Abstract
Background:
Spirometry is the gold standard for diagnosing chronic obstructive pulmonary disease (COPD). Although there are a number of different guideline criteria for deciding who should be selected for spirometric screening, to date it is not known which criteria are the best based on sensitivity and specificity.
Aims:
Firstly, to evaluate the proportion of subjects in the PLATINO Study that would be recommended for spirometry testing according to Global initiative for Obstructive Lung Disease (GOLD)-modified, American College of Chest Physicians (ACCP), National Lung Health Education Program (NLHEP), GOLD and American Thoracic Society/European Respiratory Society (ATS/ERS) criteria. Secondly, we aimed to compare the sensitivity, specificity, and positive predictive and negative predictive values, of these five different criteria.
Methods:
Data from the PLATINO study included information on respiratory symptoms, smoking and previous spirometry testing. The GOLD-modified spirometry indication criteria are based on three positive answers out of five questions: the presence of cough, phlegm in the morning, dyspnoea, age over 40 years and smoking status.
Results:
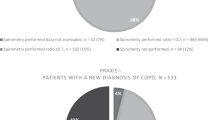
Data from 5,315 subjects were reviewed. Fewer people had an indication for spirometry (41.3%) according to the GOLD-modified criteria, and more people had an indication for spirometry (80.4%) by the GOLD and ATS/ERS criteria. A low percentage had previously had spirometry performed: GOLD-modified (14.5%); ACCP (13.2%); NLHEP (12.6%); and GOLD and ATS/ERS (12.3%). The GOLD-modified criteria showed the least sensitivity (54.9) and the highest specificity (61.0) for detecting COPD, whereas GOLD and ATS/ERS criteria showed the highest sensitivity (87.9) and the least specificity (20.8).
Conclusion:
There is a considerable difference in the indication for spirometry according to the five different guideline criteria. The GOLD-modified criteria recruit less people with the greatest sum of sensitivity and specificity.
Similar content being viewed by others
Introduction
Chronic obstructive pulmonary disease (COPD) is a preventable and treatable disease with some significant extra-pulmonary effects that may contribute to the severity of the disease. It is characterised by persistent airflow limitation. The obstruction is usually progressive and is associated with an increased chronic inflammatory response to noxious particles or gases in the lung.1,2
Early diagnosis of COPD is based on symptoms, identification of risk factors and spirometry.1 The American College of Chest Physicians (ACCP)3 recommends spirometry only for patients with respiratory symptoms, especially dyspnoea, and states that spirometry should not be performed in asymptomatic individuals, even in those with known risk factors for COPD. The National Lung Health Education Program (NLHEP)4 recommends that spirometry should be considered in individuals over 45 years old with a smoking history, or at any age in individuals with respiratory symptoms. The Global initiative for Obstructive Lung Disease (GOLD)1 guideline recommends spirometry in individuals over 40 years old who have been exposed to tobacco, with a family history of chronic lung disease and/or any respiratory symptom. The American Thoracic Society and European Respiratory Society (ATS/ERS) guideline2 recommends spirometry in any individual with respiratory symptoms and/or previous exposure to risk factors for the disease.
However, only considering symptoms, or age above 40, or a smoking history, as indicators for spirometry is likely to prove a poor cost-effective strategy for population screening for COPD. Post-nasal drip, asthma and gastroesophageal reflux are the most common causes of chronic cough in immunocompetent non-smoking individuals.5 Chronic cough may affect up to 40% of the population.6 Nasal blockage, nasal secretion, sneezing, asthma, cold air sensitivity and aspirin intolerance are also related to cough.7 In addition, productive cough is one of the main symptoms reported by patients with bronchiectasis.8 Dyspnoea, another common respiratory symptom, varies based on behavioural and physiologic responses. The main causes of dyspnoea are asthma, heart failure, myocardial ischaemia, COPD and interstitial lung disease.9 Dyspnoea is also a symptom of anxiety, especially in individuals with panic disorder or hyperventilation syndrome.10 According to the PLATINO study, the prevalence of smokers and ex-smokers aged 40 years or more was 57% in São Paulo, 67% in Santiago, 44% in Mexico City, 57% in Montevideo and 58.4% in Caracas.11 Similar results were found in London, where 44.8% of the population above 35 years of age were smokers or ex-smokers.12
The adoption of any of the above four guideline criteria seems likely to be too non-specific, leading to a large number of unnecessary spirometries. Our hypothesis is that the adoption of more rigid criteria for spirometry could increase specificity, decrease the number of unnecessary spirometries and make spirometry screening more cost-effective. Based on modification of the GOLD criteria for spirometry, we believe that spirometry would be more specific if performed in individuals with at least three positive answers out of the five following questions: ‘Do you cough most days?’; ‘Do you bring up phlegm in the morning?’; ‘Do you walk at a slower pace than other people of the same age?’; ‘Are you older than 40 years?’; and ‘Are you a smoker or ex-smoker?’. These are our ‘GOLD-modified’ criteria.
