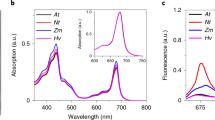
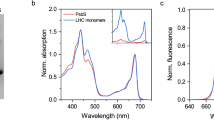
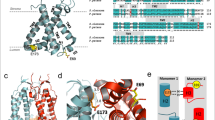
Abstract
To maintain high photosynthetic rates, plants must adapt to their light environment on a timescale of seconds to minutes. Therefore, the light-harvesting antenna system of photosystem II in thylakoid membranes, light-harvesting complex II (LHCII), has a feedback mechanism, which determines the proportion of absorbed energy dissipated as heat: non-photochemical chlorophyll fluorescence quenching (NPQ). This is crucial to prevent photo-oxidative damage to photosystem II (PSII) and is controlled by the transmembrane pH differences (ΔpH). High ΔpH activates NPQ by protonation of the protein PsbS and the enzymatic de-epoxidation of LHCII-bound violaxanthin to zeaxanthin. But the precise role of PsbS and its interactions with different LHCII complexes remain uncertain. We have investigated PsbS–LHCII interactions in native thylakoid membranes using magnetic-bead-linked antibody pull-downs. The interaction of PsbS with the antenna system is affected by both ΔpH and the level of zeaxanthin. In the presence of ΔpH alone, PsbS is found to be mainly associated with the trimeric LHCII protein polypeptides, Lhcb1, Lhcb2 and Lhcb3. However, a combination of ΔpH and zeaxanthin increases the proportion of PsbS bound to the minor LHCII antenna complex proteins Lhcb4, Lhcb5 and Lhcb6. This pattern of interaction is not influenced by the presence of PSII reactions centres. Similar to LHCII particles in the photosynthetic membrane, PsbS protein forms clusters in the NPQ state. NPQ recovery in the dark requires uncoupling of PsbS. We suggest that PsbS acts as a ‘seeding’ centre for the LHCII antenna rearrangement that is involved in NPQ.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Demmig-Adams, B., Garab, G., Adams, W. III & Govindjee . Non-Photochemical Quenching and Energy Dissipation in Plants, Algae and Cyanobacteria (Springer, 2014).
Jansson, S. A guide to the Lhc genes and their relatives in Arabidopsis. Trends Plant Sci. 4, 236–240 (1999).
Powles, S. B. Photoinhibition of photosynthesis induced by visible-light. Annu. Rev. Plant Physiol. Plant Mol. Biol. 35, 15–44 (1984).
Barber, J. Molecular basis of the vulnerability of photosystem II to damage by light. Aust. J. Plant Physiol. 22, 201–208 (1995).
Osmond, C. B. in Photoinhibition of Photosynthesis (eds Baker, N. R. & Bowyer, J. R. ) 1–24 (Bios Scientific Publishers, 1994).
Demmig-Adams, B. & Adams, W. W. Photoprotection and other responses of plants to high light stress. Annu. Rev. Plant Physiol. Plant Mol. Biol. 43, 599–626 (1992).
Li, Z., Wakao, S., Fischer, B. B. & Niyogi, K. K. Sensing and responding to excess light. Annu. Rev. Plant Biol. 60, 239–260 (2009).
Horton, P., Ruban, A. V. & Walters, R. G. Regulation of light harvesting in green plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 47, 655–684 (1996).
Niyogi, K. K. & Truong, T. B. Evolution of flexible non-photochemical quenching mechanisms that regulate light harvesting in oxygenic photosynthesis. Curr. Opin. Plant Biol. 16, 307–314 (2013).
Ruban, A. V. Evolution under the sun: optimizing light harvesting in photosynthesis. J. Exp. Bot. 66, 7–23 (2015).
Ruban, A. V. Non-photochemical chlorophyll fluorescence quenching: mechanism and effectiveness in protection against photodamage. Plant Physiol. 170, 1903–1916 (2016).
Kramer, D. M., Sacksteder, C. A. & Cruz, J. A. How acidic is the lumen? Photosynth. Res. 60, 151–163 (1999).
Liu, Z. et al. Crystal structure of spinach major light-harvesting complex at 2.72 Å resolution. Nature 428, 287–229 (2004).
Demmig-Adams, B. Carotenoids and photoprotection: a role for the xanthophyll zeaxanthin. Biochim. Biophys. Acta 1020, 1–2 (1990).
Yamamoto, H. Y., Nakayama, T. O. M. & Chichester, C. O. Studies on the light and dark interconversions of leaf xanthophylls. Arch. Biochem. Biophys. 97, 168–173 (1962).
Li, X. P. et al. A pigment-binding protein essential for regulation of photosynthetic light harvesting. Nature 403, 391–395 (2000).
Li, X. P. et al. Regulation of photosynthetic light harvesting involves intrathylakoid lumen pH sensing by the PsbS protein. J. Biol. Chem. 279, 22866–22287 (2004).
Fan, M. et al. Crystal structures of the PsbS protein essential for photoprotection. Nat. Struc. Mol. Biol. 22, 729–735 (2015).
Bergantino, E. et al. Light- and pH-dependent structural changes in the PsbS protein of photosystem II. Proc. Natl Acad. Sci. USA 100, 15265–15270 (2003).
Teardo, E. et al. Evidences for interaction of PsbS with photosynthetic complexes in maize thylakoids. Biochim. Biophys. Acta 1767, 703–711 (2007).
