Key Points
-
The spectrum of known and putative oncogenic drivers with companion targeted therapies continues to increase. As broader mutational testing becomes more clinically available, a greater proportion of patients with non-small-cell lung cancer (NSCLC) will be eligible for targeted therapies.
-
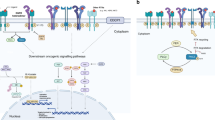
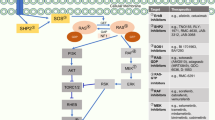
Mechanisms of resistance to targeted therapies can be divided into 'on-target' alterations in the primary drug target and 'off-target' changes that influence downstream and parallel bypass signalling pathways.
-
There is both signalling crosstalk and overlap among downstream and bypass signalling pathways that lead to resistance to the different targeted therapies currently in clinical use for treating NSCLC, suggesting that common themes in the development of drug resistance can be leveraged to guide further development of therapeutic agents and strategies.
-
The upfront combination of therapies targeting both the oncogenic driver and common bypass pathways might delay the onset of disease progression in NSCLC and is the subject of ongoing clinical trials.
-
Bidirectional signalling between tumour cells and components of the tumour microenvironment (TME) shapes both the characteristics of the TME and the propensity of a tumour cell towards therapeutic resistance.
-
Tissue biopsy at disease progression might identify modes of therapeutic resistance to guide the rational selection of subsequent lines of therapy. Serial assessment of circulating tumour DNA might provide a complementary approach to capture heterogeneous and evolving resistance mechanisms in patients.
Abstract
The expanding spectrum of both established and candidate oncogenic driver mutations identified in non-small-cell lung cancer (NSCLC), coupled with the increasing number of clinically available signal transduction pathway inhibitors targeting these driver mutations, offers a tremendous opportunity to enhance patient outcomes. Despite these molecular advances, advanced-stage NSCLC remains largely incurable due to therapeutic resistance. In this Review, we discuss alterations in the targeted oncogene ('on-target' resistance) and in other downstream and parallel pathways ('off-target' resistance) leading to resistance to targeted therapies in NSCLC, and we provide an overview of the current understanding of the bidirectional interactions with the tumour microenvironment that promote therapeutic resistance. We highlight common mechanistic themes underpinning resistance to targeted therapies that are shared by NSCLC subtypes, including those with oncogenic alterations in epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), ROS1 proto-oncogene receptor tyrosine kinase (ROS1), serine/threonine-protein kinase b-raf (BRAF) and other less established oncoproteins. Finally, we discuss how understanding these themes can inform therapeutic strategies, including combination therapy approaches, and overcome the challenge of tumour heterogeneity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Meza, R., Meernik, C., Jeon, J. & Cote, M. L. Lung cancer incidence trends by gender, race and histology in the United States, 1973–2010. PLoS ONE 10, e0121323 (2015).
Kris, M. G. et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA 311, 1998–2006 (2014).
Zhou, C. et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 12, 735–742 (2011).
Solomon, B. J. et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N. Engl. J. Med. 371, 2167–2177 (2014).
Shaw, A. T. et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N. Engl. J. Med. 371, 1963–1971 (2014).
Planchard, D. et al. Dabrafenib plus trametinib in patients with previously treated BRAF(V600E)-mutant metastatic non-small cell lung cancer: an open-label, multicentre phase 2 trial. Lancet Oncol. 17, 984–993 (2016). This paper reported improved survival in BRAF-V600E-mutant NSCLC through the use of combined BRAF and MEK inhibition, providing an example of a combination therapy that is now FDA-approved for use in treating NSCLC.
Bivona, T. G. & Doebele, R. C. A framework for understanding and targeting residual disease in oncogene-driven solid cancers. Nat. Med. 22, 472–478 (2016).
Wu, J. Y. et al. Lung cancer with epidermal growth factor receptor exon 20 mutations is associated with poor gefitinib treatment response. Clin. Cancer Res. 14, 4877–4882 (2008).
Bivona, T. G. et al. FAS and NF-κB signalling modulate dependence of lung cancers on mutant EGFR. Nature 471, 523–526 (2011). This study was among the first to use an unbiased genetic screen to identify a mechanism of EGFR oncogene addiction and innate resistance to EGFR TKI treatment in NSCLC.
Ng, K. P. et al. A common BIM deletion polymorphism mediates intrinsic resistance and inferior responses to tyrosine kinase inhibitors in cancer. Nat. Med. 18, 521–528 (2012).
Alam, N. et al. Small-cell carcinoma with an epidermal growth factor receptor mutation in a never-smoker with gefitinib-responsive adenocarcinoma of the lung. Clin. Lung Cancer 11, E1–E4 (2010).
Yamada, T. et al. Paracrine receptor activation by microenvironment triggers bypass survival signals and ALK inhibitor resistance in EML4-ALK lung cancer cells. Clin. Cancer Res. 18, 3592–3602 (2012).
Budha, N. R. et al. Drug absorption interactions between oral targeted anticancer agents and PPIs: is pH-dependent solubility the Achilles heel of targeted therapy? Clin. Pharmacol. Ther. 92, 203–213 (2012).
Rosell, R. et al. Screening for epidermal growth factor receptor mutations in lung cancer. N. Engl. J. Med. 361, 958–967 (2009).
Sequist, L. V. et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J. Clin. Oncol. 31, 3327–3334 (2013).
Mok, T. S. et al. Osimertinib or Platinum–Pemetrexed in EGFR T790M–Positive Lung Cancer. N. Engl. J. Med. 376, 629–640 (2017).
Kancha, R. K., von Bubnoff, N., Peschel, C. & Duyster, J. Functional analysis of epidermal growth factor receptor (EGFR) mutations and potential implications for EGFR targeted therapy. Clin. Cancer Res. 15, 460–467 (2009).
Lee, C. K. et al. Impact of specific epidermal growth factor receptor (EGFR) mutations and clinical characteristics on outcomes after treatment with EGFR tyrosine kinase inhibitors versus chemotherapy in EGFR-mutant lung cancer: a meta-analysis. J. Clin. Oncol. 33, 1958–1965 (2015).
Yarden, Y. & Sliwkowski, M. X. Untangling the ErbB signalling network. Nat. Rev. Mol. Cell Biol. 2, 127–137 (2001).
Yasuda, H. et al. Structural, biochemical, and clinical characterization of epidermal growth factor receptor (EGFR) exon 20 insertion mutations in lung cancer. Sci. Transl Med. 5, 216ra177 (2013).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02716116 (2017).
Yu, H. A. et al. Poor response to erlotinib in patients with tumors containing baseline EGFR T790M mutations found by routine clinical molecular testing. Ann. Oncol. 25, 423–428 (2014).
Watanabe, S. et al. Effectiveness of gefitinib against non-small-cell lung cancer with the uncommon EGFR mutations G719X and L861Q. J. Thorac. Oncol. 9, 189–194 (2014).
Klughammer, B. et al. Examining treatment outcomes with erlotinib in patients with advanced non-small cell lung cancer whose tumors harbor uncommon EGFR mutations. J. Thorac. Oncol. 11, 545–555 (2016).
Soda, M. et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007).
Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature 511, 543–550 (2014).
Armstrong, F. et al. Differential effects of X-ALK fusion proteins on proliferation, transformation, and invasion properties of NIH3T3 cells. Oncogene 23, 6071–6082 (2004).
Lin, J. J., Riely, G. J. & Shaw, A. T. Targeting ALK: precision medicine takes on drug resistance. Cancer Discov. 7, 137–155 (2017).
Friboulet, L. et al. The ALK inhibitor ceritinib overcomes crizotinib resistance in non-small cell lung cancer. Cancer Discov. 4, 662–673 (2014).
Gainor, J. F. et al. Molecular mechanisms of resistance to first- and second-generation ALK inhibitors in ALK-rearranged lung cancer. Cancer Discov. 6, 1118–1133 (2016). This study characterized the comparative spectrum of secondary ALK mutations occurring at resistance to early-generation and late-generation ALK TKIs.
Zhang, S. et al. The potent ALK inhibitor Brigatinib (AP26113) overcomes mechanisms of resistance to first- and second-generation ALK inhibitors in preclinical models. Clin. Cancer Res. 22, 5527–5538 (2016).
Johnson, T. W. et al. Discovery of (10R)-7-amino-12- fluoro-2,10,16-trimethyl-15-oxo-10,15,16,17- tetrahydro-2H-8,4-(metheno)pyrazolo[4,3-h][2,5,11]- benzoxadiazacyclotetradecine-3-carbonitrile (PF-06463922), a macrocyclic inhibitor of anaplastic lymphoma kinase (ALK) and c-ros oncogene 1 (ROS1) with preclinical brain exposure and broad-spectrum potency against ALK-resistant mutations. J. Med. Chem. 57, 4720–4744 (2014).
