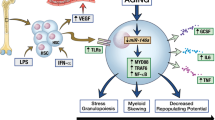
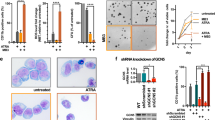
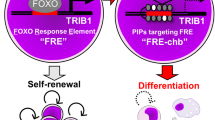
Abstract
The concept of differentiation therapy emerged from the fact that hormones or cytokines may promote differentiation ex vivo, thereby irreversibly changing the phenotype of cancer cells. Its hallmark success has been the treatment of acute promyelocytic leukaemia (APL), a condition that is now highly curable by the combination of retinoic acid (RA) and arsenic. Recently, drugs that trigger differentiation in a variety of primary tumour cells have been identified, suggesting that they are clinically useful. This Opinion article analyses the basis for the clinical successes of RA or arsenic in APL by assessing the respective roles of terminal maturation and loss of self-renewal. By reviewing other successful examples of drug-induced tumour cell differentiation, novel approaches to transform differentiating drugs into more efficient therapies are proposed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lotem, J. & Sachs, L. In vivo control of differentiation of myeloid leukemic cells by recombinant granulocyte-macrophage colony-stimulating factor and interleukin 3. Blood 71, 375–382 (1988).
Sachs, L. The control of hematopoiesis and leukemia: from basic biology to the clinic. Proc. Natl Acad. Sci. USA 93, 4742–4749 (1996).
Ferrero, D., Pessano, S., Pagliardi, G. L. & Rovera, G. Induction of differentiation of human myeloid leukemias: surface changes probed with monoclonal antibodies. Blood 61, 171–179 (1983).
Bradley, T. R., Metcalf, D. & Robinson, W. Stimulation by leukaemic sera of colony formation in solid agar cultures by proliferation of mouse bone marrow cells. Nature 213, 926–927 (1967).
Bradley, T. R., Robinson, W. & Metcalf, D. Colony production in vitro by normal polycythaemic and anaemic bone marrow. Nature 214, 511 (1967).
Nilsson, B. Probable in vivo induction of differentiation by retinoic acid of promyelocytes in acute promyelocytic leukaemia. Br. J. Haematol. 57, 365–371 (1984).
Sampi, K., Honma, Y., Hozumi, M. & Sakurai, M. Discrepancy between in-vitro and in-vivo inductions of differentiation by retinoids of human acute promyelocytic leukemia cells in relapse. Leuk. Res. 9, 1475–1478 (1985).
Wisch, J. S., Griffin, J. D. & Kufe, D. W. Response of preleukemic syndromes to continuous infusion of low-dose cytarabine. N. Engl. J. Med. 309, 1599–1602 (1983).
Housset, M., Daniel, M. T. & Degos, L. Small doses of ARA-C in the treatment of acute myeloid leukaemia: differentiation of myeloid leukaemia cells? Br. J. Haematol. 51, 125–129 (1982).
Tilly, H. et al. Low-dose cytarabine: persistence of a clonal abnormality during complete remission of acute nonlymphocytic leukemia. N. Engl. J. Med. 314, 246–247 (1986).
Shlush, L. I. et al. Identification of pre-leukaemic haematopoietic stem cells in acute leukaemia. Nature 506, 328–333 (2014).
Jan, M. et al. Clonal evolution of preleukemic hematopoietic stem cells precedes human acute myeloid leukemia. Sci. Transl Med. 4, 149ra118 (2012).
Huang, M. et al. Use of all trans retinoic acid in the treatment of acute promyelocytic leukaemia. Blood 72, 567–572 (1988).
Degos, L. et al. All-trans retinoic acid as a differentiating agent in the treatment of acute promyelocytic leukemia. Blood 85, 2643–2653 (1995).
Warrell, R., de Thé, H., Wang, Z. & Degos, L. Acute promyelocytic leukemia. N. Engl. J. Med. 329, 177–189 (1993).
Hillestad, L. K. Acute promyelocytic leukemia. Acta Med. Scand. 159, 189–194 (1957).
de The, H. & Chen, Z. Acute promyelocytic leukaemia: novel insights into the mechanisms of cure. Nat. Rev. Cancer 10, 775–783 (2010).
Dos Santos, G. A., Kats, L. & Pandolfi, P. P. Synergy against PML-RARa: targeting transcription, proteolysis, differentiation, and self-renewal in acute promyelocytic leukemia. J. Exp. Med. 210, 2793–2802 (2013).
Sanz, M. A. & Lo-Coco, F. Modern approaches to treating acute promyelocytic leukemia. J. Clin. Oncol. 29, 495–503 (2011).
de Thé, H., Pandolfi, P. & Chen, Z. Acute promyelocytic leukemia: a paradigm for oncoprotein-targeted cure. Cancer Cell (in press).
Rowley, J. D., Golomb, H. M. & Dougherty, C. 15/17 translocation, a consistent chromosomal change in acute promyelocytic leukaemia. Lancet 1, 549–550 (1977).
de The, H., Chomienne, C., Lanotte, M., Degos, L. & Dejean, A. The t(15;17) translocation of acute promyelocytic leukaemia fuses the retinoic acid receptor alpha gene to a novel transcribed locus. Nature 347, 558–561 (1990).
de Thé, H. et al. The PML-RAR alpha fusion mRNA generated by the t(15;17) translocation in acute promyelocytic leukemia encodes a functionally altered RAR. Cell 66, 675–684 (1991).
Vickers, M., Jackson, G. & Taylor, P. The incidence of acute promyelocytic leukemia appears constant over most of a human lifespan, implying only one rate limiting mutation. Leukemia 14, 722–726 (2000).
