Abstract
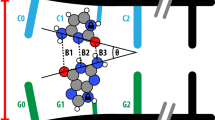
Multiple H-bonded base-pairing as a fundamental element of DNA structure was first described by Watson and Crick using stable keto tautomer forms. In their analysis, they considered the possibility of mutations via tautomeric proton transfer shifts. Among other phenomena, such shifts can be caused by electronic excitation; for example, anomalous adenine-cytosine pairing may be a result of two-proton phototautomerism. One suitable model base pair for two-proton translocating tautomerization is the C2h dimer of 7-azaindole (7AI) proposed by Taylor et al. in 1969. The most salient contribu&x00AC;tion of their work was that the double proton transfer in such a dimer occurs in a concerted manner. After the strong controversy raised in 1995 by the proposal of Douhal et al. of a stepwise mechanism for the process was overcome, its concerted nature has been strongly supported by available evidence (see references and references therein). Very recently, however, Kwon and Zewail claimed to have obtained new supportive evidence that the proton phototransfer in 7AI dimer in polar solvents is in fact a stepwise rather than concerted process. In this communication, we conducted a systematic spectroscopic study of the dimerization of 7AI at a 0.1 M concentration in acetonitrile that may allow us to demonstrate whether the 0.1 M 7AI solutions in acetonitrile at room temperature used by Kwon and Zewail contained enough 7AI doubly hydrogen bonded dimer to enable its photoselection and hence its photophysical characterization with femtosecond / fluorescence spectroscopy.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Catalan, J. On the Doubly hydrogen bonded dimer of 7-azaindole (0.1 M) as a model for DNA base pairs in acetonitrile solutions at rt. Nat Prec (2008). https://doi.org/10.1038/npre.2008.1926.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2008.1926.1