Abstract
In 2008, as part of the changes to develop integrated health care services in the Canterbury region of New Zealand, the local health board in collaboration with general practitioners, respiratory specialists and scientists introduced a programme for general practices to provide laboratory-quality spirometry in the community. The service adhered to the 2005 ATS/ERS international spirometry standards. The spirometry service was provided by trained practice nurses and community respiratory nurses, and was monitored and quality assured by certified respiratory scientists in the Respiratory Physiology Laboratory, Christchurch Hospital and CISO (Canterbury Initiative Services Organisation). These two organisations were responsible for organising training seminars and refresher courses on spirometry technique and interpretation of results. A total of 10 practices have now become approved spirometry providers, with the number of tests carried out in the primary care setting increasing gradually. Consistently high-quality spirometry tests have been obtained and are now presented on a centrally available results database for all hospital and community clinicians to review. Although the service has proved to be more convenient for patients, the tests have not been delivered as quickly as those carried out by the Respiratory Physiology Laboratory. However, the time scales for testing achieved by the community service is considered suitable for investigation of chronic disease. The success of the service has been dependent on several key factors including hospital and clinical support and a centralised quality assurance programme, a comprehensive training schedule and online clinical guidance and close integration between primary and secondary care clinicians.
Similar content being viewed by others
Introduction
The diagnosis of chronic obstructive pulmonary disease (COPD) requires the demonstration of airflow obstruction that is not fully reversible.1 Good-quality spirometry therefore needs to be readily available in primary care settings, where the majority of COPD diagnosis should be occurring. Without the availability of spirometry, COPD is misdiagnosed in 27% of patients.2 Unfortunately, spirometry performed in primary care settings is often of variable quality owing to inadequate training, poor quality control and insufficient patient throughput.3 To address this issue, primary and secondary care clinicians and respiratory scientists in Canterbury developed a laboratory-quality spirometry service in the community. This was delivered by general practices, with an agreed quality framework supported by Respiratory Specialist Services and the central hospital physiology laboratory.
The required development steps included provision of web-based clinical information for referring clinicians, linkage with an electronic referral management system, standardised testing systems, processes, training and education, web-based reporting of tests and filing of results in an electronic shared care view of the medical record.
Development and details of the community-based spirometry service
The Canterbury region has a population of 510,000, with 436,000 people living in greater Christchurch city.4 Before 2007, only Christchurch Hospital was performing spirometry to 2005 ATS/ERS standards. The Christchurch Respiratory Physiology Laboratory is a Thoracic Society of Australia and New Zealand (TSANZ) accredited laboratory employing Australian and New Zealand Society of Respiratory Science (ANZSRS) certified physiologists, and it performs ~850 spirometry tests on referrals from general practice each year, and in excess of 7,000 spirometry tests on hospital-referred patients. The number of referrals from general practitioners was anticipated to increase. Rather than increasing the capacity using respiratory scientists, it was considered more appropriate to establish spirometry services in primary care settings. The perceived advantages of this service, rather than an outreach service provided by the hospital respiratory laboratory, were to increase the knowledge and skills of general practice staff and to provide a familiar testing environment close to patients’ homes. This would build on existing infrastructure and skills within general practice.
In 2008, a spirometry working group agreed on a framework and specifications for a service in which general practice teams would be contracted to carry out spirometry testing in the community. The working group included primary and secondary clinicians, senior laboratory respiratory physiologists and senior management. Targeted general practices were given the opportunity to become contracted primary care providers for this testing, with the aim of establishing approved provider practices throughout Canterbury. The responsibility for referral management and testing remained with the approved provider practice, with the hospital respiratory department and physiology laboratory providing support, guidance and quality assurance. In areas where a local practice could not provide the service, mobile respiratory nurses were employed to deliver spirometry services in a local practice or other health care environment such as a community pharmacy.