Welch, J. S. et al. The origin and evolution of mutations in acute myeloid leukemia. Cell 150, 264–278 (2012).
Karnan, S. et al. Genomewide array-based comparative genomic hybridization analysis of acute promyelocytic leukemia. Genes Chromosomes Cancer 45, 420–425 (2006).
Cancer Genome Atlas Research Network. et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N. Engl. J. Med. 368, 2059–2074 (2013).
Akagi, T. et al. Hidden abnormalities and novel classification of t(15;17) acute promyelocytic leukemia (APL) based on genomic alterations. Blood 113, 1741–1748 (2009).
Ibanez, M. et al. The mutational landscape of acute promyelocytic leukemia reveals an interacting network of co-occurrences and recurrent mutations. PLoS ONE 11, e0148346 (2016).
Ronchini, C. et al. PML-RARA-associated cooperating mutations belong to a transcriptional network that is deregulated in myeloid leukemias. Leukemia 31, 1975–1986 (2017).
Riva, L. et al. Acute promyelocytic leukemias share cooperative mutations with other myeloid-leukemia subgroups. Blood Cancer J. 4, e195 (2014).
Miller, C. A., Wilson, R. K. & Ley, T. J. Genomic landscapes and clonality of de novo AML. N. Engl. J. Med. 369, 1473 (2013).
Tocci, A. et al. Dual action of retinoic acid on human embryonic/fetal hematopoiesis: blockade of primitive progenitor proliferation and shift from multipotent/erythroid/monocytic to granulocytic differentiation program. Blood 88, 2878–2888 (1996).
Kastner, P. et al. Positive and negative regulation of granulopoiesis by endogenous RARα. Blood 97, 1314–1320 (2001).
Strickland, S. & Mahdavi, V. The induction of differentiation in teratocarcinoma stem cells by retinoic acid. Cell 15, 393–403 (1978).
Sidell, N. Retinoic acid-induced growth inhibition and morphologic differentiation of human neuroblastoma cells in vitro. J. Natl Cancer Inst. 68, 589–596 (1982).
Chanda, B., Ditadi, A., Iscove, N. N. & Keller, G. Retinoic acid signaling is essential for embryonic hematopoietic stem cell development. Cell 155, 215–227 (2013).
Niederreither, K. & Dolle, P. Retinoic acid in development: towards an integrated view. Nat. Rev. Genet. 9, 541–553 (2008).
Wendel, H. G. et al. Loss of p53 impedes the antileukemic response to BCR-ABL inhibition. Proc. Natl Acad. Sci. USA 103, 7444–7449 (2006).
El Hajj, H. et al. Retinoic acid and arsenic trioxide trigger degradation of mutated NPM1, resulting in apoptosis of AML cells. Blood 125, 3447–3454 (2015).
Ablain, J. et al. Activation of a promyelocytic leukemia-tumor protein 53 axis underlies acute promyelocytic leukemia cure. Nat. Med. 20, 167–174 (2014).
Zhao, Z. et al. p53 loss promotes acute myeloid leukemia by enabling aberrant self-renewal. Genes Dev. 24, 1389–1402 (2010).
Zuber, J. et al. Mouse models of human AML accurately predict chemotherapy response. Genes Dev. 23, 877–889 (2009).
Lallemand-Breitenbach, V. & de The, H. PML nuclear bodies. Cold Spring Harb. Perspect. Biol. 2, a000661 (2010).
Pearson, M. et al. PML regulates p53 acetylation and premature senescence induced by oncogenic Ras. Nature 406, 207–210 (2000).
Guo, A. et al. The function of PML in p53-dependent apoptosis. Nat. Cell Biol. 2, 730–736 (2000).
Ferbeyre, G. et al. PML is induced by oncogenic ras and promotes premature senescence. Genes Dev. 14, 2015–2027 (2000).
Bernardi, R. & Pandolfi, P. P. Structure, dynamics and functions of promyelocytic leukaemia nuclear bodies. Nat. Rev. Mol. Cell Biol. 8, 1006–1016 (2007).
Takahashi, Y., Lallemand-Breitenbach, V., Zhu, J. & de The, H. PML nuclear bodies and apoptosis. Oncogene 23, 2819–2824 (2004).
Nebral, K. et al. Identification of PML as novel PAX5 fusion partner in childhood acute lymphoblastic leukaemia. Br. J. Haematol. 139, 269–274 (2007).
Kurahashi, S. et al. PAX5-PML acts as a dual dominant-negative form of both PAX5 and PML. Oncogene 30, 1822–1830 (2011).
Tan, J. et al. Genomic landscapes of breast fibroepithelial tumors. Nat. Genet. 47, 1341–1345 (2015).
Khetchoumian, K. et al. Loss of Trim24 (Tif1 α) gene function confers oncogenic activity to retinoic acid receptor alpha. Nat. Genet. 39, 1500–1506 (2007).
Du, C., Redner, R. L., Cooke, M. P. & Lavau, C. Overexpression of wild-type retinoic acid receptor α (RARα) recapitulates retinoic acid-sensitive transformation of primary myeloid progenitors by acute promyelocytic leukemia RARα-fusion genes. Blood 94, 793–802 (1999).
McKeown, M. R. et al. Superenhancer analysis defines novel epigenomic subtypes of non-APL AML, including an RARα dependency targetable by SY-1425, a potent and selective RARα agonist. Cancer Discov. 7, 1136–1153 (2017).
Gurrieri, C. et al. Loss of the tumor suppressor PML in human cancers of multiple histologic origins. J. Natl Cancer Inst. 96, 269–279 (2004).