Gettinger, S. N. et al. Activity and safety of brigatinib in ALK-rearranged non-small-cell lung cancer and other malignancies: a single-arm, open-label, phase 1/2 trial. Lancet Oncol. 17, 1683–1696 (2016).
Lin, J. J. et al. Clinical activity of Alectinib in advanced RET-rearranged non-small cell lung cancer. J. Thorac. Oncol. 11, 2027–2032 (2016).
Subbiah, V., Hong, D. S. & Meric-Bernstam, F. Clinical activity of ceritinib in ROS1-rearranged non-small cell lung cancer: bench to bedside report. Proc. Natl Acad. Sci. USA 113, E1419–E1420 (2016).
Peters, S. et al. Alectinib versus Crizotinib in untreated ALK-positive non-small-cell lung cancer. N. Engl. J. Med. 377, 829–838 (2017).
Bergethon, K. et al. ROS1 rearrangements define a unique molecular class of lung cancers. J. Clin. Oncol. 30, 863–870 (2012).
Ou, S. H., Tan, J., Yen, Y. & Soo, R. A. ROS1 as a 'druggable' receptor tyrosine kinase: lessons learned from inhibiting the ALK pathway. Expert Rev. Anticancer Ther. 12, 447–456 (2012).
Paik, P. K. et al. Clinical characteristics of patients with lung adenocarcinomas harboring BRAF mutations. J. Clin. Oncol. 29, 2046–2051 (2011).
Imielinski, M. et al. Mapping the hallmarks of lung adenocarcinoma with massively parallel sequencing. Cell 150, 1107–1120 (2012).
Marchetti, A. et al. Clinical features and outcome of patients with non-small-cell lung cancer harboring BRAF mutations. J. Clin. Oncol. 29, 3574–3579 (2011).
Litvak, A. M. et al. Clinical characteristics and course of 63 patients with BRAF mutant lung cancers. J. Thorac. Oncol. 9, 1669–1674 (2014).
Yao, Z. et al. BRAF mutants evade ERK-dependent feedback by different mechanisms that determine their sensitivity to pharmacologic inhibition. Cancer Cell 28, 370–383 (2015).
Planchard, D. et al. Dabrafenib in patients with BRAFV600E-positive advanced non-small-cell lung cancer: a single-arm, multicentre, open-label, phase 2 trial. Lancet. Oncol. 17, 642–650 (2016).
Hyman, D. M. et al. Vemurafenib in multiple nonmelanoma cancers with BRAF V600 mutations. N. Engl. J. Med. 373, 726–736 (2015).
Zhang, C. et al. RAF inhibitors that evade paradoxical MAPK pathway activation. Nature 526, 583–586 (2015).
Okimoto, R. A. et al. Preclinical efficacy of a RAF inhibitor that evades paradoxical MAPK pathway activation in protein kinase BRAF-mutant lung cancer. Proc. Natl Acad. Sci. USA 113, 13456–13461 (2016).
Joshi, M., Rice, S. J., Liu, X., Miller, B. & Belani, C. P. Trametinib with or without vemurafenib in BRAF mutated non-small cell lung cancer. PLoS ONE 10, e0118210 (2015).
Janne, P. A. et al. Selumetinib plus docetaxel compared with docetaxel alone and progression-free survival in patients with kras-mutant advanced non-small cell lung cancer: the SELECT-1 randomized clinical trial. JAMA 317, 1844–1853 (2017).
Tolcher, A. W. et al. Antitumor activity in RAS-driven tumors by blocking AKT and MEK. Clin. Cancer Res. 21, 739–748 (2015).
Manchado, E. et al. A combinatorial strategy for treating KRAS-mutant lung cancer. Nature 534, 647–651 (2016).
Kim, J. Y. et al. Phosphoproteomics reveals MAPK inhibitors enhance MET- and EGFR-driven AKT signaling in KRAS-mutant lung cancer. Mol. Cancer Res. 14, 1019–1029 (2016).
Lin, L. et al. The Hippo effector YAP promotes resistance to RAF- and MEK-targeted cancer therapies. Nat. Genet. 47, 250–256 (2015). A genetic screen in this study identified YAP1 as a protein promoting resistance to BRAF-targeted and MEK-targeted therapies in BRAF-mutant NSCLC.
Tao, Z. et al. Coadministration of trametinib and palbociclib radiosensitizes KRAS-mutant non-small cell lung cancers in vitro and in vivo. Clin. Cancer Res. 22, 122–133 (2016).
Goldman, J. et al. Clinical activity of LY2835219, a novel cell cycle inhibitor selective for CDK4 and CDK6, in patients with non-small cell lung cancer. J. Clin. Oncol. 35, 8026 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02152631 (2017).
Wood, K., Hensing, T., Malik, R. & Salgia, R. Prognostic and predictive value in KRAS in non-small-cell lung cancer: a review. JAMA Oncol. 2, 805–812 (2016).
Hunter, J. C. et al. In situ selectivity profiling and crystal structure of SML-8-73-1, an active site inhibitor of oncogenic K-Ras G12C. Proc. Natl Acad. Sci. USA 111, 8895–8900 (2014).
Lito, P., Solomon, M., Li, L. S., Hansen, R. & Rosen, N. Allele-specific inhibitors inactivate mutant KRAS G12C by a trapping mechanism. Science 351, 604–608 (2016).
Ostrem, J. M., Peters, U., Sos, M. L., Wells, J. A. & Shokat, K. M. K-Ras(G12C) inhibitors allosterically control GTP affinity and effector interactions. Nature 503, 548–551 (2013).
Frampton, G. M. et al. Activation of MET via diverse exon 14 splicing alterations occurs in multiple tumor types and confers clinical sensitivity to MET inhibitors. Cancer Discov. 5, 850–859 (2015).
Gandino, L., Longati, P., Medico, E., Prat, M. & Comoglio, P. M. Phosphorylation of serine 985 negatively regulates the hepatocyte growth factor receptor kinase. J. Biol. Chem. 269, 1815–1820 (1994).
Peschard, P. et al. Mutation of the c-Cbl TKB domain binding site on the Met receptor tyrosine kinase converts it into a transforming protein. Mol. Cell 8, 995–1004 (2001).
Drilon, A. E. et al. Efficacy and safety of crizotinib in patients (pts) with advanced MET exon 14-altered non-small cell lung cancer (NSCLC). J. Clin. Oncol. 34, 108 (2016).
Paik, P. K. et al. Response to MET inhibitors in patients with stage IV lung adenocarcinomas harboring MET mutations causing exon 14 skipping. Cancer Discov. 5, 842–849 (2015).
Cappuzzo, F. et al. Increased MET gene copy number negatively affects survival of surgically resected non-small-cell lung cancer patients. J. Clin. Oncol. 27, 1667–1674 (2009).
Onozato, R. et al. Activation of MET by gene amplification or by splice mutations deleting the juxtamembrane domain in primary resected lung cancers. J. Thorac. Oncol. 4, 5–11 (2009).
Drilon, A., Cappuzzo, F., Ou, S. I. & Camidge, D. R. Targeting MET in lung cancer: will expectations finally be MET? J. Thorac. Oncol. 12, 15–26 (2017).
Camidge, D. R. et al. Efficacy and safety of crizotinib in patients with advanced c-MET-amplified non-small cell lung cancer (NSCLC). J. Clin. Oncol. 32, 8001 (2014).
Mazieres, J. et al. Lung cancer that harbors an HER2 mutation: epidemiologic characteristics and therapeutic perspectives. J. Clin. Oncol. 31, 1997–2003 (2013).
Arcila, M. E. et al. Prevalence, clinicopathologic associations, and molecular spectrum of ERBB2 (HER2) tyrosine kinase mutations in lung adenocarcinomas. Clin. Cancer Res. 18, 4910–4918 (2012).
Chuang, J. C. et al. ERBB2-mutated metastatic non-small cell lung cancer: response and resistance to targeted therapies. J. Thorac. Oncol. 12, 833–842 (2017).
Mazieres, J. et al. Lung cancer patients with HER2 mutations treated with chemotherapy and HER2-targeted drugs: results from the European EUHER2 cohort. Ann. Oncol. 27, 281–286 (2016).
Kris, M. G. et al. Targeting HER2 aberrations as actionable drivers in lung cancers: phase II trial of the pan-HER tyrosine kinase inhibitor dacomitinib in patients with HER2-mutant or amplified tumors. Ann. Oncol. 26, 1421–1427 (2015).
Kosaka, T. et al. Response heterogeneity of EGFR and HER2 exon 20 insertions to covalent EGFR and HER2 inhibitors. Cancer Res. 77, 2712–2721 (2017).
Shan, Y., Arkhipov, A., Kim, E. T., Pan, A. C. & Shaw, D. E. Transitions to catalytically inactive conformations in EGFR kinase. Proc. Natl Acad. Sci. USA 110, 7270–7275 (2013).
Roskoski, R. Jr Anaplastic lymphoma kinase (ALK): structure, oncogenic activation, and pharmacological inhibition. Pharmacol. Res. 68, 68–94 (2013).
