts on DCs to suppress the T cell response and autoimmunity by inducing expression of the immunoregulatory molecule CD39. Nat. Immunol. 14, 1054–1063 (2013).
Gupta, P. K. et al. CD39 Expression identifies terminally exhausted CD8+ T cells. PLoS Pathog. 11, e1005177 (2015).
Sun, X. et al. CD39/ENTPD1 expression by CD4+Foxp3+ regulatory T cells promotes hepatic metastatic tumor growth in mice. Gastroenterology 139, 1030–1040 (2010). This paper shows that CD39 expression on regulatory T cells inhibits NK cell activity and is permissive for metastatic growth.
Jackson, S. W. et al. Disordered purinergic signaling inhibits pathological angiogenesis in Cd39/Entpd1-null mice. Am. J. Pathol. 171, 1395–1404 (2007).
Vijayan, D. et al. Selective activation of anti-CD73 mechanisms in control of primary tumors and metastases. Oncoimmunology 6, e1312044 (2017).
Stagg, J. et al. CD73-deficient mice are resistant to carcinogenesis. Cancer Res. 72, 2190–2196 (2012).
Beavis, P. A. et al. Targeting the adenosine 2A receptor enhances chimeric antigen receptor T cell efficacy. J. Clin. Invest. 127, 929–941 (2017). The potential to use the combination of CAR T cells with A2AR inhibition is demonstrated in this study.
Mittal, D. et al. Antimetastatic effects of blocking PD-1 and the adenosine A2A receptor. Cancer Res. 74, 3652–3658 (2014).
Waickman, A. T. et al. Enhancement of tumor immunotherapy by deletion of the A2A adenosine receptor. Cancer Immunol. Immunother. 61, 917–926 (2012).
Ohta, A. & Sitkovsky, M. Extracellular adenosine-mediated modulation of regulatory T cells. Front. Immunol. 5, 304 (2014).
Iannone, R., Miele, L., Maiolino, P., Pinto, A. & Morello, S. Blockade of A2b adenosine receptor reduces tumor growth and immune suppression mediated by myeloid-derived suppressor cells in a mouse model of melanoma. Neoplasia 15, 1400–1409 (2013).
Sorrentino, C., Miele, L., Porta, A., Pinto, A. & Morello, S. Myeloid-derived suppressor cells contribute to A2B adenosine receptor-induced VEGF production and angiogenesis in a mouse melanoma model. Oncotarget 6, 27478–27489 (2015). This study shows the effect of A2BR inhibition on myeloid cells and angiogenesis.
Young, A. et al. Targeting adenosine in BRAF-mutant melanoma reduces tumor growth and metastasis. Cancer Res. 77, 4684–4696 (2017).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02403193 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02655822 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02740985 (2017).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02503774 (2017).
Biswas, S. K. Metabolic reprogramming of immune cells in cancer progression. Immunity 43, 435–449 (2015).
Synnestvedt, K. et al. Ecto-5′-nucleotidase (CD73) regulation by hypoxia-inducible factor-1 mediates permeability changes in intestinal epithelia. J. Clin. Invest. 110, 993–1002 (2002).
Tak, E. et al. Protective role of hypoxia-inducible factor-1α-dependent CD39 and CD73 in fulminant acute liver failure. Toxicol. Appl. Pharmacol. 314, 72–81 (2017).
Kong, T., Westerman, K. A., Faigle, M., Eltzschig, H. K. & Colgan, S. P. HIF-dependent induction of adenosine A2B receptor in hypoxia. FASEB J. 20, 2242–2250 (2006).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Klein, M. & Bopp, T. Cyclic AMP represents a crucial component of Treg cell-mediated immune regulation. Front. Immunol. 7, 315 (2016).
Wang, P. et al. MicroRNA-128b suppresses tumor growth and promotes apoptosis by targeting A2bR in gastric cancer. Biochem. Biophys. Res. Commun. 467, 798–804 (2015).
Morello, S. et al. IL-1β and TNF-α regulation of the adenosine receptor (A2A) expression: differential requirement for NF-κB binding to the proximal promoter. J. Immunol. 177, 7173–7183 (2006).