Koken, M. H. M. et al. The PML growth-suppressor has an altered expression in human oncogenesis. Oncogene 10, 1315–1324 (1995).
Martens, J. H. et al. PML-RARα/RXR alters the epigenetic landscape in acute promyelocytic leukemia. Cancer Cell 17, 173–185 (2010).
Sunami, Y. et al. Histone acetyltransferase p300/CREB-binding protein-associated factor (PCAF) is required for all-trans-retinoic acid-induced granulocytic differentiation in leukemia cells. J. Biol. Chem. 292, 2815–2829 (2017).
Daniel, M.-T. et al. PML protein expression in hematopoietic and acute promyelocytic leukemia cells. Blood 82, 1858–1867 (1993).
Insinga, A. et al. Impairment of p53 acetylation, stability and function by an oncogenic transcription factor. EMBO J. 23, 1144–1154 (2004).
Occhionorelli, M. et al. The self-association coiled-coil domain of PML is sufficient for the oncogenic conversion of the retinoic acid receptor (RAR) alpha. Leukemia 25, 814–820 (2011).
Sternsdorf, T. et al. Forced retinoic acid receptor a homodimer prime mice for APL-like leukemia. Cancer Cell 9, 81–94 (2006).
Castaigne, S. et al. All-trans retinoic acid as a differentiation therapy for acute promyelocytic leukemia. 1. Clinical results. Blood 76, 1704–1709 (1990).
Grignani, F. et al. Fusion proteins of the retinoic acid receptor-α recruit histone deacetylase in promyelocytic leukaemia. Nature 391, 815–818 (1998).
Ablain, J. & de The, H. Revisiting the differentiation paradigm in acute promyelocytic leukemia. Blood 117, 5795–5802 (2011).
Ghavamzadeh, A. et al. Phase II study of single-agent arsenic trioxide for the front-line therapy of acute promyelocytic leukemia. J. Clin. Oncol. 29, 2753–2757 (2011).
Mathews, V. et al. Arsenic trioxide in the treatment of newly diagnosed acute promyelocytic leukemia: a single center experience. Am. J. Hematol. 70, 292–299 (2002).
Mathews, V. et al. Single-agent arsenic trioxide in the treatment of newly diagnosed acute promyelocytic leukemia: long-term follow-up data. J. Clin. Oncol. 28, 3866–3871 (2010).
Zhu, J., Chen, Z., Lallemand-Breitenbach, V. & de Thé, H. How acute promyelocytic leukemia revived arsenic. Nat. Rev. Cancer 2, 705–713 (2002).
Camacho, L. H. et al. Leukocytosis and the retinoic acid syndrome in patients with acute promyelocytic leukemia treated with arsenic trioxide. J. Clin. Oncol. 18, 2620–2625 (2000).
Guillemin, M. C. et al. In vivo activation of cAMP signaling induces growth arrest and differentiation in acute promyelocytic leukemia. J. Exp. Med. 196, 1373–1380 (2002).
Muto, A. et al. A novel differentiation-inducing therapy for acute promyelocytic leukemia with a combination of arsenic trioxide and GM-CSF. Leukemia 15, 1176–1184 (2001).
Ablain, J. et al. Uncoupling RARA transcriptional activation and degradation clarifies the bases for APL response to therapies. J. Exp. Med. 210, 647–653 (2013).
Nasr, R. et al. Eradication of acute promyelocytic leukemia-initiating cells through PML-RARA degradation. Nat. Med. 14, 1333–1342 (2008).
Quignon, F., Chen, Z. & de Thé, H. Retinoic acid and arsenic: towards oncogene targeted treatments of acute promyelocytic leukaemia. Biochim. Biophys. Acta 1333, M53–M61 (1997).
Zhu, J., Lallemand-Breitenbach, V. & de The, H. Pathways of retinoic acid- or arsenic trioxide-induced PML/RARα catabolism, role of oncogene degradation in disease remission. Oncogene 20, 7257–7265 (2001).
Jeanne, M. et al. PML/RARA oxidation and arsenic binding initiate the antileukemia response of As2O3. Cancer Cell 18, 88–98 (2010).
Zhu, J. et al. Arsenic-induced PML targeting onto nuclear bodies: implications for the treatment of acute promyelocytic leukemia. Proc. Natl Acad. Sci. USA 94, 3978–3983 (1997).
Isakson, P., Bjoras, M., Boe, S. O. & Simonsen, A. Autophagy contributes to therapy-induced degradation of the PML/RARA oncoprotein. Blood 116, 2324–2331 (2010).
Chen, G. Q. et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): I. As2O3 exerts dose-dependent dual effects on APL cells. Blood 89, 3345–3353 (1997).
Vitaliano-Prunier, A. et al. Clearance of PML/RARA-bound promoters suffice to initiate APL differentiation. Blood 124, 3772–3780 (2014).
Zhu, H. H., Qin, Y. Z. & Huang, X. J. Resistance to arsenic therapy in acute promyelocytic leukemia. N. Engl. J. Med. 370, 1864–1866 (2014).
Goto, E. et al. Missense mutations in PML-RARA are critical for the lack of responsiveness to arsenic trioxide treatment. Blood 118, 1600–1609 (2011).
Gallagher, R. E. et al. Treatment-influenced associations of PML-RARα mutations, FLT3 mutations, and additional chromosome abnormalities in relapsed acute promyelocytic leukemia. Blood 120, 2098–2108 (2012).
Lehmann-Che, J. Bally, C. & de Thé, H. Therapy resistance in APL. N. Engl. J. Med. 371, 1171–1172 (2014).