The primary objectives of the community spirometry service were to provide laboratory-quality diagnostic spirometry within the community and to improve patient access to testing. The development of systems for continuous quality control and monitoring of results was also required. The standard requirements for the service were the internationally agreed guidelines for spirometry5 and primary care spirometry6 of the American Thoracic Society and European Respiratory Society (ATS/ERS). Using these guidelines, general practice teams can perform spirometry with high levels of technical skill and accuracy and to a similar standard as trained scientists.6 This acquired level of expertise is dependent on several factors including training, practice and refresher courses, the use of standardised spirometers, and ongoing quality assurance and feedback on interpretation of results.7
Training programme
Practice nurses at general practices that chose to provide spirometry testing, and community respiratory nurses, were required to attend a 2-day accredited training course, which focused on practical training for achieving high-quality spirometry results. This course provided information on the physiological basis of spirometry, the technical details of spirometry equipment, and associated quality assurance. The nurses were trained to carry out spirometry accurately with maximal subject co-operation and to assess independently the performance of the test and quality of the tracings. The nurses received further practical tuition by carrying out testing of patients in the Respiratory Physiology Laboratory under the supervision of respiratory scientists, and were required to complete a training log. The nurses attended a 1-day refresher course 12 months later, and participated in refresher courses every 3 years. Doctors from approved provider practices attended education courses in test reporting and interpretation of the spirometry recordings. These courses were coordinated by the Respiratory Physiology Laboratory and respiratory physicians.
Spirometry equipment and infection control
A single model of spirometer, the EasyOn PC ultrasonic flow-sensing device (ndd Medical Technologies Inc., Zurich, Switzerland), was selected for use in the service. This spirometer meets the specifications of the 2005 ATS/ERS pulmonary function guidelines5 and was selected because it was portable, it incorporated a quality assurance programme and calibration verification method, and it allowed the export of data. We chose one spirometer for all providers because only one procedure for data export was desired. On each day of use, the nurses verified the accuracy of the spirometer at three different flow rates using a 3-L calibration syringe (i.e., calibration error <±3.5%). Each general practice maintained a spirometer maintenance log and were required to implement agreed biological and infection control measures.
Spirometry procedure and interpretation
Spirometry was undertaken to ATS/ERS standards6 with at least three technically acceptable and repeatable efforts being recorded. Bronchodilator reversibility was undertaken on all patients, even if pre-bronchodilator lung function was within the normal range. Patients who refused the bronchodilator or had a medical contraindication were not tested. The recordings were interpreted using the Hankinson reference values.8 In 2014, we adopted the Global Lung Initiative 2012 reference equations.9 To avoid over-diagnosing elderly patients and under-diagnosing younger patients, interpretation of the recordings used the lower limits of the normal reference range specific to each patient rather than a fixed cut-off value, which does not take into account age, height, gender or ethnicity. The severity of obstruction, if present, was graded using the percent-predicted forced expiratory volume in 1 s. To reduce inter-interpreter variability, an agreed reporting structure was established, including standard phrases. The interpretation also included advice on further investigation and management strategies, with links to specialist respiratory medical and laboratory services and a clinical guidance website, HealthPathways (http://www.cdhb.health.nz/Hospitals-Services/Health-Professionals/Pages/Health-Pathways.aspx).
Quality assurance
The Respiratory Physiology Laboratory and a Community Respiratory Physician were jointly responsible for monitoring the quality of the spirometry tests, and interpretation of the results. Initially, it was planned that each contracted general practice would have their first 100 tests re-read by the respiratory scientists, with further checks made on a periodic basis. However, this was subsequently changed to every test being scrutinised for quality using a web-based quality assurance tool. Four categories of testing quality were assessed, based on the 2005 ATS/ERS spirometry guidelines: acceptability, repeatability, technical comments and interpretation. The quality target for each practitioner was to achieve 90% in all four categories.
A clinical information system (Éclair, Sysmex New Zealand Ltd, Auckland, New Zealand) was used to store and make the test results available to referring practices. This allows clinicians to view the test results on an electronic shared care record view (e-SCRV) of the medical record.
Referral criteria for community-based spirometry
The referral criteria for funded community spirometry were defined by the original work group and listed on HealthPathways (http://www.cdhb.health.nz/Hospitals-Services/Health-Professionals/Pages/Health-Pathways.aspx). These included the following conditions in patients older than 15 years: suspected COPD; monitoring of COPD severity; chronic cough; breathlessness where a respiratory cause was being considered; and asthma. Children requiring spirometry were referred to the Christchurch Hospital Physiology Laboratory.
At the start of the programme, referral for community-based spirometry was paper based. From 2010 onwards, referrals were entered into an electronic request management system. This allowed direct transfer of the referral to the provider practice, rather than going through a central administrative point. The electronic request management system also allows easier audit and quality assurance.