Inoue, Y. et al. Prognostic impact of CD73 and A2A adenosine receptor expression in non-small-cell lung cancer. Oncotarget 8, 8738–8751 (2017).
Elliott, M. R. et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461, 282–286 (2009).
Galluzzi, L., Buque, A., Kepp, O., Zitvogel, L. & Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 17, 97–111 (2017).
Jin, D. et al. CD73 on tumor cells impairs antitumor T-cell responses: a novel mechanism of tumor-induced immune suppression. Cancer Res. 70, 2245–2255 (2010).
Zhi, X. et al. RNA interference of ecto-5′-nucleotidase (CD73) inhibits human breast cancer cell growth and invasion. Clin. Exp. Metastasis 24, 439–448 (2007).
Turcotte, M. et al. CD73 is associated with poor prognosis in high-grade serous ovarian cancer. Cancer Res. 75, 4494–4503 (2015). This study highlights a role for CD73 as a prognostic marker of patient survival and as a candidate therapeutic target in advanced serous ovarian cancers.
Sadej, R., Spychala, J. & Skladanowski, A. C. Expression of ecto-5′-nucleotidase (eN, CD73) in cell lines from various stages of human melanoma. Melanoma Res. 16, 213–222 (2006).
Terp, M. G. et al. Anti-human CD73 monoclonal antibody inhibits metastasis formation in human breast cancer by inducing clustering and internalization of CD73 expressed on the surface of cancer cells. J. Immunol. 191, 4165–4173 (2013).
Wang, L. et al. Ecto-5′-nucleotidase promotes invasion, migration and adhesion of human breast cancer cells. J. Cancer Res. Clin. Oncol. 134, 365–372 (2008).
Merighi, S. et al. Adenosine receptors as mediators of both cell proliferation and cell death of cultured human melanoma cells. J. Invest. Dermatol. 119, 923–933 (2002).
Etique, N., Grillier-Vuissoz, I., Lecomte, J. & Flament, S. Crosstalk between adenosine receptor (A2A isoform) and ERα mediates ethanol action in MCF-7 breast cancer cells. Oncol. Rep. 21, 977–981 (2009).
Auchampach, J. A. Adenosine receptors and angiogenesis. Circ. Res. 101, 1075–1077 (2007).
Feng, L. et al. Vascular CD39/ENTPD1 directly promotes tumor cell growth by scavenging extracellular adenosine triphosphate. Neoplasia 13, 206–216 (2011).
Allard, B. et al. Anti-CD73 therapy impairs tumor angiogenesis. Int. J. Cancer. 134, 1466–1473 (2014).
Wang, L. et al. CD73 has distinct roles in nonhematopoietic and hematopoietic cells to promote tumor growth in mice. J. Clin. Invest. 121, 2371–2382 (2011).
Yang, Q., Du, J. & Zu, L. Overexpression of CD73 in prostate cancer is associated with lymph node metastasis. Pathol. Oncol. Res. 19, 811–814 (2013).
Clayton, A., Al-Taei, S., Webber, J., Mason, M. D. & Tabi, Z. Cancer exosomes express CD39 and CD73, which suppress T cells through adenosine production. J. Immunol. 187, 676–683 (2011).
Srinivasan, S., Vannberg, F. O. & Dixon, J. B. Lymphatic transport of exosomes as a rapid route of information dissemination to the lymph node. Sci. Rep. 6, 24436 (2016).
Takedachi, M. et al. CD73-generated adenosine restricts lymphocyte migration into draining lymph nodes. J. Immunol. 180, 6288–6296 (2008).
Airas, L., Niemela, J. & Jalkanen, S. CD73 engagement promotes lymphocyte binding to endothelial cells via a lymphocyte function-associated antigen-1-dependent mechanism. J. Immunol. 165, 5411–5417 (2000).
Speiser, D. E., Ho, P. C. & Verdeil, G. Regulatory circuits of T cell function in cancer. Nat. Rev. Immunol. 16, 599–611 (2016).