Sequist, L. V. et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci. Transl Med. 3, 75ra26 (2011).
Yu, H. A. et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin. Cancer Res. 19, 2240–2247 (2013).
Pao, W. et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2, e73 (2005).
Kobayashi, S. et al. EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 352, 786–792 (2005).
Yun, C. H. et al. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc. Natl Acad. Sci. USA 105, 2070–2075 (2008).
Hata, A. N. et al. Tumor cells can follow distinct evolutionary paths to become resistant to epidermal growth factor receptor inhibition. Nat. Med. 22, 262–269 (2016). This study described preclinical and computational modelling of two distinct evolutionary paths to tumour cell acquisition of the EGFR-T790M mutation; expansion of an existing EGFR-T790M subclone; and de novo acquisition of the EGFR-T790M mutation during EGFR TKI therapy.
Watanabe, M. et al. Ultra-sensitive detection of the pretreatment EGFR T790M mutation in non-small cell lung cancer patients with an EGFR-activating mutation using droplet digital PCR. Clin. Cancer Res. 21, 3552–3560 (2015).
Choi, Y. L. et al. EML4-ALK mutations in lung cancer that confer resistance to ALK inhibitors. N. Engl. J. Med. 363, 1734–1739 (2010).
McCoach, C. E. et al. Resistance mechanisms to targeted therapies in ROS1+and ALK+non-small cell lung cancer. J. Clin. Oncol. 34, 9065 (2016).
Huang, Q. et al. Preclinical modeling of KIF5B-RET fusion lung adenocarcinoma. Mol. Cancer Ther. 15, 2521–2529 (2016).
Cross, D. A. et al. AZD9291, an irreversible EGFR TKI, overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discov. 4, 1046–1061 (2014).
Yu, H. A. et al. Acquired resistance of EGFR-mutant lung cancer to a T790M-specific EGFR inhibitor: emergence of a third mutation (C797S) in the EGFR tyrosine kinase domain. JAMA Oncol. 1, 982–984 (2015). This paper described the EGFR-C797S mutation as a mechanism of acquired resistance to third-generation EGFR TKI therapy.
Thress, K. S. et al. Acquired EGFR C797S mutation mediates resistance to AZD9291 in non-small cell lung cancer harboring EGFR T790M. Nat. Med. 21, 560–562 (2015).
Ou, S. I. et al. Emergence of novel and dominant acquired EGFR solvent-front mutations at Gly796 (G796S/R) together with C797S/R and L792F/H mutations in one EGFR (L858R/T790M) NSCLC patient who progressed on osimertinib. Lung Cancer 108, 228–231 (2017).
Heuckmann, J. M. et al. ALK mutations conferring differential resistance to structurally diverse ALK inhibitors. Clin. Cancer Res. 17, 7394–7401 (2011).
Katayama, R. et al. Mechanisms of acquired crizotinib resistance in ALK-rearranged lung cancers. Sci. Transl Med. 4, 120ra117 (2012).
Awad, M. M. et al. Acquired resistance to crizotinib from a mutation in CD74-ROS1. N. Engl. J. Med. 368, 2395–2401 (2013).
Gainor, J. et al. Frequency and spectrum of ROS1 resistance mutations in ROS1-positive lung cancer patients progressing on crizotinib. J. Clin. Oncol. 34, 9072 (2016).
Drilon, A. et al. A novel crizotinib-resistant solvent-front mutation responsive to cabozantinib therapy in a patient with ROS1-rearranged lung cancer. Clin. Cancer Res. 22, 2351–2358 (2016).
Russo, M. et al. Acquired resistance to the TRK inhibitor entrectinib in colorectal cancer. Cancer Discov. 6, 36–44 (2016).
Bean, J. et al. Acquired resistance to epidermal growth factor receptor kinase inhibitors associated with a novel T854A mutation in a patient with EGFR-mutant lung adenocarcinoma. Clin. Cancer Res. 14, 7519–7525 (2008).
Balak, M. N. et al. Novel D761Y and common secondary T790M mutations in epidermal growth factor receptor-mutant lung adenocarcinomas with acquired resistance to kinase inhibitors. Clin. Cancer Res. 12, 6494–6501 (2006).
Chiba, M. et al. Efficacy of irreversible EGFR-TKIs for the uncommon secondary resistant EGFR mutations L747S, D761Y, and T854A. BMC Cancer 17, 281 (2017).
Doebele, R. C. et al. Mechanisms of resistance to crizotinib in patients with ALK gene rearranged non-small cell lung cancer. Clin. Cancer Res. 18, 1472–1482 (2012).
Sasaki, T. et al. The neuroblastoma-associated F1174L ALK mutation causes resistance to an ALK kinase inhibitor in ALK-translocated cancers. Cancer Res. 70, 10038–10043 (2010).
Katayama, R. et al. Two novel ALK mutations mediate acquired resistance to the next-generation ALK inhibitor alectinib. Clin. Cancer Res. 20, 5686–5696 (2014).
Facchinetti, F. et al. Crizotinib-resistant ROS1 mutations reveal a predictive kinase inhibitor sensitivity model for ROS1- and ALK-rearranged lung cancers. Clin. Cancer Res. 22, 5983–5991 (2016). This paper discussed the use of structural homology among ROS1 and ALK to predict potential ROS1 TKI activity against secondary ROS1 resistance mutations.
Heist, R. S. et al. Acquired resistance to crizotinib in NSCLC with MET exon 14 skipping. J. Thorac. Oncol. 11, 1242–1245 (2016).
Ou, S. I. et al. Emergence of preexisting MET Y1230C mutation as a resistance mechanism to crizotinib in NSCLC with MET exon 14 skipping. J. Thorac. Oncol. 12, 137–140 (2017).
Cui, J. J. Targeting receptor tyrosine kinase MET in cancer: small molecule inhibitors and clinical progress. J. Med. Chem. 57, 4427–4453 (2014).
Bahcall, M. et al. Acquired METD1228V mutation and resistance to MET inhibition in lung cancer. Cancer Discov. 6, 1334–1341 (2016).
Yan, S. B. et al. LY2801653 is an orally bioavailable multi-kinase inhibitor with potent activity against MET, MST1R, and other oncoproteins, and displays anti-tumor activities in mouse xenograft models. Invest. New Drugs 31, 833–844 (2013).
Li, A. N. et al. Acquired MET Y1248H and D1246N mutations mediate resistance to MET inhibitors in non-small cell lung cancer. Clin. Cancer Res. 23, 4929–4937 (2017).
Niederst, M. J. et al. The allelic context of the C797S mutation acquired upon treatment with third-generation EGFR inhibitors impacts sensitivity to subsequent treatment strategies. Clin. Cancer Res. 21, 3924–3933 (2015).
Uchibori, K. et al. Brigatinib combined with anti-EGFR antibody overcomes osimertinib resistance in EGFR-mutated non-small-cell lung cancer. Nat. Commun. 8, 14768 (2017). This study demonstrated the potential for the combination of brigatinib with an EGFR monoclonal antibody to overcome EGFR-C797S-mediated resistance to EGFR TKI therapy.
Jia, Y. et al. Overcoming EGFR(T790M) and EGFR(C797S) resistance with mutant-selective allosteric inhibitors. Nature 534, 129–132 (2016).
Shaw, A. T. et al. Resensitization to crizotinib by the lorlatinib ALK resistance mutation L1198F. N. Engl. J. Med. 374, 54–61 (2016).
Kim, T. M. et al. Mechanisms of acquired resistance to AZD9291: a mutation-selective, irreversible EGFR inhibitor. J. Thorac. Oncol. 10, 1736–1744 (2015).
Katayama, R. et al. Therapeutic strategies to overcome crizotinib resistance in non-small cell lung cancers harboring the fusion oncogene EML4-ALK. Proc. Natl Acad. Sci. USA 108, 7535–7540 (2011).
Lin, L. et al. Mapping the molecular determinants of BRAF oncogene dependence in human lung cancer. Proc. Natl Acad. Sci. USA 111, E748–E757 (2014).
Ohashi, K. et al. Lung cancers with acquired resistance to EGFR inhibitors occasionally harbor BRAF gene mutations but lack mutations in KRAS, NRAS, or MEK1. Proc. Natl Acad. Sci. USA 109, E2127–E2133 (2012).
de Bruin, E. C. et al. Reduced NF1 expression confers resistance to EGFR inhibition in lung cancer. Cancer Discov. 4, 606–619 (2014).
Tricker, E. M. et al. Combined EGFR/MEK inhibition prevents the emergence of resistance in EGFR mutant lung cancer. Cancer Discov. 5, 960–971 (2015). This study reported a delayed acquisition of resistance to EGFR TKI therapy through the use of a combined therapy with EGFR TKIs and MEK inhibitors in preclinical models.
Ercan, D. et al. Reactivation of ERK signaling causes resistance to EGFR kinase inhibitors. Cancer Discov. 2, 934–947 (2012).