Iaccarino, L. et al. Mutations affecting both the rearranged and the unrearranged PML alleles in refractory acute promyelocytic leukaemia. Br. J. Haematol. 172, 909–913 (2016).
Shao, W. et al. Arsenic trioxide as an inducer of apoptosis and loss of PML/RARα protein in acute promyelocytic leukemia cells. J. Natl Cancer Inst. 90, 124–133 (1998).
Cicconi, L. et al. PML-RARα kinetics and impact of FLT3-ITD mutations in newly diagnosed acute promyelocytic leukaemia treated with ATRA and ATO or ATRA and chemotherapy. Leukemia 30, 1987–1992 (2016).
Burnett, A. K. et al. Arsenic trioxide and all-trans retinoic acid treatment for acute promyelocytic leukaemia in all risk groups (AML17): results of a randomised, controlled, phase 3 trial. Lancet Oncol. 16, 1295–1305 (2015).
Lo-Coco, F. et al. Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. N. Engl. J. Med. 369, 111–121 (2013).
Lo-Coco, F. et al. Targeted therapy alone for acute promyelocytic leukemia. N. Engl. J. Med. 374, 1197–1198 (2016).
Mann, K. K. & Miller, W. H. Jr. Death by arsenic: implications of PML sumoylation. Cancer Cell 5, 307–309 (2004).
Liu, W. et al. Induction of tumor arrest and differentiation with prolonged survival by intermittent hypoxia in a mouse model of acute myeloid leukemia. Blood 107, 698–707 (2006).
Olsson, I. L., Breitman, T. R. & Gallo, R. C. Priming of human myeloid leukemic cell lines HL-60 and U-937 with retinoic acid for differentiation effects of cyclic adenosine 3′:5′-monophosphate-inducing agents and a T-lymphocyte-derived differentiation factor. Cancer Res. 42, 3928–3933 (1982).
Lando, M., Abemayor, E., Verity, M. A. & Sidell, N. Modulation of intracellular cyclic adenosine monophosphate levels and the differentiation response of human neuroblastoma cells. Cancer Res. 50, 722–727 (1990).
Benoit, G. et al. RAR-independent RXR signaling induces t(15;17) leukemia cell maturation. EMBO J. 18, 7011–7018 (1999).
Quenech'Du, N., Ruchaud, S., Khelef, N., Guiso, N. & Lanotte, M. A sustained increase in the endogenous level of cAMP reduces the retinoid concentration required for APL cell maturation to near physiological levels. Leukemia 12, 1829–1833 (1998).
Kamashev, D. E., Vitoux, D. & de Thé, H. PML/RARA-RXR oligomers mediate retinoid- and rexinoid- /cAMP in APL cell differentiation. J. Exp. Med. 199, 1–13 (2004).
Altucci, L. et al. Rexinoid-triggered differentiation and tumours selective apoptosis of AML by protein kinase-A-mediated de-subordination of RXR. Cancer Res. 65, 8754–8765 (2005).
Gaillard, E. et al. Phosphorylation by PKA potentiates retinoic acid receptor alpha activity by means of increasing interaction with and phosphorylation by cyclin H/cdk7. Proc. Natl Acad. Sci. USA 103, 9548–9553 (2006).
Zhu, Q. et al. Synergic effects of arsenic trioxide and cAMP during acute promyelocytic leukemia cell maturation subtends a novel signaling cross- talk. Blood 99, 1014–1022 (2002).
Zhu, J. et al. Lineage restriction of the RARα gene expression in myeloid differentiation. Blood 98, 2563–2567 (2001).
Glasow, A., Prodromou, N., Xu, K., von Lindern, M. & Zelent, A. Retinoids and myelomonocytic growth factors cooperatively activate RARA and induce human myeloid leukemia cell differentiation via MAP kinase pathways. Blood 105, 341–349 (2005).
Schlenk, R. F. et al. Phase III study of all-trans retinoic acid in previously untreated patients 61 years or older with acute myeloid leukemia. Leukemia 18, 1798–1803 (2004).
Schlenk, R. F. et al. Gene mutations and response to treatment with all-trans retinoic acid in elderly patients with acute myeloid leukemia. Results from the AMLSG Trial AML HD98B. Haematologica 94, 54–60 (2009).
Martelli, M. P. et al. Arsenic trioxide and all-trans retinoic acid target NPM1 mutant oncoprotein levels and induce apoptosis in NPM1-mutated AML cells. Blood 125, 3455–3465 (2015).
Schenk, T. et al. Inhibition of the LSD1 (KDM1A) demethylase reactivates the all-trans-retinoic acid differentiation pathway in acute myeloid leukemia. Nat. Med. 18, 605–611 (2012).
Boutzen, H. et al. Isocitrate dehydrogenase 1 mutations prime the all-trans retinoic acid myeloid differentiation pathway in acute myeloid leukemia. J. Exp. Med. 213, 483–497 (2016).
Gianni, M. et al. p38αMAPK interacts with and inhibits RARα: suppression of the kinase enhances the therapeutic activity of retinoids in acute myeloid leukemia cells. Leukemia 26, 1850–1861 (2012).
Shao, X. et al. The HER2 inhibitor TAK165 sensitizes human acute myeloid leukemia cells to retinoic acid-induced myeloid differentiation by activating MEK/ERK mediated RARα/STAT1 axis. Sci. Rep. 6, 24589 (2016).
Zeng, W. et al. Targeting to the non-genomic activity of retinoic acid receptor-gamma by acacetin in hepatocellular carcinoma. Sci. Rep. 7, 348 (2017).
Verhagen, H. J. et al. Primary acute myeloid leukemia cells with overexpression of EVI-1 are sensitive to all-trans retinoic acid. Blood 127, 458–463 (2016).