Guillerey, C., Huntington, N. D. & Smyth, M. J. Targeting natural killer cells in cancer immunotherapy. Nat. Immunol. 17, 1025–1036 (2016).
Raskovalova, T. et al. Gs protein-coupled adenosine receptor signaling and lytic function of activated NK cells. J. Immunol. 175, 4383–4391 (2005).
Ohta, A. et al. A2A adenosine receptor may allow expansion of T cells lacking effector functions in extracellular adenosine-rich microenvironments. J. Immunol. 183, 5487–5493 (2009).
Hilchey, S. P. et al. Human follicular lymphoma CD39+-infiltrating T cells contribute to adenosine-mediated T cell hyporesponsiveness. J. Immunol. 183, 6157–6166 (2009).
Ngiow, S. F. et al. Agonistic CD40 mAb-driven IL12 reverses resistance to anti-PD1 in a T-cell-rich tumor. Cancer Res. 76, 6266–6277 (2016).
Kalekar, L. A. et al. CD4+ T cell anergy prevents autoimmunity and generates regulatory T cell precursors. Nat. Immunol. 17, 304–314 (2016).
Li, L. et al. Dendritic cells tolerized with adenosine A2AR agonist attenuate acute kidney injury. J. Clin. Invest. 122, 3931–3942 (2012).
Armstrong, J. M. et al. Gene dose effect reveals no Gs-coupled A2A adenosine receptor reserve in murine T-lymphocytes: studies of cells from A2A-receptor-gene-deficient mice. Biochem. J. 354, 123–130 (2001).
Sevigny, C. P. et al. Activation of adenosine 2A receptors attenuates allograft rejection and alloantigen recognition. J. Immunol. 178, 4240–4249 (2007).
Allard, B., Pommey, S., Smyth, M. J. & Stagg, J. Targeting CD73 enhances the antitumor activity of anti-PD-1 and anti-CTLA-4 mAbs. Clin. Cancer Res. 19, 5626–5635 (2013).
Zarek, P. E. et al. A2A receptor signaling promotes peripheral tolerance by inducing T-cell anergy and the generation of adaptive regulatory T cells. Blood 111, 251–259 (2008).
Sitkovsky, M. V., Kjaergaard, J., Lukashev, D. & Ohta, A. Hypoxia-adenosinergic immunosuppression: tumor protection by T regulatory cells and cancerous tissue hypoxia. Clin. Cancer Res. 14, 5947–5952 (2008).
Montalbán Del Barrio, I. et al. Adenosine-generating ovarian cancer cells attract myeloid cells which differentiate into adenosine-generating tumor associated macrophages — a self-amplifying, CD39- and CD73-dependent mechanism for tumor immune escape. J. Immunother. Cancer 4, 49 (2016).
de Lourdes Mora-Garcia, M. et al. Mesenchymal stromal cells derived from cervical cancer produce high amounts of adenosine to suppress cytotoxic T lymphocyte functions. J. Transl Med. 14, 302 (2016).
Cekic, C. et al. Adenosine A2B receptor blockade slows growth of bladder and breast tumors. J. Immunol. 188, 198–205 (2012).
Shang, B., Liu, Y., Jiang, S. J. & Liu, Y. Prognostic value of tumor-infiltrating FoxP3+ regulatory T cells in cancers: a systematic review and meta-analysis. Sci. Rep. 5, 15179 (2015).
Choi, H. S. et al. The prognostic effects of tumor infiltrating regulatory T cells and myeloid derived suppressor cells assessed by multicolor flow cytometry in gastric cancer patients. Oncotarget 7, 7940–7951 (2016).
Ohta, A. et al. The development and immunosuppressive functions of CD4+ CD25+ FoxP3+ regulatory T cells are under influence of the adenosine- A2A adenosine receptor pathway. Front. Immunol. 3, 190 (2012).
Zhang, B. et al. Circulating and tumor-infiltrating myeloid-derived suppressor cells in patients with colorectal carcinoma. PLoS ONE 8, e57114 (2013).