Ho, C. C. et al. Acquired BRAF V600E mutation as resistant mechanism after treatment with osimertinib. J. Thorac. Oncol. 12, 567–572 (2017).
Eberlein, C. A. et al. Acquired resistance to the mutant-selective EGFR inhibitor AZD9291 is associated with increased dependence on RAS signaling in preclinical models. Cancer Res. 75, 2489–2500 (2015).
Hrustanovic, G. et al. RAS-MAPK dependence underlies a rational polytherapy strategy in EML4- ALK-positive lung cancer. Nat. Med. 21, 1038–1047 (2015). This study identified the RAS–MAPK pathway as the predominant driver of tumour cell survival during treatment with ALK TKIs, and it demonstrated improved responses with a combination therapy using ALK and MEK inhibitors.
Crystal, A. S. et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science 346, 1480–1486 (2014).
Cargnelutti, M. et al. Activation of RAS family members confers resistance to ROS1 targeting drugs. Oncotarget 6, 5182–5194 (2015).
Engelman, J. A. et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 316, 1039–1043 (2007).
Ludovini, V. et al. Phosphoinositide-3-kinase catalytic alpha and KRAS mutations are important predictors of resistance to therapy with epidermal growth factor receptor tyrosine kinase inhibitors in patients with advanced non-small cell lung cancer. J. Thorac. Oncol. 6, 707–715 (2011).
Sos, M. L. et al. PTEN loss contributes to erlotinib resistance in EGFR-mutant lung cancer by activation of Akt and EGFR. Cancer Res. 69, 3256–3261 (2009).
Shen, H. et al. TGF-β1 induces erlotinib resistance in non-small cell lung cancer by down-regulating PTEN. Biomed. Pharmacother. 77, 1–6 (2016).
Engelman, J. A. et al. Allelic dilution obscures detection of a biologically significant resistance mutation in EGFR-amplified lung cancer. J. Clin. Invest. 116, 2695–2706 (2006).
Deng, Q. F. et al. Integrin β1-mediated acquired gefitinib resistance in non-small cell lung cancer cells occurs via the phosphoinositide 3-kinase-dependent pathway. Oncol. Lett. 11, 535–542 (2016).
Ihle, N. T. et al. The phosphatidylinositol-3-kinase inhibitor PX-866 overcomes resistance to the epidermal growth factor receptor inhibitor gefitinib in A-549 human non-small cell lung cancer xenografts. Mol. Cancer Ther. 4, 1349–1357 (2005).
Karachaliou, N. et al. BIM and mTOR expression levels predict outcome to erlotinib in EGFR-mutant non-small-cell lung cancer. Scientif. Rep. 5, 17499 (2015).
Kawabata, S. et al. Rapamycin prevents the development and progression of mutant epidermal growth factor receptor lung tumors with the acquired resistance mutation T790M. Cell Rep. 7, 1824–1832 (2014).
Gandhi, L. et al. MA04.02 neratinib ± temsirolimus in HER2-mutant lung cancers: an international, randomized phase II study. J. Thorac. Oncol. 12, S358–S359 (2017).
Blakely, C. M. et al. NF-κB-activating complex engaged in response to EGFR oncogene inhibition drives tumor cell survival and residual disease in lung cancer. Cell Rep. 11, 98–110 (2015). This paper described NF- κ B activation as a key signalling event underlying early tumour cell survival during EGFR TKI therapy.
Gao, S. P. et al. JAK2 inhibition sensitizes resistant EGFR-mutant lung adenocarcinoma to tyrosine kinase inhibitors. Sci. Signal. 9, ra33 (2016).
Chaib, I. et al. Co-activation of STAT3 and YES-associated protein 1 (YAP1) pathway in EGFR-mutant NSCLC. J. Natl Cancer Inst. http://dx.doi.org/10.1093/jnci/djx014 (2017).
Shou, J. et al. Cyclosporine A sensitizes human non-small cell lung cancer cells to gefitinib through inhibition of STAT3. Cancer Lett. 379, 124–133 (2016).
Song, L., Rawal, B., Nemeth, J. A. & Haura, E. B. JAK1 activates STAT3 activity in non-small-cell lung cancer cells and IL-6 neutralizing antibodies can suppress JAK1-STAT3 signaling. Mol. Cancer Ther. 10, 481–494 (2011).
Kim, S. M. et al. Activation of IL-6R/JAK1/STAT3 signaling induces de novo resistance to irreversible EGFR inhibitors in non-small cell lung cancer with T790M resistance mutation. Mol. Cancer Ther. 11, 2254–2264 (2012).
Yu, H. A. et al. A phase 1/2 trial of ruxolitinib and erlotinib in patients with EGFR-mutant lung adenocarcinomas with acquired resistance to erlotinib. J. Thorac. Oncol. 12, 102–109 (2017).
Lee, H. J. et al. Drug resistance via feedback activation of Stat3 in oncogene-addicted cancer cells. Cancer Cell 26, 207–221 (2014).
Wheeler, D. L., Iida, M. & Dunn, E. F. The role of Src in solid tumors. Oncologist 14, 667–678 (2009).
Kanda, R. et al. Erlotinib resistance in lung cancer cells mediated by integrin beta1/Src/Akt-driven bypass signaling. Cancer Res. 73, 6243–6253 (2013).
Yoshida, T. et al. Effects of Src inhibitors on cell growth and epidermal growth factor receptor and MET signaling in gefitinib-resistant non-small cell lung cancer cells with acquired MET amplification. Cancer Sci. 101, 167–172 (2010).
Gold, K. A. et al. A phase I/II study combining erlotinib and dasatinib for non-small cell lung cancer. Oncologist 19, 1040–1041 (2014).
Zhao, Y., Yang, Y., Xu, Y., Lu, S. & Jian, H. AZD0530 sensitizes drug-resistant ALK-positive lung cancer cells by inhibiting SRC signaling. FEBS Open Bio 7, 472–476 (2017).
Trusolino, L., Bertotti, A. & Comoglio, P. M. MET signalling: principles and functions in development, organ regeneration and cancer. Nat. Rev. Mol. Cell Biol. 11, 834–848 (2010).
Bean, J. et al. MET amplification occurs with or without T790M mutations in EGFR mutant lung tumors with acquired resistance to gefitinib or erlotinib. Proc. Natl Acad. Sci. USA 104, 20932–20937 (2007).
Ou, S. I. et al. Phase I results from a study of crizotinib in combination with erlotinib in patients with advanced nonsquamous non-small cell lung cancer. J. Thorac. Oncol. 12, 145–151 (2017).
Sequist, L. V. et al. Randomized phase II study of erlotinib plus tivantinib versus erlotinib plus placebo in previously treated non-small-cell lung cancer. J. Clin. Oncol. 29, 3307–3315 (2011).
Wu, Y.-L. et al. Safety and efficacy of INC280 in combination with gefitinib (gef) in patients with EGFR-mutated (mut), MET-positive NSCLC: a single-arm phase lb/ll study. J. Clin. Oncol. 32, 8017 (2014).
Gouji, T., Takashi, S., Mitsuhiro, T. & Yukito, I. Crizotinib can overcome acquired resistance to CH5424802: is amplification of the MET gene a key factor? J. Thorac. Oncol. 9, e27–e28 (2014).
Scaltriti, M., Elkabets, M. & Baselga, J. Molecular pathways: AXL, a membrane receptor mediator of resistance to therapy. Clin. Cancer Res. 22, 1313–1317 (2016).
Zhang, Z. et al. Activation of the AXL kinase causes resistance to EGFR-targeted therapy in lung cancer. Nat. Genet. 44, 852–860 (2012).
Byers, L. A. et al. An epithelial-mesenchymal transition gene signature predicts resistance to EGFR and PI3K inhibitors and identifies Axl as a therapeutic target for overcoming EGFR inhibitor resistance. Clin. Cancer Res. 19, 279–290 (2013).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02424617 (2017).
Wilson, F. H. et al. A functional landscape of resistance to ALK inhibition in lung cancer. Cancer Cell 27, 397–408 (2015).
Nelson-Taylor, S. K. et al. Resistance to RET-inhibition in RET-rearranged NSCLC is mediated by reactivation of RAS/MAPK signaling. Mol. Cancer Ther. 16, 1623–1633 (2017).
Dong, X., Fernandez-Salas, E., Li, E. & Wang, S. Elucidation of resistance mechanisms to second-generation ALK inhibitors alectinib and ceritinib in non-small cell lung cancer cells. Neoplasia 18, 162–171 (2016).
Song, A. et al. Molecular changes associated with acquired resistance to crizotinib in ROS1-rearranged non-small cell lung cancer. Clin. Cancer Res. 21, 2379–2387 (2015).
Davies, K. D. et al. Resistance to ROS1 inhibition mediated by EGFR pathway activation in non-small cell lung cancer. PLoS ONE 8, e82236 (2013).
