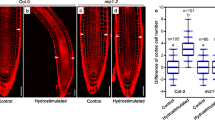
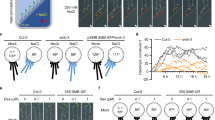
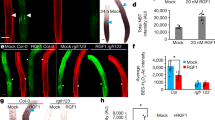
Abstract
Plants can acclimate by using tropisms to link the direction of growth to environmental conditions. Hydrotropism allows roots to forage for water, a process known to depend on abscisic acid (ABA) but whose molecular and cellular basis remains unclear. Here we show that hydrotropism still occurs in roots after laser ablation removed the meristem and root cap. Additionally, targeted expression studies reveal that hydrotropism depends on the ABA signalling kinase SnRK2.2 and the hydrotropism-specific MIZ1, both acting specifically in elongation zone cortical cells. Conversely, hydrotropism, but not gravitropism, is inhibited by preventing differential cell-length increases in the cortex, but not in other cell types. We conclude that root tropic responses to gravity and water are driven by distinct tissue-based mechanisms. In addition, unlike its role in root gravitropism, the elongation zone performs a dual function during a hydrotropic response, both sensing a water potential gradient and subsequently undergoing differential growth.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Blancaflor, E. B., Fasano, J. M. & Gilroy, S. Mapping the functional roles of cap cells in the response of Arabidopsis primary roots to gravity. Plant Physiol. 116, 213–222 (1998).
Ottenschlager, I. et al. Gravity-regulated differential auxin transport from columella to lateral root cap cells. Proc. Natl Acad. Sci. USA 100, 2987–2991 (2003).
Swarup, R. et al. Root gravitropism requires lateral root cap and epidermal cells for transport and response to a mobile auxin signal. Nat. Cell Biol. 7, 1057–1065 (2005).
Rahman, A. et al. Gravitropism of Arabidopsis thaliana roots requires the polarization of PIN2 toward the root tip in meristematic cortical cells. Plant Cell 22, 1762–1776 (2010).
Friml, J. Subcellular trafficking of PIN auxin efflux carriers in auxin transport. Eur. J. Cell Biol. 89, 231–235 (2010).
Jaffe, M. J., Takahashi, H. & Biro, R. L. A pea mutant for the study of hydrotropism in roots. Science 230, 445–447 (1985).
Takahashi, H. & Suge, H. Root hydrotropism of an agravitropic pea mutant, ageotropum. Physiol. Plant. 82, 24–31 (1991).
Takahashi, H. & Scott, T. K. Intensity of hydrostimulation for the induction of root hydrotropism and its sensing by the root cap. Plant Cell Environ. 16, 99–103 (1993).
Miyazawa, Y. et al. Effects of locally targeted heavy-ion and laser microbeam on root hydrotropism in Arabidopsis thaliana. J. Radiat. Res. 49, 373–379 (2008).
Miyamoto, N., Ookawa, T., Takahashi, H. & Hirasawa, T. Water uptake and hydraulic properties of elongating cells in hydrotropically bending roots of Pisum sativum L. Plant Cell Physiol. 43, 393–401 (2002).
Kaneyasu, T. et al. Auxin response, but not its polar transport, plays a role in hydrotropism of Arabidopsis roots. J. Exp. Bot. 58, 1143–1150 (2007).
Takahashi, N., Goto, N., Okada, K. & Takahashi, H. Hydrotropism in abscisic acid, wavy, and gravitropic mutants of Arabidopsis thaliana. Planta 216, 203–211 (2002).
Takahashi, H., Miyazawa, Y. & Fujii, N. Hormonal interactions during root tropic growth: hydrotropism versus gravitropism. Plant Mol. Biol. 69, 489–502 (2009).
Shkolnik, D., Krieger, G., Nuriel, R. & Fromm, H. Hydrotropism: root bending does not require auxin redistribution. Mol. Plant 9, 757–759 (2016).
Shkolnik, D. & Fromm, H. The Cholodny-Went theory does not explain hydrotropism. Plant Sci. 252, 400–403 (2016).
Antoni, R. et al. PYRABACTIN RESISTANCE1-LIKE8 plays an important role for the regulation of abscisic acid signaling in root. Plant Physiol. 161, 931–941 (2013).
Kobayashi, A. et al. A gene essential for hydrotropism in roots. Proc. Natl Acad. Sci. USA 104, 4724–4729 (2007).
Moriwaki, T., Miyazawa, Y., Fujii, N. & Takahashi, H. Light and abscisic acid signalling are integrated by MIZ1 gene expression and regulate hydrotropic response in roots of Arabidopsis thaliana. Plant Cell Environ. 35, 1359–1368 (2012).