Novitskiy, S. V. et al. Adenosine receptors in regulation of dendritic cell differentiation and function. Blood 112, 1822–1831 (2008).
Loi, S. et al. CD73 promotes anthracycline resistance and poor prognosis in triple negative breast cancer. Proc. Natl Acad. Sci. USA 110, 11091–11096 (2013).
Ren, Z. H. et al. CD73 is associated with poor prognosis in HNSCC. Oncotarget 7, 61690–61702 (2016).
Kolachala, V. et al. TNF-α upregulates adenosine 2b (A2b) receptor expression and signaling in intestinal epithelial cells: a basis for A2bR overexpression in colitis. Cell. Mol. Life Sci. 62, 2647–2657 (2005).
Reinhardt, J. et al. MAPK signaling and inflammation link melanoma phenotype switching to induction of CD73 during immunotherapy. Cancer Res. 77, 4697–4709 (2017).
Hung, S. P., Yang, M. H., Tseng, K. F. & Lee, O. K. Hypoxia-induced secretion of TGF-β1 in mesenchymal stem cell promotes breast cancer cell progression. Cell Transplant. 22, 1869–1882 (2013).
Cortez, V. S. et al. Transforming growth factor-β signaling guides the differentiation of innate lymphoid cells in salivary glands. Immunity 44, 1127–1139 (2016).
Regateiro, F. S. et al. Generation of anti-inflammatory adenosine by leukocytes is regulated by TGF-β. Euro. J. Immunol. 41, 2955–2965 (2011).
Olivier, M., Hollstein, M. & Hainaut, P. TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harb. Perspect. Biol. 2, a001008 (2010).
Wu, X. R. et al. High expression of CD73 as a poor prognostic biomarker in human colorectal cancer. J. Surg. Oncol. 106, 130–137 (2012).
Long, J. S. et al. Extracellular adenosine sensing-a metabolic cell death priming mechanism downstream of p53. Mol. Cell. 50, 394–406 (2013).
Emens, L. et al. CPI-444, an oral adenosine A2a receptor (A2aR) antagonist, demonstrates clinical activity in patients with advanced solid tumors [abstract]. Cancer Res. 77 (Suppl. 13), CT119 (2017).
Hay, C. M. et al. Targeting CD73 in the tumor microenvironment with MEDI9447. Oncoimmunology 5, e1208875 (2016). This study demonstrates the preclinical efficacy of MEDI9447, a cross-reacting CD73 mAb that is currently undergoing a phase I clinical trial.
Schindler, U. et al. Novel small-molecule inhibitors of ecto-nucleotidase CD73 promote activation of human CD4+ and CD8+ T cells and have profound effects in experimental tumor models [abstract]. Third CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference, 224 https://static1.squarespace.com/static/56dee71e555986fb3ae583e2/t/59ad08b1b8a79b086c865d6c/1504512189107/CIMT_Abstracts_170904.pdf (2017).
Jaakola, V. P. et al. The 2.6 angstrom crystal structure of a human A2A adenosine receptor bound to an antagonist. Science 322, 1211–1217 (2008).
Borodovsky, A. et al. Preclinical pharmacodynamics and antitumor activity of AZD4635, a novel adenosine 2A receptor inhibitor that reverses adenosine mediated T cell suppression [abstract]. Cancer Res. 77 (Suppl. 13), 5580 (2017).
Walters, M. J. et al. Characterization of the potent and selectiveA2aR antagonist AB928 for the treatment of cancer [abstract]. Cancer Res. 77 (Suppl. 13), 4572 (2017).
Fons, P. et al. Targeting the adenosine immunosuppressive pathway for cancer immunotherapy with small molecule agents [abstract]. Cancer Res. 77 (Suppl. 13), 3970 (2017).
Geoghegan, J. C. et al. Inhibition of CD73 AMP hydrolysis by a therapeutic antibody with a dual, non-competitive mechanism of action. mAbs 8, 454–467 (2016).
Piccione, E. C. et al. A novel CD73-blocking antibody reduces production of immunosuppressive adenosine and restores T cell function [abstract]. Cancer Res. 77 (Suppl. 13), 5577 (2017).