Vaishnavi, A. et al. EGFR mediates responses to small molecule drugs targeting oncogenic fusion kinases. Cancer Res. 77, 3551–3563 (2017). This study reported bypass activation of EGFR signalling as a shared resistance mechanism in the setting of TKI therapy targeted against different oncogenic fusion proteins, including ALK, ROS1, RET and NTRK1 fusions.
Tani, T. et al. Activation of EGFR bypass signaling by TGFα overexpression induces acquired resistance to alectinib in ALK-translocated lung cancer cells. Mol. Cancer Ther. 15, 162–171 (2016).
Taniguchi, H. et al. Amphiregulin triggered epidermal growth factor receptor activation confers in vivo crizotinib-resistance of EML4-ALK lung cancer and circumvention by epidermal growth factor receptor inhibitors. Cancer Sci. 108, 53–60 (2017).
Yamaguchi, N. et al. Dual ALK and EGFR inhibition targets a mechanism of acquired resistance to the tyrosine kinase inhibitor crizotinib in ALK rearranged lung cancer. Lung Cancer 83, 37–43 (2014).
Arteaga, C. L. & Engelman, J. A. ERBB receptors: from oncogene discovery to basic science to mechanism-based cancer therapeutics. Cancer Cell 25, 282–303 (2014).
Tabara, K. et al. Loss of activating EGFR mutant gene contributes to acquired resistance to EGFR tyrosine kinase inhibitors in lung cancer cells. PLoS ONE 7, e41017 (2012).
Heinmoller, P. et al. HER2 status in non-small cell lung cancer: results from patient screening for enrollment to a phase II study of herceptin. Clin. Cancer Res. 9, 5238–5243 (2003).
Takezawa, K. et al. HER2 amplification: a potential mechanism of acquired resistance to EGFR inhibition in EGFR-mutant lung cancers that lack the second-site EGFRT790M mutation. Cancer Discov. 2, 922–933 (2012).
Tanizaki, J. et al. Activation of HER family signaling as a mechanism of acquired resistance to ALK inhibitors in EML4-ALK-positive non-small cell lung cancer. Clin. Cancer Res. 18, 6219–6226 (2012).
Gu, F. F. et al. Lung adenocarcinoma harboring concomitant SPTBN1-ALK fusion, c-Met overexpression, and HER-2 amplification with inherent resistance to crizotinib, chemotherapy, and radiotherapy. J. Hematol. Oncol. 9, 66 (2016).
Clamon, G. et al. Lack of trastuzumab activity in nonsmall cell lung carcinoma with overexpression of erb-B2: 39810: a phase II trial of Cancer and Leukemia Group B. Cancer 103, 1670–1675 (2005).
Lara, P. N. Jr et al. Trastuzumab plus docetaxel in HER2/neu-positive non-small-cell lung cancer: a California Cancer Consortium screening and phase II trial. Clin. Lung Cancer 5, 231–236 (2004).
Langer, C. J., Stephenson, P., Thor, A., Vangel, M. & Johnson, D. H. Trastuzumab in the treatment of advanced non-small-cell lung cancer: is there a role? Focus on Eastern Cooperative Oncology Group study 2598. J. Clin. Oncol. 22, 1180–1187 (2004).
Gatzemeier, U. et al. Randomized phase II trial of gemcitabine-cisplatin with or without trastuzumab in HER2-positive non-small-cell lung cancer. Ann. Oncol. 15, 19–27 (2004).
Terai, H. et al. Activation of the FGF2-FGFR1 autocrine pathway: a novel mechanism of acquired resistance to gefitinib in NSCLC. Mol. Cancer Res. 11, 759–767 (2013).
Amato, K. R. et al. EPHA2 blockade overcomes acquired resistance to EGFR kinase inhibitors in lung cancer. Cancer Res. 76, 305–318 (2016).
Lovly, C. M. et al. Rationale for co-targeting IGF-1R and ALK in ALK fusion-positive lung cancer. Nat. Med. 20, 1027–1034 (2014).
Morgillo, F., Woo, J. K., Kim, E. S., Hong, W. K. & Lee, H. Y. Heterodimerization of insulin-like growth factor receptor/epidermal growth factor receptor and induction of survivin expression counteract the antitumor action of erlotinib. Cancer Res. 66, 10100–10111 (2006).
Dziadziuszko, R. et al. An activating KIT mutation induces crizotinib resistance in ROS1-positive lung cancer. J. Thorac. Oncol. 11, 1273–1281 (2016).
Gong, Y. et al. Induction of BIM is essential for apoptosis triggered by EGFR kinase inhibitors in mutant EGFR-dependent lung adenocarcinomas. PLoS Med. 4, e294 (2007).
Zhang, L. et al. Clinical features of Bim deletion polymorphism and its relation with crizotinib primary resistance in Chinese patients with ALK/ROS1 fusion-positive non-small cell lung cancer. Cancer 123, 2927–2935 (2017).
Tanimoto, A. et al. Histone deacetylase 3 inhibition overcomes BIM deletion polymorphism-mediated osimertinib resistance in EGFR-mutant lung cancer. Clin. Cancer Res. 23, 3139–3149 (2016).
Costa, C. et al. The impact of EGFR T790M mutations and BIM mRNA expression on outcome in patients with EGFR-mutant NSCLC treated with erlotinib or chemotherapy in the randomized phase III EURTAC trial. Clin. Cancer Res. 20, 2001–2010 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02520778 (2017).
Kane, L. P., Shapiro, V. S., Stokoe, D. & Weiss, A. Induction of NF-κB by the Akt/PKB kinase. Curr. Biol. 9, 601–604 (1999).
Moroishi, T., Hansen, C. G. & Guan, K. L. The emerging roles of YAP and TAZ in cancer. Nat. Rev. Cancer 15, 73–79 (2015).
Wang, Y. et al. Overexpression of yes-associated protein contributes to progression and poor prognosis of non-small-cell lung cancer. Cancer Sci. 101, 1279–1285 (2010).
Lee, J. E. et al. Hippo pathway effector YAP inhibition restores the sensitivity of EGFR-TKI in lung adenocarcinoma having primary or acquired EGFR-TKI resistance. Biochem. Biophys. Res. Commun. 474, 154–160 (2016).
Hsu, P. C. et al. YAP promotes erlotinib resistance in human non-small cell lung cancer cells. Oncotarget 7, 51922–51933 (2016).
Zhang, J. et al. YAP-dependent induction of amphiregulin identifies a non-cell-autonomous component of the Hippo pathway. Nat. Cell Biol. 11, 1444 (2009).
Jiang, J. et al. Coexistence of p16/CDKN2A homozygous deletions and activating EGFR mutations in lung adenocarcinoma patients signifies a poor response to EGFR-TKIs. Lung Cancer 102, 101–107 (2016).
Liu, M. et al. PD 0332991, a selective cyclin D kinase 4/6 inhibitor, sensitizes lung cancer cells to treatment with epidermal growth factor receptor tyrosine kinase inhibitors. Oncotarget 7, 84951–84964 (2016).
Cha, Y. J., Cho, B. C., Kim, H. R., Lee, H. J. & Shim, H. S. A. Case of ALK-rearranged adenocarcinoma with small cell carcinoma-like transformation and resistance to crizotinib. J. Thorac. Oncol. 11, e55–e58 (2016).
Niederst, M. J. et al. RB loss in resistant EGFR mutant lung adenocarcinomas that transform to small-cell lung cancer. Nat. Commun. 6, 6377 (2015).
Kobayashi, Y. et al. Transformation to sarcomatoid carcinoma in ALK-rearranged adenocarcinoma, which developed acquired resistance to crizotinib and received subsequent chemotherapies. J. Thorac. Oncol. 8, e75–e78 (2013).
Gower, A., Hsu, W. H., Hsu, S. T., Wang, Y. & Giaccone, G. EMT is associated with, but does not drive resistance to ALK inhibitors among EML4-ALK non-small cell lung cancer. Mol. Oncol. 10, 601–609 (2016).
Kim, H. R. et al. Epithelial-mesenchymal transition leads to crizotinib resistance in H2228 lung cancer cells with EML4-ALK translocation. Mol. Oncol. 7, 1093–1102 (2013).
Witta, S. E. et al. Restoring E-cadherin expression increases sensitivity to epidermal growth factor receptor inhibitors in lung cancer cell lines. Cancer Res. 66, 944–950 (2006).
Yao, Z. et al. TGF-β IL-6 axis mediates selective and adaptive mechanisms of resistance to molecular targeted therapy in lung cancer. Proc. Natl Acad. Sci. USA 107, 15535–15540 (2010).
Zhang, T. et al. A genetic cell context-dependent role for ZEB1 in lung cancer. Nat. Commun. 7, 12231 (2016).
Yoshida, T. et al. ZEB1 mediates acquired resistance to the epidermal growth factor receptor-tyrosine kinase inhibitors in non-small cell lung cancer. PLoS ONE 11, e0147344 (2016).