Churchman, M. L. et al. Efficacy of retinoids in IKZF1-mutated BCR-ABL1 acute lymphoblastic leukemia. Cancer Cell 28, 343–356 (2015).
Lagishetty, V., Liu, N. Q. & Hewison, M. Vitamin D metabolism and innate immunity. Mol. Cell Endocrinol. 347, 97–105 (2011).
Rigby, W. F., Shen, L., Ball, E. D., Guyre, P. M. & Fanger, M. W. Differentiation of a human monocytic cell line by 1,25-dihydroxyvitamin D3 (calcitriol): a morphologic, phenotypic, and functional analysis. Blood 64, 1110–1115 (1984).
Howell, A. L., Stukel, T. A., Bloomfield, C. D., Davey, F. R. & Ball, E. D. Induction of differentiation in blast cells and leukemia colony-forming cells from patients with acute myeloid leukemia. Blood 75, 721–729 (1990).
Hughes, P. J., Marcinkowska, E., Gocek, E., Studzinski, G. P. & Brown, G. Vitamin D3-driven signals for myeloid cell differentiation — implications for differentiation therapy. Leuk. Res. 34, 553–565 (2010).
Paubelle, E. et al. Deferasirox and vitamin D improves overall survival in elderly patients with acute myeloid leukemia after demethylating agents failure. PLoS ONE 8, e65998 (2013).
Shadel, G. S. & Horvath, T. L. Mitochondrial ROS signaling in organismal homeostasis. Cell 163, 560–569 (2015).
Bigarella, C. L., Liang, R. & Ghaffari, S. Stem cells and the impact of ROS signaling. Development 141, 4206–4218 (2014).
Abdel-Wahab, O. & Levine, R. L. Metabolism and the leukemic stem cell. J. Exp. Med. 207, 677–680 (2010).
Liu, C. X., Zhou, H. C., Yin, Q. Q., Wu, Y. L. & Chen, G. Q. Targeting peroxiredoxins against leukemia. Exp. Cell Res. 319, 170–176 (2013).
Roth, M. et al. Eltrombopag inhibits the proliferation of leukemia cells via reduction of intracellular iron and induction of differentiation. Blood 120, 386–394 (2012).
Callens, C. et al. Targeting iron homeostasis induces cellular differentiation and synergizes with differentiating agents in acute myeloid leukemia. J. Exp. Med. 207, 731–750 (2010).
Sykes, D. B. et al. Inhibition of dihydroorotate dehydrogenase overcomes differentiation blockade in acute myeloid leukemia. Cell 167, 171–186.e15 (2016).
Wen, Q. J. et al. Targeting megakaryocytic-induced fibrosis in myeloproliferative neoplasms by AURKA inhibition. Nat. Med. 21, 1473–1480 (2015).
Wen, Q. et al. Identification of regulators of polyploidization presents therapeutic targets for treatment of AMKL. Cell 150, 575–589 (2012).
Ferrero, D., Tarella, C., Gallo, E., Ruscetti, F. W. & Breitman, T. R. Terminal differentiation of the human promyelocytic leukemia cell line, HL-60, in the absence of cell proliferation. Cancer Res. 42, 4421–4426 (1982).
Griffin, J., Munroe, D., Major, P. & Kufe, D. Induction of differentiation of human myeloid leukemia cells by inhibitors of DNA synthesis. Exp. Hematol. 10, 774–781 (1982).
Marks, P. et al. Histone deacetylases and cancer: causes and therapies. Nat. Rev. Cancer 1, 194–202 (2001).
Johnstone, R. W. Histone-deacetylase inhibitors: novel drugs for the treatment of cancer. Nat. Rev. Drug Discov. 1, 287–299 (2002).
Ferrara, F. F. et al. Histone deacetylase-targeted treatment restores retinoic acid signaling and differentiation in acute myeloid leukemia. Cancer Res. 61, 2–7 (2001).
Gottlicher, M. et al. Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. EMBO J. 20, 6969–6978 (2001).
Minucci, S. & Pelicci, P. G. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat. Rev. Cancer 6, 38–51 (2006).
Bolden, J. E., Peart, M. J. & Johnstone, R. W. Anticancer activities of histone deacetylase inhibitors. Nat Rev. Drug Discov. 5, 769–784 (2006).
Leiva, M. et al. Valproic acid induces differentiation and transient tumor regression, but spares leukemia-initiating activity in mouse models of APL. Leukemia 26, 1630–1637 (2012).
Matthews, G. M. et al. Functional-genetic dissection of HDAC dependencies in mouse lymphoid and myeloid malignancies. Blood 126, 2392–2403 (2015).
Santoro, F. et al. A dual role for Hdac1: oncosuppressor in tumorigenesis, oncogene in tumor maintenance. Blood 121, 3459–3468 (2013).
Falkenberg, K. J. & Johnstone, R. W. Histone deacetylases and their inhibitors in cancer, neurological diseases and immune disorders. Nat. Rev. Drug Discov. 13, 673–691 (2014).
Loven, J. et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 153, 320–334 (2013).
Puissant, A. et al. Targeting MYCN in neuroblastoma by BET bromodomain inhibition. Cancer Discov. 3, 308–323 (2013).
Zuber, J. et al. RNAi screen identifies Brd4 as a therapeutic target in acute myeloid leukaemia. Nature 478, 524–528 (2011).
Filippakopoulos, P. et al. Selective inhibition of BET bromodomains. Nature 468, 1067–1073 (2010).