The objective of this study, therefore, was to evaluate the proportion of individuals from the PLATINO study that met the criteria for spirometry assessment based on GOLD-modified, ACCP, NLHEP, GOLD and ATS/ERS guideline criteria, and to evaluate the applicability of each of these guideline criteria for the diagnosis of COPD as defined by a forced expiratory volume in 1 s/forced vital capacity (FEV1/FVC) ratio below 0.7.
Materials and methods
Study design
This study was based on data from the Latin American Project for the Investigation of Obstructive Lung Disease (PLATINO).11
The inclusion criteria were males and females aged 40 years or more living in selected households of the metropolitan areas of São Paulo (Brazil), Santiago (Chile), Mexico City (Mexico), Montevideo (Uruguay) and Caracas (Venezuela). COPD was defined as an FEV1/FVC ratio below 0.70. The exclusion criteria were: institutionalised individuals; history of mental illness; thoracic, abdominal or ophthalmic surgery in the past 3 months; angina or myocardial infarction in the past 3 months; diagnosis of active tuberculosis; pregnancy; and acute respiratory infection within 3 weeks before the interview.
Respiratory symptoms, smoking and previous spirometry assessment were evaluated using a questionnaire based on the following questionnaires: American Thoracic Society Division of Lung Diseases (ATS/DLD);13 European Community Respiratory Health Survey II (ECRHS II);14 Lung Health Study (LHS);15 and Short Form 12.16
Equipment and techniques
Spirometry (Easy One; NDD; Medical Technologies, Switzerland) was performed according to ATS guidelines17 and it was repeated after 15 min of bronchodilator inhalation (salbutamol, 200 µg). The criteria for COPD diagnosis was based on a post-bronchodilator FEV1/FVC ratio <0.70 (GOLD).1 The criteria for spirometry are shown in Table 1.
Statistical analysis
For the descriptive analysis, numerical data are shown as mean and standard deviation. The categorical variables are presented in absolute value and percentage. The t-test was used to compare numerical variables between two independent groups. Categorical variables' proportions between independent groups were compared using the Chi-squared test. Analysis of variance was used for comparison of Latin American Cities. P values <0.05 were considered statistically significant.
Results
Demographic data
From a total of 6,711 eligible subjects in all locations, 5,571 individuals completed questionnaires and 5,315 spirometry tests were obtained. The mean age was 56.3±11.7 years. The proportion of women was higher (mean 60.5% of the total) and the prevalence of smokers in the population was 29.8% (Table 2). The COPD prevalence in Latin America was 14.3%.
Indication for spirometry assessment
According to our GOLD-modified criteria, 2,195 (41.3%) individuals fulfilled the indication criteria for spirometry. There were 3,013 individuals (56.7%) who met the spirometry indication criteria according to ACCP, 4,010 individuals (75.5%) according to NLHEP and 4,273 (80.4%) according to GOLD and ATS/ERS (Table 3). Table 4 shows the prevalence of spirometry indication in each of the five cities.
Indication of spirometry for the diagnosis of COPD
In order to identify the most effective criteria for the screening of COPD, we compared the number of individuals who met the criteria for spirometry assessment according to GOLD-modified, ACCP, NHLEP, GOLD and ATS/ERS and the presence or absence of COPD diagnosis (Table 5). GOLD-modified had the lowest number of individuals with indication for spirometry (41.3%). On the contrary, GOLD and ATS/ERS criteria had the highest indication for spirometry (80.4%).
The sensitivity, specificity, positive predictive (PPV) and negative predictive values (NPV) for the COPD screening criteria are presented in Table 6. The GOLD-modified criteria showed the lowest sensitivity (54.9%)—i.e., the subjects with COPD that were correctly identified, and the highest specificity (61%)—i.e. the number of individuals without COPD who were identified correctly. GOLD and ATS/ERS criteria showed the highest sensitivity (87.9%) and the lowest specificity (20.8%).