Wu, F., Li, J., Jang, C., Wang, J. & Xiong, J. The role of Axl in drug resistance and epithelial-to-mesenchymal transition of non-small cell lung carcinoma. Int. J. Clin. Exp. Pathol. 7, 6653–6661 (2014).
Wilson, C. et al. Overcoming EMT-associated resistance to anti-cancer drugs via Src/FAK pathway inhibition. Oncotarget 5, 7328–7341 (2014).
Li, L. et al. Metformin sensitizes EGFR-TKI-resistant human lung cancer cells in vitro and in vivo through inhibition of IL-6 signaling and EMT reversal. Clin. Cancer Res. 20, 2714–2726 (2014).
Sharma, S. V. et al. A chromatin-mediated reversible drug-tolerant state in cancer cell subpopulations. Cell 141, 69–80 (2010).
Greve, G. et al. The pan-HDAC inhibitor panobinostat acts as a sensitizer for erlotinib activity in EGFR-mutated and -wildtype non-small cell lung cancer cells. BMC Cancer 15, 947 (2015).
Choe, C. et al. Crosstalk with cancer-associated fibroblasts induces resistance of non-small cell lung cancer cells to epidermal growth factor receptor tyrosine kinase inhibition. Onco Targets Ther. 8, 3665–3678 (2015).
Yoshida, T. et al. Podoplanin-positive cancer-associated fibroblasts in the tumor microenvironment induce primary resistance to EGFR-TKIs in lung adenocarcinoma with EGFR mutation. Clin. Cancer Res. 21, 642–651 (2015).
Mink, S. R. et al. Cancer-associated fibroblasts derived from EGFR-TKI-resistant tumors reverse EGFR pathway inhibition by EGFR-TKIs. Mol. Cancer Res. 8, 809–820 (2010).
Wang, W. et al. Crosstalk to stromal fibroblasts induces resistance of lung cancer to epidermal growth factor receptor tyrosine kinase inhibitors. Clin. Cancer Res. 15, 6630–6638 (2009).
Lotti, F. et al. Chemotherapy activates cancer-associated fibroblasts to maintain colorectal cancer-initiating cells by IL-17A. J. Exp. Med. 210, 2851–2872 (2013).
Chang, H. et al. EGF induced RET inhibitor resistance in CCDC6-RET lung cancer cells. Yonsei Med. J. 58, 9–18 (2017).
An, J. et al. Significance of cancer-associated fibroblasts in the regulation of gene expression in the leading cells of invasive lung cancer. J. Cancer Res. Clin. Oncol. 139, 379–388 (2013).
Bae, S. Y., Park, H. J., Hong, J. Y., Lee, H. J. & Lee, S. K. Down-regulation of SerpinB2 is associated with gefitinib resistance in non-small cell lung cancer and enhances invadopodia-like structure protrusions. Scientif. Rep. 6, 32258 (2016).
Yamauchi, M. et al. N-Cadherin expression is a potential survival mechanism of gefitinib-resistant lung cancer cells. Am. J. Cancer Res. 1, 823–833 (2011).
Wald, O. et al. Interaction between neoplastic cells and cancer-associated fibroblasts through the CXCL12/CXCR4 axis: role in non-small cell lung cancer tumor proliferation. J. Thorac. Cardiovasc. Surg. 141, 1503–1512 (2011).
Wang, Y. et al. Abstract 4119: Cytoplasmic location of CXCR4 is correlated to loss of EMT marker and activation of downstream signaling pathway in non-small cell lung cancer. Cancer Res. 70, abstr. 4119 (2014).
Muz, B., de la Puente, P., Azab, F. & Azab, A. K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 3, 83–92 (2015).
Murakami, A. et al. Hypoxia increases gefitinib-resistant lung cancer stem cells through the activation of insulin-like growth factor 1 receptor. PLoS ONE 9, e86459 (2014).
Minakata, K. et al. Hypoxia induces gefitinib resistance in non-small-cell lung cancer with both mutant and wild-type epidermal growth factor receptors. Cancer Sci. 103, 1946–1954 (2012).
Kogita, A. et al. Hypoxia induces resistance to ALK inhibitors in the H3122 non-small cell lung cancer cell line with an ALK rearrangement via epithelial-mesenchymal transition. Int. J. Oncol. 45, 1430–1436 (2014).
Chatterjee, S. et al. Tumor VEGF:VEGFR2 autocrine feed-forward loop triggers angiogenesis in lung cancer. J. Clin. Invest. 123, 1732–1740 (2013).
Chung, F. T. et al. Tumor-associated macrophages correlate with response to epidermal growth factor receptor-tyrosine kinase inhibitors in advanced non-small cell lung cancer. Int. J. Cancer 131, E227–E235 (2012).
Choi, H. et al. Transcriptome analysis of individual stromal cell populations identifies stroma-tumor crosstalk in mouse lung cancer model. Cell Rep. 10, 1187–1201 (2015). This paper described a computational model of signalling interactions between tumour cells and stromal cells, which was generated based on transcriptome analysis of sorted tumour and myeloid cells.
Akbay, E. A. et al. Activation of the PD-1 pathway contributes to immune escape in EGFR-driven lung tumors. Cancer Discov. 3, 1355–1363 (2013).
Ota, K. et al. Induction of PD-L1 expression by the EML4-ALK oncoprotein and downstream signaling pathways in non-small cell lung cancer. Clin. Cancer Res. 21, 4014–4021 (2015).
Gainor, J. F. et al. EGFR mutations and ALK rearrangements are associated with low response rates to PD-1 pathway blockade in non-small cell lung cancer: a retrospective analysis. Clin. Cancer Res. 22, 4585–4593 (2016).
Jamal-Hanjani, M. et al. Tracking the evolution of non-small-cell lung cancer. N. Engl. J. Med. 376, 2109–2121 (2017). This paper described the spectrum of intratumoural heterogeneity within resected NSCLC tissue and correlated these findings with the risk of recurrence.
Suda, K. et al. Heterogeneity in resistance mechanisms causes shorter duration of epidermal growth factor receptor kinase inhibitor treatment in lung cancer. Lung Cancer 91, 36–40 (2016).
Sundaresan, T. K. et al. Detection of T790M, the acquired resistance EGFR mutation, by tumor biopsy versus noninvasive blood-based analyses. Clin. Cancer Res. 22, 1103–1110 (2016).
Sorensen, B. S. et al. Monitoring of epidermal growth factor receptor tyrosine kinase inhibitor-sensitizing and resistance mutations in the plasma DNA of patients with advanced non-small cell lung cancer during treatment with erlotinib. Cancer 120, 3896–3901 (2014).
Oxnard, G. R. et al. Noninvasive detection of response and resistance in EGFR-mutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin. Cancer Res. 20, 1698–1705 (2014).
Abbosh, C. et al. Phylogenetic ctDNA analysis depicts early stage lung cancer evolution. Nature 545, 446–451 (2017). This study used ctDNA analysis to identify patients progressing towards resistance to systemic therapies and towards tumour recurrence, as well as to monitor for subclonal molecular changes within metastatic disease.
Ramirez, M. et al. Diverse drug-resistance mechanisms can emerge from drug-tolerant cancer persister cells. Nat. Commun. 7, 10690 (2016).
Gomez, D. R. et al. Local consolidative therapy versus maintenance therapy or observation for patients with oligometastatic non-small-cell lung cancer without progression after first-line systemic therapy: a multicentre, randomised, controlled, phase 2 study. Lancet Oncol. 17, 1672–1682 (2016).
Mok, T. et al. Dacomitinib versus gefitinib for the first-line treatment of advanced EGFR mutation positive non-small cell lung cancer (ARCHER 1050): a randomized, open-label phase III trial. J. Clin. Oncol. http://dx.doi.org/10.1200/jco.2017.35.18_suppl.lba9007 (2017).
Ramalingam, S. et al. LBA1_PR: Osimertinb as first-line treatment for EGFR mutation-positive advanced NSCLC: updated efficacy and safety results from two Phase I expansion cohorts. J. Thorac. Oncol. 11, S152 (2016).
Jonsson, V. D. et al. Novel computational method for predicting polytherapy switching strategies to overcome tumor heterogeneity and evolution. Sci. Rep. 7, 44206 (2017). Computational modelling was used in this study to predict optimal schedules for the sequencing of targeted therapy, including the prediction that switching strategies directed by mutational changes might provide for the greatest therapeutic efficacy.
Weickhardt, A. J. et al. Local ablative therapy of oligoprogressive disease prolongs disease control by tyrosine kinase inhibitors in oncogene-addicted non-small-cell lung cancer. J. Thorac. Oncol. 7, 1807–1814 (2012).
Qiu, B. et al. Local therapy for oligoprogressive disease in patients with advanced stage non-small-cell lung cancer harboring epidermal growth factor receptor mutation. Clin. Lung Cancer http://dx.doi.org/10.1016/j.cllc.2017.04.002 (2017).
