Abstract
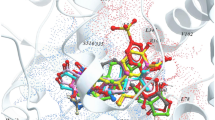
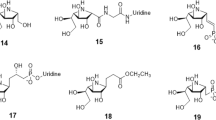
CD59 is a potent inhibitor of the complement membrane attack complex (MAC) action that acts by binding to the C8 and C9 complements of the assembling MAC, thereby preventing incorporation of the multiple copies of C9 required for complete formation of the osmolytic pore. Human CD59 low expression leads to atherosclerosis. Therefore, computational approach methods were implemented herein to design novel activators for CD59. Crystal structure (PDB ID: 1CDR) of CD59 was investigated to locate N-acetyl glucosamine (NAG) active site residues (Leu1, Gln2, Val17, Asn18, Ser20, Ser21, Asp22 and Asp67). CD59 is known for its binding affinity towards NAG and Alpha-L-Fucose, hence, were explored against more than one million entries of Ligand.Info metadatabase to create an in-house library of 708 compounds. Ligand dataset was prepared using LigPrep and filtered based on Lipinski’s rule of five and reactive group constraints. The crystal structure was optimized and energy was minimized applying OPLS force field in Maestro v9.2. Sixty four ligands were found to have binding affinity towards CD59 through virtual screening workflow of Maestro. Two ligands namely mizoribine (-8.56 kcal/mol) and gluconolactone (-8.39 kcal/mol) with better XPG score compared to NAG (-8.02 kcal/mol) were proposed potential CD59 activators. Analysis of docking complexes for both proposed leads revealed a stable hydrogen bond network, good van der Waal interactions and good agreement with NAG binding orientations. Thus, mizoribine and gluconolactone would be useful for developing potential drug molecules for atherosclerosis.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lakshmi Narayana Rao, C., Pradhan, D. & Umamaheswari, A. Exploring NAG active site of human CD59 towards discovery of novel activators for treatment of atherosclerosis. Nat Prec (2011). https://doi.org/10.1038/npre.2011.6531.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2011.6531.1