Governance of the service
Funding for the service was provided by the Canterbury District Health Board. This involved contracting general practices to provide fully subsidised spirometry services. Training of practice nurses was funded by individual practices, who also purchased the required spirometer, whereas quality assurance software and calibration syringes were funded by the Canterbury District Health Board. General practitioners were funded to attend training courses for test interpretation and reporting. A community respiratory physician was appointed, with one of their responsibilities being to oversee the spirometry service and provide medical and spirometry interpretation guidance to approved provider practices. The Canterbury District Health Board funded this position (half-day weekly) and an additional 2-day weekly position for quality oversight and support by respiratory scientists.
An Integrated Respiratory Service Governance Group was convened to monitor the service, including service specifications and key performance indicators, and if necessary update the service.
Impact of the community-based spirometry service
Since its establishment in 2008, a total of 10 practices have become approved providers, with six mobile respiratory nurses providing testing at various locations in the community. The details of testing at the first six approved practices are summarised in Table 1. In 2013, only 5 of the 10 certified practice nurses performed the annual target of 100 spirometry tests, considered necessary to retain optimal testing quality. The five nurses not reaching 100 tests per annum included rural practices (two) with a small population, or were secondary testers within an urban practice.
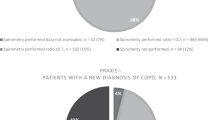
There has been a gradual increase in total spirometry testing in community settings. In February 2011, there was a decrease in referrals, owing to the Christchurch earthquakes (Figure 1). After 2013, the number of tests undertaken by community respiratory nurses decreased, whereas tests undertaken in general practice increased. This was owing to a decision to increase Approved Provider practice numbers, to free up community respiratory nurse resources for other projects. The ratio of patients referred by another general practitioner for community-based spirometry to those who were registered patients of the approved provider was ~2:1 (64 vs. 36%). Overall, the number of spirometry tests provided in Canterbury has increased.
An audit of timelines from referral to completion, undertaken in the first quarter of 2014, showed that the median time from referral to the spirometry report was 25 days (range 2–107 days). The main driver for the time taken to complete the test was the time required to organise the test appointment and to undertake spirometry. The median time from referral to test date was 22 days (range 0–105 days). Table 2 shows the specific indications for referral in a subset of patients and a summary of the results of the spirometry tests carried out in the community since introduction of the service. Spirometric abnormalities were identified in 53% of the tests performed. In addition, 7% of patients with normal spirometry and 24% of patients with a restrictive pattern had a significant bronchodilator response (>200 ml and ⩾12% increase in either forced expiratory volume in 1 s or forced vital capacity).
After the introduction of the community spirometry service, test quality reached levels similar to those seen in the Respiratory Physiology Laboratory following five sessions of feedback from respiratory scientists to the practice nurses.10 Repeatability, technical comments and interpretive pattern all reached >90%, and acceptability reached 86% after five feedback sessions.
Overview
This paper describes the establishment of a community-based, laboratory-standard spirometry service, delivered predominantly by approved general practices. This service delivered 5,409 spirometry tests in community settings between 2009 and 2013 that fulfilled the ATS/ERS quality criteria. All aspects of the delivery of the test, including interpretation and reporting, are undertaken by clinicians based in primary care. This has led to the situation where general practitioners have the confidence and support to refer patients for investigations to other general practices.
A number of factors have been critical to this process. These include hospital clinician and laboratory support, especially around interpretation and quality assurance. In addition, there needs to be a comprehensive training programme with ongoing support, centralised quality assurance, appropriate funding, a clear governance structure and close integration between primary and secondary care clinicians, as well as the central respiratory physiology laboratory. Central to the success of this programme was the insistence on the delivery of laboratory-quality spirometry testing. Inadequate quality of lung function testing is a common cause of misclassification of respiratory disease.3
The programme has evolved to become almost paper free, using web-based resources that were developed in Canterbury as part of the Canterbury Initiative (http://www.canterburyinitiative.org.nz/).11 These include web-based clinical (HealthPathways (http://www.cdhb.health.nz/Hospitals-Services/Health-Professionals/Pages/Health-Pathways.aspx)) and patient advice portals (HealthInfo (www.healthinfo.org.nz/)), an electronic request management system, a web-based interpretation and reporting tool, sending the results automatically to the referring doctor, electronic claiming for tests and uploading to the central electronic medical record.
The tests were not delivered more quickly than the service provided by the Respiratory Physiology Laboratory. However, the service was more convenient to patients, and the agreed time scales for testing were deemed suitable by the spirometry working group for the investigation of chronic disease. More acute lung function testing remained the responsibility of the central laboratory.