Gruenbacher, G. et al. Ecto-ATPase CD39 inactivates isoprenoid-derived Vγ9Vδ2 T cell phosphoantigens. Cell Rep. 16, 444–456 (2016). This is an informative study that demonstrates a previously unrecognized role for CD39 in the dephosphorylation of T cell phosphoantigens.
Xu, S. et al. Synergy between the ectoenzymes CD39 and CD73 contributes to adenosinergic immunosuppression in human malignant gliomas. Neuro Oncol. 15, 1160–1172 (2013).
Serra, S. et al. CD73-generated extracellular adenosine in chronic lymphocytic leukemia creates local conditions counteracting drug-induced cell death. Blood 118, 6141–6152 (2011).
Fernandez, P. et al. Extracellular generation of adenosine by the ectonucleotidases CD39 and CD73 promotes dermal fibrosis. Am. J. Pathol. 183, 1740–1746 (2013).
Di Virgilio, F. & Adinolfi, E. Extracellular purines, purinergic receptors and tumor growth. Oncogene 36, 293–303 (2017).
Sun, X. et al. Disordered purinergic signaling and abnormal cellular metabolism are associated with development of liver cancer in Cd39/ENTPD1 null mice. Hepatology 57, 205–216 (2013).
Horenstein, A. L. et al. NAD+-metabolizing ectoenzymes in remodeling tumor-host interactions: the human myeloma model. Cells 4, 520–537 (2015).
Vaisitti, T. et al. NAD+-metabolizing ecto-enzymes shape tumor-host interactions: the chronic lymphocytic leukemia model. FEBS Lett. 585, 1514–1520 (2011).
Antonioli, L., Pacher, P., Vizi, E. S. & Hasko, G. CD39 and CD73 in immunity and inflammation. Trends Mol. Med. 19, 355–367 (2013).
Sharif, T. et al. The NAD+ salvage pathway modulates cancer cell viability via p73. Cell Death Differ. 23, 669–680 (2016).
US National Library of Medicine. ClinicalTrial.gov https://clinicaltrials.gov/ct2/show/NCT02944565 (2017).
US National Library of Medicine. ClinicalTrial.gov https://clinicaltrials.gov/ct2/show/NCT01084252 (2017).
Zimmermann, H. Prostatic acid phosphatase, a neglected ectonucleotidase. Purinerg. Signal. 5, 273–275 (2009).
Yegutkin, G. G. et al. Consequences of the lack of CD73 and prostatic acid phosphatase in the lymphoid organs. Mediators Inflamm. 2014, 485743 (2014).
Kim, S. H. et al. Reassessment of alkaline phosphatase as serum tumor marker with high specificity in osteosarcoma. Cancer Med. 6, 1311–1322 (2017).
Rao, S. R. et al. Tumour-derived alkaline phosphatase regulates tumour growth, epithelial plasticity and disease-free survival in metastatic prostate cancer. Br. J. Cancer. 116, 227–236 (2017).
Pettengill, M. et al. Soluble ecto-5′-nucleotidase (5′-NT), alkaline phosphatase, and adenosine deaminase (ADA1) activities in neonatal blood favor elevated extracellular adenosine. J. Biol. Chem. 288, 27315–27326 (2013).
Michaud, M. et al. Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science 334, 1573–1577 (2011).
Iannone, R., Miele, L., Maiolino, P., Pinto, A. & Morello, S. Adenosine limits the therapeutic effectiveness of anti-CTLA4 mAb in a mouse melanoma model. Am. J. Cancer Res. 4, 172–181 (2014).
Gray, L. H., Conger, A. D., Ebert, M., Hornsey, S. & Scott, O. C. The concentration of oxygen dissolved in tissues at the time of irradiation as a factor in radiotherapy. Br. J. Radiol. 26, 638–648 (1953).
Moeller, B. J., Cao, Y., Li, C. Y. & Dewhirst, M. W. Radiation activates HIF-1 to regulate vascular radiosensitivity in tumors: role of reoxygenation, free radicals, and stress granules. Cancer Cell 5, 429–441 (2004).