Moriwaki, T., Miyazawa, Y., Kobayashi, A. & Takahashi, H. Molecular mechanisms of hydrotropism in seedling roots of Arabidopsis thaliana (Brassicaceae). Am. J. Bot. 100, 25–34 (2013).
Cutler, S. R., Rodriguez, P. L., Finkelstein, R. R. & Abrams, S. R. Abscisic acid: Emergence of a core signaling network. Annu. Rev. Plant Biol. 61, 651–679 (2010).
Ma, Y. et al. Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 324, 1064–1068 (2009).
Park, S. Y. et al. Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324, 1068–1071 (2009).
Fujii, H. et al. In vitro reconstitution of an abscisic acid signalling pathway. Nature 462, 660–664 (2009).
Fujii, H., Verslues, P. E. & Zhu, J. K. Identification of two protein kinases required for abscisic acid regulation of seed germination, root growth, and gene expression in Arabidopsis. Plant Cell 19, 485–494 (2007).
Antoni, R., Dietrich, D., Bennett, M. J. & Rodriguez, P. L. Hydrotropism: analysis of the root response to a moisture gradient. Methods Mol. Biol. 1398, 3–9 (2016).
Kline, K. G., Barrett-Wilt, G. A. & Sussman, M. R. In planta changes in protein phosphorylation induced by the plant hormone abscisic acid. Proc. Natl Acad. Sci. USA 107, 15986–15991 (2010).
Wang, P. et al. Quantitative phosphoproteomics identifies SnRK2 protein kinase substrates and reveals the effectors of abscisic acid action. Proc. Natl Acad. Sci. USA 110, 11205–11210 (2013).
Casamitjana-Martinez, E. et al. Root-specific CLE19 overexpression and the sol1/2 suppressors implicate a CLV-like pathway in the control of Arabidopsis root meristem maintenance. Curr. Biol. 13, 1435–1441 (2003).
Willemsen, V. et al. The NAC domain transcription factors FEZ and SOMBRERO control the orientation of cell division plane in Arabidopsis root stem cells. Dev. Cell 15, 913–922 (2008).
Lee, M. M. & Schiefelbein, J. WEREWOLF, a MYB-related protein in Arabidopsis, is a position-dependent regulator of epidermal cell patterning. Cell 99, 473–483 (1999).
Wysocka-Diller, J. W., Helariutta, Y., Fukaki, H., Malamy, J. E. & Benfey, P. N. Molecular analysis of SCARECROW function reveals a radial patterning mechanism common to root and shoot. Development 127, 595–603 (2000).
Heidstra, R., Welch, D. & Scheres, B. Mosaic analyses using marked activation and deletion clones dissect Arabidopsis SCARECROW action in asymmetric cell division. Genes & Dev. 18, 1964–1969 (2004).
Lee, J. Y. et al. Transcriptional and posttranscriptional regulation of transcription factor expression in Arabidopsis roots. Proc. Natl Acad. Sci. USA 103, 6055–6060 (2006).
Ondzighi-Assoume, C. A., Chakraborty, S. & Harris, J. M. Environmental nitrate stimulates abscisic acid accumulation in Arabidopsis root tips by releasing it from inactive stores. Plant Cell 28, 729–745 (2016).
Sharp, R. E., Wu, Y. J., Voetberg, G. S., Saab, I. N. & Lenoble, M. E. Confirmation that abscisic-acid accumulation is required for maize primary root elongation at low water potentials. J. Exp. Bot. 45, 1743–1751 (1994).
Xu, W. F. et al. Abscisic acid accumulation modulates auxin transport in the root tip to enhance proton secretion for maintaining root growth under moderate water stress. New Phytol. 197, 139–150 (2013).
Rowe, J. H., Topping, J. F., Liu, J. & Lindsey, K. Abscisic acid regulates root growth under osmotic stress conditions via an interacting hormonal network with cytokinin, ethylene and auxin. New Phytol. 211, 225–239 (2016).
Dyson, R. J. et al. Mechanical modelling quantifies the functional importance of outer tissue layers during root elongation and bending. New Phytol. 202, 1212–1222 (2014).
Churchman, M. L. et al. SIAMESE, a plant-specific cell cycle regulator, controls endoreplication onset in Arabidopsis thaliana. Plant Cell 18, 3145–3157 (2006).
Brunoud, G. et al. A novel sensor to map auxin response and distribution at high spatio-temporal resolution. Nature 482, 103–106 (2012).
Band, L. R. et al. Root gravitropism is regulated by a transient lateral auxin gradient controlled by a tipping-point mechanism. Proc. Natl Acad. Sci. USA 109, 4668–4673 (2012).
Mullen, J. L., Ishikawa, H. & Evans, M. L. Analysis of changes in relative elemental growth rate patterns in the elongation zone of Arabidopsis roots upon gravistimulation. Planta 206, 598–603 (1998).