Liu, S. & Lee, J. J. An overview of the design and conduct of the BATTLE trials. Chin. Clin. Oncol. 4, 33 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02465060 (2017).
Rangachari, D. et al. Brain metastases in patients with EGFR-mutated or ALK-rearranged non-small-cell lung cancers. Lung Cancer 88, 108–111 (2015).
Deeken, J. F. & Loscher, W. The blood-brain barrier and cancer: transporters, treatment, and Trojan horses. Clin. Cancer Res. 13, 1663–1674 (2007).
Togashi, Y. et al. Cerebrospinal fluid concentration of erlotinib and its active metabolite OSI-420 in patients with central nervous system metastases of non-small cell lung cancer. J. Thorac. Oncol. 5, 950–955 (2010).
Togashi, Y. et al. Cerebrospinal fluid concentration of gefitinib and erlotinib in patients with non-small cell lung cancer. Cancer Chemother. Pharmacol. 70, 399–405 (2012).
Zhao, J. et al. Cerebrospinal fluid concentrations of gefitinib in patients with lung adenocarcinoma. Clin. Lung Cancer 14, 188–193 (2013).
Tamiya, A. et al. OA08.05 Efficacy and cerebrospinal fluid concentration of afatinib in NSCLC patients with EGFR mutation developing leptomeningeal carcinomatosis. J. Thorac. Oncol. Abstr. 12, S273 (2017).
Hoffknecht, P. et al. Efficacy of the irreversible ErbB family blocker afatinib in epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI)-pretreated non-small-cell lung cancer patients with brain metastases or leptomeningeal disease. J. Thorac. Oncol. 10, 156–163 (2015).
Ballard, P. et al. Preclinical comparison of osimertinib with other EGFR-TKIs in EGFR-mutant NSCLC brain metastases models, and early evidence of clinical brain metastases activity. Clin. Cancer Res. 22, 5130–5140 (2016).
Costa, D. B. et al. CSF concentration of the anaplastic lymphoma kinase inhibitor crizotinib. J. Clin. Oncol. 29, e443–e445 (2011).
Novartis. Zykadia prescribing information Novartis https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/zykadia.pdf (2016).
Kodama, T. et al. Antitumor activity of the selective ALK inhibitor alectinib in models of intracranial metastases. Cancer Chemother. Pharmacol. 74, 1023–1028 (2014).
Dudnik, E. et al. Durable brain response with pulse-dose crizotinib and ceritinib in ALK-positive non-small cell lung cancer compared with brain radiotherapy. J. Clin. Neurosci. 26, 46–49 (2016).
Jackman, D. et al. Pulsed dosing of erlotinib for central nervous system progression in EGFR-mutant non-small cell lung cancer. J. Clin. Oncol. 31, 8116 (2013).
Rho, J. K. et al. Superior efficacy and selectivity of novel small-molecule kinase inhibitors of T790M-mutant EGFR in preclinical models of lung cancer. Cancer Res. 77, 1200–1211 (2017).
Ariyasu, R. et al. Efficacy of bevacizumab and erlotinib combination for leptomeningeal carcinomatosis after failure of erlotinib. Anticancer Drugs 28, 565–567 (2017).
Tang, S. C. et al. Increased oral availability and brain accumulation of the ALK inhibitor crizotinib by coadministration of the P-glycoprotein (ABCB1) and breast cancer resistance protein (ABCG2) inhibitor elacridar. Int. J. Cancer 134, 1484–1494 (2014).
Das, M. et al. Specific radiolabeling of a cell surface receptor for epidermal growth factor. Proc. Natl Acad. Sci. USA 74, 2790–2794 (1977).
Nakano, H. et al. Isolation of transforming sequences of two human lung carcinomas: structural and functional analysis of the activated c-K-ras oncogenes. Proc. Natl Acad. Sci. USA 81, 71–75 (1984).
Takahashi, M., Ritz, J. & Cooper, G. M. Activation of a novel human transforming gene, ret, by DNA rearrangement. Cell 42, 581–588 (1985).
Ward, W. H. et al. Epidermal growth factor receptor tyrosine kinase. Investigation of catalytic mechanism, structure-based searching and discovery of a potent inhibitor. Biochem. Pharmacol. 48, 659–666 (1994).
Morris, S. W. et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin's lymphoma. Science 263, 1281–1284 (1994).
Davies, H. et al. Mutations of the BRAF gene in human cancer. Nature 417, 949–954 (2002).
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–2139 (2004).
Stephens, P. et al. Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature 431, 525–526 (2004).
Ma, P. C. et al. Functional expression and mutations of c-Met and its therapeutic inhibition with SU11274and small interfering RNA in non-small cell lung cancer. Cancer Res. 65, 1479–1488 (2005).
Cappuzzo, F., Bemis, L. & Varella-Garcia, M. HER2 mutation and response to trastuzumab therapy in non-small-cell lung cancer. N. Engl. J. Med. 354, 2619–2621 (2006).
Rikova, K. et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 131, 1190–1203 (2007).
McDermott, U. et al. Genomic alterations of anaplastic lymphoma kinase may sensitize tumors to anaplastic lymphoma kinase inhibitors. Cancer Res. 68, 3389–3395 (2008).
Brahmer, J. R. et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J. Clin. Oncol. 28, 3167–3175 (2010).
Kohno, T. et al. KIF5B-RET fusions in lung adenocarcinoma. Nat. Med. 18, 375–377 (2012).
Lipson, D. et al. Identification of new ALK and RET gene fusions from colorectal and lung cancer biopsies. Nat. Med. 18, 382–384 (2012).
Takeuchi, K. et al. RET, ROS1 and ALK fusions in lung cancer. Nat. Med. 18, 378–381 (2012).
Gautschi, O. et al. A patient with BRAF V600E lung adenocarcinoma responding to vemurafenib. J. Thorac. Oncol. 7, e23–e24 (2012).
De Greve, J. et al. Clinical activity of afatinib (BIBW 2992) in patients with lung adenocarcinoma with mutations in the kinase domain of HER2/neu. Lung Cancer 76, 123–127 (2012).
Walter, A. O. et al. Discovery of a mutant-selective covalent inhibitor of EGFR that overcomes T790M-mediated resistance in NSCLC. Cancer Discov. 3, 1404–1415 (2013).
Drilon, A. et al. Response to Cabozantinib in patients with RET fusion-positive lung adenocarcinomas. Cancer Discov. 3, 630–635 (2013).
Vaishnavi, A. et al. Oncogenic and drug-sensitive NTRK1 rearrangements in lung cancer. Nat. Med. 19, 1469–1472 (2013).
Farago, A. F. et al. Durable clinical response to entrectinib in NTRK1-rearranged non-small cell lung cancer. J. Thorac. Oncol. 10, 1670–1674 (2015).
Zandi, R., Larsen, A. B., Andersen, P., Stockhausen, M. T. & Poulsen, H. S. Mechanisms for oncogenic activation of the epidermal growth factor receptor. Cell. Signal. 19, 2013–2023 (2007).
Kobayashi, Y. & Mitsudomi, T. Not all epidermal growth factor receptor mutations in lung cancer are created equal: perspectives for individualized treatment strategy. Cancer Sci. 107, 1179–1186 (2016).
Shigematsu, H. et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. J. Natl Cancer Inst. 97, 339–346 (2005).
Kim, E. K., Kim, K. A., Lee, C. Y. & Shim, H. S. The frequency and clinical impact of HER2 alterations in lung adenocarcinoma. PLoS ONE 12, e0171280 (2017).
Ju, Y. S. et al. A transforming KIF5B and RET gene fusion in lung adenocarcinoma revealed from whole-genome and transcriptome sequencing. Genome Res. 22, 436–445 (2012).
Gautschi, O. et al. Targeting RET in patients with RET-rearranged lung cancers: results from the global, multicenter RET registry. J. Clin. Oncol. 35, 1403–1410 (2017).
Velcheti, V. et al. FRMD4A/RET: a novel RET oncogenic fusion variant in non-small cell lung carcinoma. J. Thorac. Oncol. 12, e15–e16 (2017).
Matsubara, D. et al. Identification of CCDC6-RET fusion in the human lung adenocarcinoma cell line. LC-2/ad. J. Thorac. Oncol. 7, 1872–1876 (2012).
Lee, M. S. et al. Identification of a novel partner gene, KIAA1217, fused to RET: functional characterization and inhibitor sensitivity of two isoforms in lung adenocarcinoma. Oncotarget 7, 36101–36114 (2016).
Lira, M. E. et al. A single-tube multiplexed assay for detecting ALK, ROS1, and RET fusions in lung cancer. J. Mol. Diagn. 16, 229–243 (2014).
Kodama, T. et al. Alectinib shows potent antitumor activity against RET-rearranged non-small cell lung cancer. Mol. Cancer Ther. 13, 2910–2918 (2014).