Cekic, C., Sag, D., Day, Y. J. & Linden, J. Extracellular adenosine regulates naive T cell development and peripheral maintenance. J. Exp. Med. 210, 2693–2706 (2013).
Chen, D. S. & Mellman, I. Oncology meets immunology: the cancer-immunity cycle. Immunity 39, 1–10 (2013).
Moriyama, K. & Sitkovsky, M. V. Adenosine A2A receptor is involved in cell surface expression of A2B receptor. J. Biol. Chem. 285, 39271–39288 (2010).
Stagg, J. et al. Anti-CD73 antibody therapy inhibits breast tumor growth and metastasis. Proc. Natl Acad. Sci. USA 107, 1547–1552 (2010). This study shows the first preclinical demonstration that targeting CD73 might be an effective cancer therapy.
Tan, E. Y., Mujoomdar, M. & Blay, J. Adenosine down-regulates the surface expression of dipeptidyl peptidase IV on HT-29 human colorectal carcinoma cells: implications for cancer cell behavior. Am. J. Pathol. 165, 319–330 (2004).
Sorrentino, C., Miele, L., Porta, A., Pinto, A. & Morello, S. Activation of the A2B adenosine receptor in B16 melanomas induces CXCL12 expression in FAP-positive tumor stromal cells, enhancing tumor progression. Oncotarget 7, 64274–64288 (2016).
Cai, X. Y. et al. Overexpression of CD39 in hepatocellular carcinoma is an independent indicator of poor outcome after radical resection. Medicine 95, e4989 (2016).
Cai, X. Y. et al. Overexpression of CD39 and high tumoral CD39+/CD8+ ratio are associated with adverse prognosis in resectable gastric cancer. Int. J. Clin. Exp. Pathol. 8, 14757–14764 (2015).
Abousamra, N. K., Salah El-Din, M., Hamza Elzahaf, E. & Esmael, M. E. Ectonucleoside triphosphate diphosphohydrolase-1 (E-NTPDase1/CD39) as a new prognostic marker in chronic lymphocytic leukemia. Leuk. Lymphoma 56, 113–119 (2015).
Lu, X. X. et al. Expression and clinical significance of CD73 and hypoxia-inducible factor-1α in gastric carcinoma. World J. Gastroenterol. 19, 1912–1918 (2013).
Zhang, B. et al. The expression and clinical significance of CD73 molecule in human rectal adenocarcinoma. Tumour Biol. 36, 5459–5466 (2015).
Yu, Y. I. et al. Ecto-5′-nucleotidase expression is associated with the progression of renal cell carcinoma. Oncol. Lett. 9, 2485–2494 (2015).
Leclerc, B. G. et al. CD73 expression is an independent prognostic factor in prostate cancer. Clin. Cancer Res. 22, 158–166 (2016). This study shows that CD73 expression in the prostate epithelium suppresses immunosurveillance by CD8+ T cells, whereas CD73 expression in the tumour stroma reduces NF-κB signalling in tumour cells via A2BR signalling.
Wettstein, M. S. et al. CD73 predicts favorable prognosis in patients with nonmuscle-invasive urothelial bladder cancer. Dis. Markers 2015, 785461 (2015).
Bowser, J. L. et al. Loss of CD73-mediated actin polymerization promotes endometrial tumor progression. J. Clin. Invest. 126, 220–238 (2016). This study shows that CD73-generated adenosine promotes epithelial integrity and suggests why loss of CD73 in endometrial cancer enables tumour progression.
Acknowledgements
The authors wish to thank J. Jaen (Arcus Biosciences) and K. Sachsenmeier (AstraZeneca) for helpful discussions. M.J.S. was supported by a National Health and Medical Research Council of Australia (NHMRC) Senior Research Fellowship (1078671), an NHMRC Project Grant (1120887) and a Research Agreement from MedImmune. M.W.L.T. was supported by an NHMRC Project Grant (1120887).