Krieger, G., Shkolnik, D., Miller, G. & Fromm, H. Reactive oxygen species tune root tropic responses. Plant Physiol. 172, 1209–1220 (2016).
Wells, D. M. et al. Recovering the dynamics of root growth and development using novel image acquisition and analysis methods. Philos. T. R. Soc. B 367, 1517–1524 (2012).
French, A. P. et al. Identifying biological landmarks using a novel cell measuring image analysis tool: Cell-o-Tape. Plant Methods 8, 7 (2012).
Baskin, T. I. Patterns of root growth acclimation: constant processes, changing boundaries. WIREs Dev. Biol. 2, 65–73 (2013).
Acknowledgements
The authors thank C. Howells, K. Swarup and M. Whitworth for technical assistance, J.-K. Zhu for providing snrk2.2 snrk2.3 seeds, W. Grunewald for pDONR-L1-GAL4-VP16-R2 and S. Tsukinoki for generating WER:MIZ1-GFP(HSPter) and PIN2:MIZ1-GFP(HSPter) transgenic plants and acknowledge the following funding agencies for financial support: D.D., J.F., R.A., T.N., D.W., S.T., C.S., S.M., M.R.O., L.R.B., R.D., O.J., J.K., J.R., T.B. and M.J.B. thank the Biological and Biotechnology Science Research Council (BBSRC) for responsive mode and CISB awards to the Centre for Plant Integrative Biology; D.W., C.S., S.M., M.R.O., J.K., T.P. and M.J.B. thank the European Research Council (ERC) for FUTUREROOTS project funding; L.R.B. thanks the Leverhulme Trust for an Early Career Fellowship; V.B., R.B. and L.D.V. are supported by grants of the Research Foundation Flanders (G.002911N). R.B. and M.J.B. thank the Royal Society for Newton and Wolfson Research Fellowship awards; R.A., T.I.B. and M.J.B. thank the FP7 Marie Curie Fellowship Scheme; R.D. thanks the Engineering and Physical Sciences Research Council, J.D. and M.J.B. thank the GII scheme; and V.B., R.B., L.D.V. and M.J.B. thank the Interuniversity Attraction Poles Programme (IUAP P7/29 “MARS”), initiated by the Belgian Science Policy Office. R.B.P. was funded by grants from the Knut and Alice Wallenberg Foundation. This work was also supported by a Grant-in-Aid for Scientific Research on Innovative Areas (No. 22120004) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan to H.T., a Grant-in-Aid for Young Scientists (B) (No. 26870057) from the Japan Society for the Promotion of Science (JSPS) to A.K., a Grant-in-Aid for Scientific Research on Innovative Areas (No. 22120002) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan to A.N., a Grant-in-Aid for Scientific Research on Innovative Areas (No. 22120010) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan to Y.H. and the Funding Program for Next-Generation World-Leading Researchers (GS002) to Y.M. L.P. was financially supported by a scholarship from the Japanese government. T.-W.B. was financially supported by the Funding Program for Next-Generation World-Leading Researchers (GS002) and the Grant-in-Aid for Scientific Research on Innovative Areas (No. 22120004).
Author information
Authors and Affiliations
Contributions
D.D., L.P., A.K., J.F., V.B., R.B., R.A., T.N., S.H., T.-W.B., Y.M., D.M.W., S.T. and C.J.S. performed experimental work and data analysis and mathematical modelling. D.M.W., M.R.O., L.R.B., R.D., O.J., J.R.K., S.J.M., J.R., R.B., J.D., P.L.R., T.I.B., T.P., L.D.V., N.F., Y.M., A.N., Y.H., H.T. and M.J.B. oversaw project planning and discussed experimental results and modelling simulations. D.D., L.P., A.K., N.F., Y.M., T.I.B., H.T. and M.J.B. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–8, Supplementary Methods, Supplementary References, Supplementary Table 1, Supplementary Notes 1 and 2. (PDF 6852 kb)
Rights and permissions
About this article
Cite this article
Dietrich, D., Pang, L., Kobayashi, A. et al. Root hydrotropism is controlled via a cortex-specific growth mechanism. Nature Plants 3, 17057 (2017). https://doi.org/10.1038/nplants.2017.57
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2017.57
This article is cited by
-
Defects in the cell wall and its deposition caused by loss-of-function of three RLKs alter root hydrotropism in Arabidopsis thaliana
Nature Communications (2024)
-
Don’t be MIZguided, know where to grow!
Communications Biology (2024)
-
Protective role of manganese, proline and rice straw extract in wheat against drought driven oxidative stress
Acta Physiologiae Plantarum (2024)
-
Plant biotechnology research with single-cell transcriptome: recent advancements and prospects
Plant Cell Reports (2024)
-
In arid regions, the different intercropping systems reduce interspecific competition to improve system productivity
Plant and Soil (2024)