Coude, M. M. et al. BET inhibitor OTX015 targets BRD2 and BRD4 and decreases c-MYC in acute leukemia cells. Oncotarget 6, 17698–17712 (2015).
Berthon, C. et al. Bromodomain inhibitor OTX015 in patients with acute leukaemia: a dose-escalation, phase 1 study. Lancet Haematol. 3, e186–e195 (2016).
Rathert, P. et al. Transcriptional plasticity promotes primary and acquired resistance to BET inhibition. Nature 525, 543–547 (2015).
Fong, C. Y. et al. BET inhibitor resistance emerges from leukaemia stem cells. Nature 525, 538–542 (2015).
Schwartz, B. E. et al. Differentiation of NUT midline carcinoma by epigenomic reprogramming. Cancer Res. 71, 2686–2696 (2011).
Stathis, A. et al. Clinical response of carcinomas harboring the BRD4-NUT oncoprotein to the targeted bromodomain inhibitor OTX015/MK-8628. Cancer Discov. 6, 492–500 (2016).
Cheson, B. D., Jasperse, D. M., Simon, R. & Friedman, M. A. A critical appraisal of low-dose cytosine arabinoside in patients with acute non-lymphocytic leukemia and myelodysplastic syndromes. J. Clin. Oncol. 4, 1857–1864 (1986).
Rasmussen, K. D. & Helin, K. Role of TET enzymes in DNA methylation, development, and cancer. Genes Dev. 30, 733–750 (2016).
Yang, L., Rau, R. & Goodell, M. A. DNMT3A in haematological malignancies. Nat. Rev. Cancer 15, 152–165 (2015).
Navada, S. C., Steinmann, J., Lubbert, M. & Silverman, L. R. Clinical development of demethylating agents in hematology. J. Clin. Invest. 124, 40–46 (2014).
Dawson, M. A. & Kouzarides, T. Cancer epigenetics: from mechanism to therapy. Cell 150, 12–27 (2012).
Jain, M. et al. Sustained loss of a neoplastic phenotype by brief inactivation of MYC. Science 297, 102–104 (2002).
Shachaf, C. M. et al. MYC inactivation uncovers pluripotent differentiation and tumour dormancy in hepatocellular cancer. Nature 431, 1112–1117 (2004).
Zuber, J. et al. An integrated approach to dissecting oncogene addiction implicates a Myb-coordinated self-renewal program as essential for leukemia maintenance. Genes Dev. 25, 1628–1640 (2011).
Zuber, J. et al. Toolkit for evaluating genes required for proliferation and survival using tetracycline-regulated RNAi. Nat. Biotechnol. 29, 79–83 (2011).
Bots, M. et al. Differentiation therapy for the treatment of t(8;21) acute myeloid leukemia using histone deacetylase inhibitors. Blood 123, 1341–1352 (2014).
El Hajj, H. et al. Therapy-induced selective loss of leukemia-initiating activity in murine adult T cell leukemia. J. Exp. Med. 207, 2785–2792 (2010).
El-Sabban, M. E. et al. Arsenic-interferon-α-triggered apoptosis in HTLV-I transformed cells is associated with tax down-regulation and reversal of NF-κB activation. Blood 96, 2849–2855 (2000).
Kchour, G. et al. Phase 2 study of the efficacy and safety of the combination of arsenic trioxide, interferon α, and zidovudine in newly diagnosed chronic adult T-cell leukemia/lymphoma (ATL). Blood 113, 6528–6532 (2009).
Figueroa, M. E. et al. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell 18, 553–567 (2010).
Lu, C. et al. IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature 483, 474–478 (2012).
Losman, J. A. et al. (R)-2-hydroxyglutarate is sufficient to promote leukemogenesis and its effects are reversible. Science 339, 1621–1625 (2013).
Chaturvedi, A. et al. Mutant IDH1 promotes leukemogenesis in vivo and can be specifically targeted in human AML. Blood 122, 2877–2887 (2013).
Kats, L. M. et al. Proto-oncogenic role of mutant IDH2 in leukemia initiation and maintenance. Cell Stem Cell 14, 329–341 (2014).
Wang, F. et al. Targeted inhibition of mutant IDH2 in leukemia cells induces cellular differentiation. Science 340, 622–626 (2013).
Kernytsky, A. et al. IDH2 mutation-induced histone and DNA hypermethylation is progressively reversed by small-molecule inhibition. Blood 125, 296–303 (2015).
Stein, E. M. et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood 130, 722–731 (2017).
Chaturvedi, A. et al. Pan-mutant-IDH1 inhibitor BAY1436032 is highly effective against human IDH1 mutant acute myeloid leukemia in vivo. Leukemia 31, 2020–2028 (2017).
Amatangelo, M. D. et al. Enasidenib induces acute myeloid leukemia cell differentiation to promote clinical response. Blood 130, 732–741 (2017).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Storm, E. E. et al. Targeting PTPRK-RSPO3 colon tumours promotes differentiation and loss of stem-cell function. Nature 529, 97–100 (2016).
Yan, M. et al. IKKalpha restoration via EZH2 suppression induces nasopharyngeal carcinoma differentiation. Nat. Commun. 5, 3661 (2014).
Grunwald, F. et al. Redifferentiation therapy with retinoic acid in follicular thyroid cancer. J. Nucl. Med. 39, 1555–1558 (1998).
Schmutzler, C., Winzer, R., Meissner-Weigl, J. & Kohrle, J. Retinoic acid increases sodium/iodide symporter mRNA levels in human thyroid cancer cell lines and suppresses expression of functional symporter in nontransformed FRTL-5 rat thyroid cells. Biochem. Biophys. Res. Commun. 240, 832–838 (1997).