A fundamental component of this service was the relationships between general practice, hospital clinicians, management and funders. This service would not have been created without active enthusiastic participation of all parties, working to agreed values and philosophies within supportive governance structures.11
The ongoing resources required for quality assurance and training and the sustainability of skills in individual practices are a potential cause for concern. The loss of a small number of staff from an individual practice can lead to geographical gaps in service delivery that are not easily covered.
We are unable to demonstrate definitively that the establishment of this service has improved the outcomes for patients with respiratory disease in Canterbury. This is because the service was started at the same time as a number of other initiatives aimed at improving care of patients with chronic diseases including COPD. However, we are now providing more spirometry tests for the target population in Canterbury.
Conclusion
In conclusion, we have shown that laboratory-quality spirometry can be carried out in the community when the service is supported by specialist respiratory clinicians and certified respiratory scientists and includes centralised quality assurance monitoring and a certified ongoing training programme.
References
Global initiative for chronic obstructive lung disease (GOLD). Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (updated 2013). Available at http://www.goldcopd.org/uploads/users/files/GOLD_Report_2013_Feb20.pdf.
Jones RC, Dickson-Spillmann M, Mather MJ, Marks D, Shhakell BS . Accuracy of diagnostic registers and management of chronic obstructive pulmonary disease: the Devon primary care audit. Respir Res 2008; 9: 62.
Enright P . The use and abuse of office spirometry. Prim Care Respir J 2008; 17: 238–242.
Statistics New Zealand 2013 New Zealand Census. Available at http://www.stats.govt.nz/Census/2013-census/profile-and-summary-reports.aspx.
Miller MR, Hankinson J, Brusaco V, Burgos F, Casaburi R, Coates A et al. Standardisation of spirometry. Eur Respir J 2005; 26: 319–338.
Levy ML, Quanjer PH, Booker R, Cooper BG, Holmes S . Diagnostic spirometry in primary care. Proposed standards for general practice compliant with American Thoracic Society and European Respiratory Society recommendations. Prim Care Respir J 2009; 18: 130–147.
Derom E, van Weel C, Liistro G, Buffels J, Schermer T, Lammers E et al. Primary care spirometry. Eur Respir J 2008; 31: 197–203.
Hankinson JL, Odencrantz JR, Fedan KB . Spirometric reference values from a sample of the general U.S. population. Am J Resp Crit Care Med 1999; 159: 179–187.
Quanjer PH, Stanojevic S, Cole TJ, Baur X, Hall GL, Culver BH et al. Multi-ethnic reference values for spirometry for the 3–95-yr age range: the global lung function 2012 equations. Eur Respir J 2012; 40: 1324–1343.
Swanney MP, Stanton JD . Acceptable community spirometry quality can be achieved, but at a cost. Am J Respir Crit Care Med 2011; 183: A3130.
Timmins N, Ham C . The Quest for Integrated Health and Social Care: a Case Study in Canterbury. The King’s Fund: Canterbury, New Zealand, 2013. Available at http://www.kingsfund.org.uk/publications/quest-integrated-health-and-social-care.
Acknowledgements
The authors acknowledge the contribution of Dr Roland Meyer and Associate Professor Lutz Beckert in the development of the spirometry service and our gratitude to Dr Nikki Elliot, Canterbury Initiative, and Deborah Callaghan and Paul Bridgford, Pegasus Health, Christchurch for their assistance in the preparation of this paper.
Author information
Authors and Affiliations
Contributions
MJE was the respiratory physician. JDS was the respiratory scientist. GRBMcG was the general practitioner and project clinical leader. BIS was the research scientist. MPS was the scientific director. All the authors were involved in the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Epton, M., Stanton, J., McGeoch, G. et al. The development of a community-based spirometry service in the Canterbury region of New Zealand: observations on new service delivery. npj Prim Care Resp Med 25, 15003 (2015). https://doi.org/10.1038/npjpcrm.2015.3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/npjpcrm.2015.3
This article is cited by
-
Effects of the Spirometry Learning Module on the knowledge, confidence, and experience of spirometry operators
npj Primary Care Respiratory Medicine (2019)
-
Development and outcomes of a primary care-based sleep assessment service in Canterbury, New Zealand
npj Primary Care Respiratory Medicine (2017)