Wilk, L., Grunwald, M., Liao, P. N., Walla, P. J. & Kühlbrandt, W. Direct interaction of the major light-harvesting complex II and PsbS in nonphotochemical quenching. Proc. Natl Acad. Sci. USA 110, 5452–5456 (2013).
Gerotto, C., Franchin, C., Arrigoni, G. & Morosinotto, T. In vivo identification of photosystem II light harvesting complexes interacting with photosystem II subunit S. Plant Physiol. 168, 1747–1761 (2015).
Correa-Galvis, V., Poschmann, G., Melzer, M., Stühler, K. & Jahns, P. PsbS interactions involved in the activation of energy dissipation in Arabidopsis. Nat. Plants. 2, 15225 (2016).
Albanese, P. et al. Dynamic reorganization of photosystem II supercomplexes in response to variations in light intensities. Biochim. Biophys. Acta. 10, 651–660 (2016).
Johnson, M. P. & Ruban, A. V. Arabidopsis plants lacking PsbS protein possess photoprotective energy dissipation. Plant J. 61, 283–289 (2010).
Zia, A., Johnson, M. P. & Ruban, A. V. Acclimation- and mutation-induced enhancement of PsbS levels affects the kinetics of nonphotochemical quenching in Arabidopsis thaliana. Planta 233, 1253–1264 (2011).
Belgio, E., Johnson, M. P., Jurić, S. & Ruban, A. V. Higher plant photosystem II light-harvesting antenna, not the reaction center, determines the excited-state lifetime – both the maximum and the nonphotochemically quenched. Biophys. J. 102, 2761–2771 (2012).
Belgio, E., Ungerer, P. & Ruban, A. V. Light-harvesting superstructures of green plant chloroplasts lacking photosystems. Plant Cell Environ. 38, 2035–2047 (2015).
Ilioaia, C., Duffy, C. D. P. & Ruban, A. V. Changes in the energy transfer pathways within photosystem II antenna induced by the xanthophyll cycle activity. J. Phys. Chem. B 117, 5841–5847 (2013).
Johnson, M. P. et al. Photoprotective energy dissipation involves the reorganization of photosystem II light-harvesting complexes in the grana membranes of spinach chloroplasts. Plant Cell 23, 1468–1479 (2011).
Ware, M. A., Giovagnetti, V., Belgio, E. & Ruban, A. V. PsbS protein modulates non-photochemical chlorophyll fluorescence quenching in membranes depleted from photosystems. J. Photochem. Photobiol. B 152, 301–307 (2015).
Crouchman, S., Ruban, A. & Horton, P. PsbS enhances nonphotochemical fluorescence quenching in the absence of zeaxanthin. FEBS Lett. 580, 2053–2058 (2006).
Kereïche, S., Kiss, A. Z., Kouril, R., Boekema, E. & Horton, P. The PsbS protein controls the macro-organisation of photosystem II complexes in the grana membranes of higher plant chloroplasts. FEBS Lett. 584, 754–764 (2010).
Goral, T. et al. Light-harvesting antenna composition controls the macrostructure and dynamics of thylakoid membranes in Arabidopsis. Plant J. 69, 289–301 (2012).
Porra, R. J., Thompson, W. A. & Kriedemann, E. E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta. 975, 384–394 (1989).
Ruban, A. V. et al. Plasticity in the composition of the light harvesting antenna of higher plants preserves structural integrity and biological function. J. Biol. Chem. 281, 14981–14990 (2006).
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Kouřil, R., Dekker, J. P. & Boekema, E. J. Supramolecular structure of photosystem II in green plants. Biochim. Biophys. Acta 1817, 2–12 (2012).
Acknowledgements
A.V.R. acknowledges funding from the Biotechnology and Biological Sciences Research Council of the UK and the Leverhulme Trust. The authors acknowledge Y. Tian for the help with plant growth, as well as M. Johnson and C. Duffy for critical reading of the manuscript.
Author information
Authors and Affiliations
Contributions
J.S. and A.V.R. designed the experiments. J.S. performed biochemistry, PAM fluorescence measurements, preparation for TEM and particle analysis. V.G. performed PAM fluorescence measurements. P.U. assisted with the experiments. G.M. operated TEM and assisted with sample preparation. All authors discussed the results and commented on the manuscript. J.S., V.G. and A.V.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–5. (PDF 923 kb)
Rights and permissions
About this article
Cite this article
Sacharz, J., Giovagnetti, V., Ungerer, P. et al. The xanthophyll cycle affects reversible interactions between PsbS and light-harvesting complex II to control non-photochemical quenching. Nature Plants 3, 16225 (2017). https://doi.org/10.1038/nplants.2016.225
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2016.225
This article is cited by
-
A perspective on the major light-harvesting complex dynamics under the effect of pH, salts, and the photoprotective PsbS protein
Photosynthesis Research (2023)
-
Plant carotenoids: recent advances and future perspectives
Molecular Horticulture (2022)
-
A foliar pigment-based bioassay for interrogating chloroplast signalling revealed that carotenoid isomerisation regulates chlorophyll abundance
Plant Methods (2022)
-
The role of photosynthesis related pigments in light harvesting, photoprotection and enhancement of photosynthetic yield in planta
Photosynthesis Research (2022)
-
The PsbS protein and low pH are necessary and sufficient to induce quenching in the light-harvesting complex of plants LHCII
Scientific Reports (2021)