Handkiewicz-Junak, D. et al. 13-cis-retinoic acid re-differentiation therapy and recombinant human thyrotropin-aided radioiodine treatment of non-functional metastatic thyroid cancer: a single-center, 53-patient phase 2 study. Thyroid Res. 2, 8 (2009).
Fernandez, C. A. et al. Effectiveness of retinoic acid treatment for redifferentiation of thyroid cancer in relation to recovery of radioiodine uptake. J. Endocrinol. Invest. 32, 228–233 (2009).
Marquette, A., Andre, J., Bagot, M., Bensussan, A. & Dumaz, N. ERK and PDE4 cooperate to induce RAF isoform switching in melanoma. Nat. Struct. Mol. Biol. 18, 584–591 (2011).
Lehraiki, A. et al. Inhibition of melanogenesis by the antidiabetic metformin. J. Invest. Dermatol. 134, 2589–2597 (2014).
Pattabiraman, D. R. et al. Activation of PKA leads to mesenchymal-to-epithelial transition and loss of tumor-initiating ability. Science 351, aad3680 (2016).
Pattabiraman, D. R. & Weinberg, R. A. Targeting the epithelial-to-mesenchymal transition: the case for differentiation-based therapy. Cold Spring Harb. Symp. Quant. Biol. 81, 11–19 (2017).
Xing, F. et al. The anti-Warburg effect elicited by the camp-pgc1alpha pathway drives differentiation of glioblastoma cells into astrocytes. Cell Rep. 18, 468–481 (2017).
Saha, S. K. et al. Mutant IDH inhibits HNF-4α to block hepatocyte differentiation and promote biliary cancer. Nature 513, 110–114 (2014).
Pusch, S. et al. Pan-mutant IDH1 inhibitor BAY 1436032 for effective treatment of IDH1 mutant astrocytoma in vivo. Acta Neuropathol. 133, 629–644 (2017).
Turcan, S. et al. Efficient induction of differentiation and growth inhibition in IDH1 mutant glioma cells by the DNMT inhibitor decitabine. Oncotarget 4, 1729–1736 (2013).
Abemayor, E., Chang, B. & Sidell, N. Effects of retinoic acid on the in vivo growth of human neuroblastoma cells. Cancer Lett. 55, 1–5 (1990).
Robson, J. A. & Sidell, N. Ultrastructural features of a human neuroblastoma cell line treated with retinoic acid. Neuroscience 14, 1149–1162 (1985).
Thiele, C. J., Reynolds, C. P. & Israel, M. A. Decreased expression of N-myc precedes retinoic acid-induced morphological differentiation of human neuroblastoma. Nature 313, 404–406 (1985).
Moore, T. B., Koeffler, H. P., Yamashiro, J. M. & Wada, R. K. Vitamin D3 analogs inhibit growth and induce differentiation in LA-N-5 human neuroblastoma cells. Clin. Exp. Metastasis 14, 239–245 (1996).
Rupniak, H. T. et al. Characteristics of a new human neuroblastoma cell line which differentiates in response to cyclic adenosine 3′:5′-monophosphate. Cancer Res. 44, 2600–2607 (1984).
Wuarin, L., Verity, M. A. & Sidell, N. Effects of interferon-gamma and its interaction with retinoic acid on human neuroblastoma differentiation. Int. J. Cancer 48, 136–141 (1991).
Villablanca, J. G. et al. Phase I trial of 13-cis-retinoic acid in children with neuroblastoma following bone marrow transplantation. J. Clin. Oncol. 13, 894–901 (1995).
Matthay, K. K. et al. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. N. Engl. J. Med. 341, 1165–1173 (1999).
Matthay, K. K. et al. Long-term results for children with high-risk neuroblastoma treated on a randomized trial of myeloablative therapy followed by 13-cis-retinoic acid: a children's oncology group study. J. Clin. Oncol. 27, 1007–1013 (2009).
Han, G., Chang, B., Connor, M. J. & Sidell, N. Enhanced potency of 9-cis versus all-trans-retinoic acid to induce the differentiation of human neuroblastoma cells. Differentiation 59, 61–69 (1995).
Yen, K. et al. AG-221, a first-in-class therapy targeting acute myeloid leukemia harboring oncogenic IDH2 mutations. Cancer Discov. 7, 478–493 (2017).
Nebbioso, A. et al. Tumor-selective action of HDAC inhibitors involves TRAIL induction in acute myeloid leukemia cells. Nat. Med. 11, 77–84 (2005).
Kurimchak, A. M. et al. Resistance to BET bromodomain inhibitors is mediated by kinome reprogramming in ovarian cancer. Cell Rep. 16, 1273–1286 (2016).
Prost, S. et al. Erosion of the chronic myeloid leukaemia stem cell pool by PPARγ agonists. Nature 525, 380–383 (2015).
Medyouf, H. The microenvironment in human myeloid malignancies: emerging concepts and therapeutic implications. Blood 129, 1617–1626 (2017).
Ghiaur, G. et al. Regulation of human hematopoietic stem cell self-renewal by the microenvironment's control of retinoic acid signaling. Proc. Natl Acad. Sci. USA 110, 16121–16126 (2013).
Lujambio, A. et al. Non-cell-autonomous tumor suppression by p53. Cell 153, 449–460 (2013).
Xue, W. et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445, 656–660 (2007).
Rakhra, K. et al. CD4+ T cells contribute to the remodeling of the microenvironment required for sustained tumor regression upon oncogene inactivation. Cancer Cell 18, 485–498 (2010).
Casey, S. C. et al. MYC regulates the antitumor immune response through CD47 and PD-L1. Science 352, 227–231 (2016).