Stransky, N., Cerami, E., Schalm, S., Kim, J. L. & Lengauer, C. The landscape of kinase fusions in cancer. Nat. Commun. 5, 4846 (2014).
Engelman, J. A. et al. Effective use of PI3K and MEK inhibitors to treat mutant Kras G12D and PIK3CA H1047R murine lung cancers. Nat. Med. 14, 1351–1356 (2008).
Redig, A. J. et al. Clinical and molecular characteristics of NF1-mutant lung cancer. Clin. Cancer Res. 22, 3148–3156 (2016).
Wildemann, B. C. et al. Phase I study of the MEK 1/2 inhibitor selumetinib (AZD6244) hydrogen sulfate in children and young adults with neurofibromatosis type 1 (NF1) and inoperable plexiform neurofibromas. J. Clin. Oncol. 32, 10018 (2014).
Kosaka, T. et al. Analysis of epidermal growth factor receptor gene mutation in patients with non-small cell lung cancer and acquired resistance to gefitinib. Clin. Cancer Res. 12, 5764–5769 (2006).
Tiedt, R. et al. A drug resistance screen using a selective MET inhibitor reveals a spectrum of mutations that partially overlap with activating mutations found in cancer patients. Cancer Res. 71, 5255–5264 (2011).
Oxnard, G. R. et al. Preliminary results of TATTON, a multi-arm phase Ib trial of AZD9291 combined with MEDI4736, AZD6094 or selumetinib in EGFR-mutant lung cancer. J. Clin. Oncol. 33, abstr. 2509 (2015).
Janne, P. A. et al. Phase I safety and pharmacokinetic study of the PI3K/mTOR inhibitor SAR245409 (XL765) in combination with erlotinib in patients with advanced solid tumors. J. Thorac. Oncol. 9, 316–323 (2014).
Tan, D. S. et al. A phase Ib safety and tolerability study of a pan class I PI3K inhibitor buparlisib (BKM120) and gefitinib in EGFR TKI-resistance NSCLC. J. Clin. Oncol. 31, 8107 (2013).
Price, K. A. et al. Phase II trial of gefitinib and everolimus in advanced non-small cell lung cancer. J. Thorac. Oncol. 5, 1623–1629 (2010).
Park, J. S., Kim, H. R., Hong, M. H. & Cho, B. C. P2.06-003 a phase Ib study of the combination of afatinib and ruxolitinib in EGFR mutant non-small cell lung cancer (NSCLC) progressed on EGFR-TKI. J. Thorac. Oncol. 12, S1069–S1070 (2017).
Haura, E. B. et al. Phase I/II study of the Src inhibitor dasatinib in combination with erlotinib in advanced non-small-cell lung cancer. J. Clin. Oncol. 28, 1387–1394 (2010).
Wakelee, H. A. et al. A phase Ib/II study of cabozantinib (XL184) with or without erlotinib in patients with non-small cell lung cancer. Cancer Chemother. Pharmacol. 79, 923–932 (2017).
Neal, J. W. et al. Erlotinib, cabozantinib, or erlotinib plus cabozantinib as second-line or third-line treatment of patients with EGFR wild-type advanced non-small-cell lung cancer (ECOG-ACRIN 1512): a randomised, controlled, open-label, multicentre, phase 2 trial. Lancet Oncol. 17, 1661–1671 (2016).
Spigel, D. R. et al. Randomized phase II trial of Onartuzumab in combination with erlotinib in patients with advanced non-small-cell lung cancer. J. Clin. Oncol. 31, 4105–4114 (2013).
Von Pawel, J. et al. Phase 2 HERALD study of patritumab (P) with erlotinib (E) in advanced NSCLC subjects (SBJs). J. Clin. Oncol. Abstr. 32, 8045 (2014).
Rosell, R. et al. Erlotinib and bevacizumab in patients with advanced non-small-cell lung cancer and activating EGFR mutations (BELIEF): an international, multicentre, single-arm, phase 2 trial. Lancet Respir. Med. 5, 435–444 (2017).
Reguart, N. et al. Phase I/II trial of vorinostat (SAHA) and erlotinib for non-small cell lung cancer (NSCLC) patients with epidermal growth factor receptor (EGFR) mutations after erlotinib progression. Lung Cancer 84, 161–167 (2014).
Han, J. Y. et al. Phase I/II study of gefitinib (Iressa®) and vorinostat (IVORI) in previously treated patients with advanced non-small cell lung cancer. Cancer Chemother. Pharmacol. 75, 475–483 (2015).
Moran, T. et al. Activity of dalotuzumab, a selective anti-IGF1R antibody, in combination with erlotinib in unselected patients with Non-small-cell lung cancer: a phase I/II randomized trial. Exp. Hematol. Oncol. 3, 26 (2014).
Janne, P. A. et al. Combined pan-HER and ALK/ROS1/MET inhibition with dacomitinib and crizotinib in advanced non-small cell lung cancer: results of a phase I study. J. Thorac. Oncol. 11, 737–747 (2016).
Riely, G. et al. A phase 1 study of crizotinib and ganetespib (STA-9090) in ALK positive lung cancers. J. Clin. Oncol. 33, 8064 (2015).
Lee, J. et al. Addition of HSP90 inhibitor onalespib to crizotinib prior to progression in patients with ALK-pos NSCLC: results of a randomized phase 2 study [abstract]. J. Clin. Oncol. 34, 9059 (2016).
Author information
Authors and Affiliations
Contributions
J.R. researched data for the article. Both authors contributed equally to the discussion of the content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
T.G.B. declares that he is a co-founder of Driver; a consultant for Novartis, AstraZeneca, Ariad and Teva; a member of the scientific advisory board for Array Biopharma and Revolution Medicines; and supported by Ignyta and Revolution Medicines for research funding. J.R. declares a relationship with Revolution Medicines as a consultant.
Glossary
- Intrinsic resistance
-
Tumour cell resistance to therapy due to baseline characteristics present before therapy exposure.
- Adaptive resistance
-
Dynamic changes in tumour cell signalling occurring during treatment with targeted therapy that promote therapy resistance.
- Acquired resistance
-
New molecular alterations leading to the development of targeted therapy resistance after an initial period of drug sensitivity.
- Tyrosine kinase inhibitors
-
A class of small-molecule inhibitors that antagonize receptor tyrosine kinase signalling.
- Non-covalent inhibitors
-
Inhibitors that bind to a target protein in a non-covalent, reversible manner.
- Covalent inhibitor
-
An inhibitor that binds to a target protein via irreversible, covalent bonds.
- Synthetic lethality
-
Induction of tumour cell death upon simultaneous inhibition of two signalling pathways, the individual loss of which does not lead to cell death.
- Activation loop
-
A structural component of receptor tyrosine kinases that is important for the regulation of catalytic activity.
- Steric hindrance
-
Interference with protein binding due to physical interference related to protein structure.
- Type I TKI
-
ATP-competitive small-molecule TKIs that bind at the ATP binding site while in the active kinase conformation.
- Type II TKIs
-
Small-molecule TKIs that bind at and near the ATP binding site in the inactive kinase conformation.
- Sarcomatoid carcinoma
-
Pulmonary sarcomatoid carcinoma is an uncommon and aggressive poorly differentiated form of NSCLC.
- Intratumoural heterogeneity
-
Variation in tumour cell genomic and phenotypic characteristics within a given tumour.
- Convergent evolution
-
The independent development of alterations within the same signalling pathways among different tumour cell clones during the course of tumour cell evolution.
- Radiographic progression
-
Tumour enlargement and/or new lesion development that are visible on radiographic studies and meet specific criteria.
- Residual disease
-
Persistent tumour burden despite disease stabilization and/or an objective response to antineoplastic therapy.
- Oligoprogressive disease
-
Isolated growth of malignant lesions despite continued control of overall tumour disease burden.
Rights and permissions
About this article
Cite this article
Rotow, J., Bivona, T. Understanding and targeting resistance mechanisms in NSCLC. Nat Rev Cancer 17, 637–658 (2017). https://doi.org/10.1038/nrc.2017.84
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2017.84
This article is cited by
-
RGS20 promotes non-small cell lung carcinoma proliferation via autophagy activation and inhibition of the PKA-Hippo signaling pathway
Cancer Cell International (2024)
-
The role of leptin in regulation of the soluble amyloid precursor protein α (sAPPα) levels in lung cancer cell media
Scientific Reports (2024)
-
DUSP1 Promotes Osimertinib Drug-Tolerant Persistence by Inhibiting MAPK/ERK Signaling in Non-small Cell Lung Cancer
Molecular Biotechnology (2024)
-
YES1 as a potential target to overcome drug resistance in EGFR-deregulated non-small cell lung cancer
Archives of Toxicology (2024)
-
B7H3 targeting gold nanocage pH-sensitive conjugates for precise and synergistic chemo-photothermal therapy against NSCLC
Journal of Nanobiotechnology (2023)