Author information
Authors and Affiliations
Contributions
D.V., M.W.L.T, and M.J.S. researched the data for the article. A.Y. provided a substantial contribution to discussions of the content. D.V. wrote the article, and all authors contributed equally to reviewing and/or editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.J.S. declares scientific research agreements with Bristol-Myers Squibb, Corvus Pharmaceuticals and Aduro Biotech. All other authors declare no conflict of interest.
Glossary
- Hypoxia
-
The disorganized arrangement of blood vessels around a tissue such as cancer, which often results in irregular distribution of oxygen within that tissue; low oxygen levels are often seen in regions of tissues further away from blood vessels.
- Ectonucleotidases
-
Families of nucleotide-metabolizing enzymes that possess an active catalytic site and are expressed on the plasma membrane. These enzymes are associated with the catalysis of nucleotides to their corresponding nucleosides.
- Regulatory T cells
-
(Treg cells). A subpopulation of CD4+ T cells that are involved in modulating inflammation and preventing autoimmunity. However, in the tumour microenvironment, the accumulated presence of these suppressor populations has an important role in impairing antitumour immunity.
- Adoptive cellular therapies
-
(ACTs). Treatments used to help the immune system fight diseases, such as cancer and infections with certain viruses. T cells are collected from a patient and grown ex vivo to increase the number of T cells that are able to kill cancer cells or fight infections. These T cells are then infused back into the patient. Also called cellular adoptive immunotherapy.
- Recurrence-free survival
-
Relating to cancer therapy, refers to the time after a treatment when patients show no signs of disease re-appearance (that is, these patients are cancer-free). This is also called disease-free survival or relapse-free survival.
- Myeloid-derived suppressor cells
-
(MDSCs). A heterogeneous population of myeloid immune cells that originate from the bone marrow and exhibit potent suppressive functions.
- Exosomes
-
Microvesicles of endocytic origin that are secreted by several cells, including tumour cells.
- Mesothelioma
-
An aggressive form of cancer originating around the lining (mesothelium) of organs such as the lungs, abdomen or heart.
- Exhausted or dysfunctional T cells
-
A state of T cells generally associated with progressive loss of T cell effector functions, resulting in exhaustion or dysfunction. Exhausted T cells are commonly observed during many chronic infections and cancer.
- Hyperoxic conditions
-
A condition where cells or tissues are exposed to an elevated concentration of oxygen.
- Stable disease
-
A term commonly used in cancer to describe the condition where tumours neither progress to distant organs nor regress.
- G protein-coupled receptors
-
(GPCRs). Transmembrane receptors that detect extracellular molecules to initiate signalling pathways essential for cellular processes and maintenance of homeostasis.
- Proliferative centres
-
Regions within a tumour microenvironment that are characterized by increased tumour proliferation and are commonly identified by elevated Ki-67 staining.
- Scleroderma
-
An autoimmune condition that affects the connective tissue in the body. Scleroderma commonly results in thickening and hardening of skin in areas such as the hands and face.
- Salvage pathway
-
A pathway in which nucleosides that have been released during RNA and DNA degradation are synthesized to form nucleotides. The activation of this pathway is usually observed in cells or tissues that are unable to undergo de novo synthesis.
Rights and permissions
About this article
Cite this article
Vijayan, D., Young, A., Teng, M. et al. Targeting immunosuppressive adenosine in cancer. Nat Rev Cancer 17, 709–724 (2017). https://doi.org/10.1038/nrc.2017.86
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2017.86
This article is cited by
-
cAMP-PKA/EPAC signaling and cancer: the interplay in tumor microenvironment
Journal of Hematology & Oncology (2024)
-
Immune evasion in cell-based immunotherapy: unraveling challenges and novel strategies
Journal of Biomedical Science (2024)
-
Neutrophils in cancer: dual roles through intercellular interactions
Oncogene (2024)
-
A novel small molecule inhibitor of CD73 triggers immune-mediated multiple myeloma cell death
Blood Cancer Journal (2024)
-
A2AR-mediated CXCL5 upregulation on macrophages promotes NSCLC progression via NETosis
Cancer Immunology, Immunotherapy (2024)