Padua, R. A. et al. PML-RARA-targeted DNA vaccine induces protective immunity in a mouse model of leukemia. Nat. Med. 9, 1413–1417 (2003).
Westervelt, P. et al. Adaptive immunity cooperates with liposomal all-trans-retinoic acid (ATRA) to facilitate long-term molecular remissions in mice with acute promyelocytic leukemia. Proc. Natl Acad. Sci. USA 99, 9468–9473 (2002).
Guerriero, J. L. et al. Class IIa HDAC inhibition reduces breast tumours and metastases through anti-tumour macrophages. Nature 543, 428–432 (2017).
Hogg, S. J. et al. BET-bromodomain inhibitors engage the host immune system and regulate expression of the immune checkpoint ligand PD-L1. Cell Rep. 18, 2162–2174 (2017).
Kagoya, Y. et al. BET bromodomain inhibition enhances T cell persistence and function in adoptive immunotherapy models. J. Clin. Invest. 126, 3479–3494 (2016).
Hall, J. A., Grainger, J. R., Spencer, S. P. & Belkaid, Y. The role of retinoic acid in tolerance and immunity. Immunity 35, 13–22 (2011).
Amankulor, N. M. et al. Mutant IDH1 regulates the tumor-associated immune system in gliomas. Genes Dev. 31, 774–786 (2017).
Chen, J. Y. et al. The oncometabolite R-2-hydroxyglutarate activates NF-κB-dependent tumor-promoting stromal niche for acute myeloid leukemia cells. Sci. Rep. 6, 32428 (2016).
Licht, J. D. DNA methylation inhibitors in cancer therapy: the immunity dimension. Cell 162, 938–939 (2015).
Kreso, A. & Dick, J. E. Evolution of the cancer stem cell model. Cell Stem Cell 14, 275–291 (2014).
Beck, B. & Blanpain, C. Unravelling cancer stem cell potential. Nat. Rev. Cancer 13, 727–738 (2013).
Breitman, T. R., Collins, S. J. & Keene, B. R. Terminal differentiation of human promyelocytic leukemic cells in primary culture in response to retinoic acid. Blood 57, 1000–1004 (1981).
Breitman, T. R., Selonick, S. E. & Collins, S. J. Induction of differentiation of the human promyelocytic leukemia cell line (HL-60) by retinoic acid. Proc. Natl Acad. Sci. USA 77, 2936 (1980).
Borrow, J., Goddart, A., Sheer, D. & Solomon, E. Molecular analysis of acute promyelocytic leukemia breakpoint cluster region on chromosome 17. Science 249, 1577–1580 (1990).
Lin, R. J. et al. Role of the histone deacetylase complex in acute promyelocytic leukaemia. Nature 391, 811–814 (1998).
de Thé, H., Vivanco-Ruiz, M.d. M., Tiollais, P., Stunnenberg, H. & Dejean, A. Identification of a retinoic acid responsive element in the retinoic acid receptor beta gene. Nature 343, 177–180 (1990).
Oussama, A., Lo-Coco, F. & Sanz, M. A. Acute Promyelocytic Leukemia: A Clinical Guide (Springer International Publishing, 2018).
Rego, E. M. et al. Retinoic acid (RA) and As2O3 treatment in transgenic models of acute promyelocytic leukemia (APL) unravel the distinct nature of the leukemogenic process induced by the PML-RARα and PLZF-RARα oncoproteins. Proc. Natl Acad. Sci. USA 97, 10173–10178 (2000).
He, L.-Z. et al. Distinct interactions of PML-RARα and PLZF-RARα with co-repressors determine differential responses to RA in APL. Nat. Genet. 18, 126–135 (1998).
He, L. et al. Two critical hits for promyelocytic leukemia. Mol. Cell 6, 1131–1141 (2000).
Koken, M. H. M. et al. Retinoic acid, but not arsenic trioxide, degrades the PLZF/RARα fusion protein, without inducing terminal differentiation or apoptosis, in a RA-therapy resistant tt(11;17)(q23;q21) APL patient. Oncogene 18, 1113–1118 (1999).
Lallemand-Breitenbach, V. et al. Retinoic acid and arsenic synergize to eradicate leukemic cells in a mouse model of acute promyelocytic leukemia. J. Exp. Med. 189, 1043–1052 (1999).
Acknowledgements
The author's laboratory is supported by Collège de France, INSERM, Centre National de la Recherche Scientifique (CNRS), Université Paris-Diderot, Ligue Contre le Cancer, Institut National du Cancer and the European Research Council. The author apologizes to investigators whose work could not be cited because of space constraints. The author thanks F. Maloumian for help with the figures and friends and colleagues for critical reading of the manuscript, in particular A. Bazarbachi, O. Hermine, L. Degos, P. Fenaux and V. Lallemand-Breitenbach.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
de Thé, H. Differentiation therapy revisited. Nat Rev Cancer 18, 117–127 (2018). https://doi.org/10.1038/nrc.2017.103
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2017.103
This article is cited by
-
Nucleic acid therapeutics as differentiation agents for myeloid leukemias
Leukemia (2024)
-
Identifying regulators of aberrant stem cell and differentiation activity in colorectal cancer using a dual endogenous reporter system
Nature Communications (2024)
-
Resveratrol as sensitizer in colorectal cancer plasticity
Cancer and Metastasis Reviews (2024)
-
The strategies to cure cancer patients by eradicating cancer stem-like cells
Molecular Cancer (2023)
-
Rutaecarpine induces the differentiation of triple-negative breast cancer cells through inhibiting fumarate hydratase
Journal of Translational Medicine (2023)