The PPV and NPV analysis showed a small variability among the different criteria. The PPV varied from 15.6% for the GOLD and ATS/ERS criteria to 19% for the GOLD-modified; the NPV varied from 89% for the GOLD-modified to 91.2% for GOLD and ATS/ERS criteria. Since NLHEP, GOLD and ATS/ERS criteria for spirometry screening are very similar, with the only difference being the age cut-off, the values for sensitivity, specificity, PPV and NPV were also similar.
Discussion
Main findings
Considering the high mortality and treatment costs of COPD, the disease should be diagnosed in its early phases. Risk factors such as tobacco smoking, exposure to air pollution (burning of wood), a family history of COPD, or a history of recurrent respiratory infections during childhood,18 should highlight the need for screening and early intervention.19 Undiagnosed COPD is a common problem and is usually associated with poor quality of life and poor functional status.20 Spirometry is still the gold standard for the diagnosis of COPD and it should be performed in all individuals with clinical signs of the disease.1 Despite the benefits from early diagnosis, the results of this study show that spirometry assessment is underused in Latin America.
Use of our GOLD-modified criteria (which consider the presence of respiratory symptoms and risk factors) as an indication for COPD screening was shown to be the most cost-effective method, as it produced the lowest number of individuals requiring evaluation and provided the highest proportion of COPD diagnoses.
Interpretation of findings in relation to previously published work
Our results come from the database of the PLATINO study, which was a cross-sectional, population-based study designed to evaluate the prevalence and impact of COPD in the metropolitan areas of five large cities in Latin America.11 However, the study also offers the opportunity to evaluate the use of different criteria for the early diagnosis of COPD and the relevance of these criteria. For early diagnosis, it is crucial to consider the cost-effectiveness of,21 and the best criteria for,22 screening of individual subjects.
Despite world guidance, only 12–14% of individuals who met the GOLD-modified, ACCP, NLHEP, GOLD and ATS/ERS criteria had had spirometry assessment in the past. Our data confirm that spirometry is underused. As the presence of at least one symptom is recommended in the GOLD-modified and ACCP criteria, the absence of symptoms could be an explanation for the low rates of spirometry assessment according to these criteria.23 Usually, individuals believe that respiratory symptoms such as chronic cough and sputum production are not important, and typically they have been neglected by doctors for years.23 However, even considering the more comprehensive criteria of NLHEP, GOLD and ATS/ERS, which do not require the presence of respiratory symptoms, the use of spirometry was low. According to GOLD-modified and ACCP criteria, 41.3 and 56.7% of the study population, respectively, had an indication for spirometry testing, whereas the recommendation was higher with NLHEP and GOLD/ATS/ERS—i.e., 75.5 and 80.4%, respectively.
The underutilisation of spirometry is not an isolated fact in Latin America. In 1997, the epidemiological study IBERPOC (Estudio Epidemiológico de EPOC en España)24 showed that only 16.5% of diagnosed COPD patients had had previous spirometry. However, a decade later, the EPI-SCAN study (Epidemiologic Study of COPD in Spain)25 demonstrated that 58.5% of COPD patients had had spirometry performed. An American study in primary care showed that in a group of individuals identified as having potential risk factors for COPD according to the GOLD and ATS/ERS criteria, only 50% had had previous spirometry.26 Even in developed countries, the criteria for spirometry assessment are not followed, and the criteria do not receive the attention needed for the diagnosis of COPD. In Italy, a survey evaluating the use of spirometry for COPD diagnosis among clinicians showed that 30% of doctors do not use it.27 In Japan, a sample of general and specialist physicians were provided with clinical cases and a survey regarding diagnosis and treatment of COPD; the results showed that 81.9% of physicians recommended a chest X-ray, 49.1% spirometry and 17.7% a computed tomography scan for the diagnosis of COPD.28
According to Soriano et al.,21 COPD could be prevented on three levels: primary prevention, with modification or reduction of risk factors, specially smoking; secondary prevention, with screening and early detection of COPD; and tertiary prevention, with the purpose of improving health status, decreasing or slowing disease progression and reducing exacerbation rate. Spirometry has an important role at all three preventive levels. The DIDASCO study from Belgium showed a high accuracy with the use of a portable spirometer by trained general practitioners and they were able to incorporate spirometry into their clinical routine.29
Among the criteria considered in our study, our GOLD-modified criteria showed the highest sum of sensitivity and specificity, whereas the GOLD and ATS/ERS criteria had the lowest sensitivity and specificity sum. This is an important finding, as the GOLD criteria have been used worldwide for the diagnosis and treatment of COPD patients. Isolated respiratory symptoms are a poor indicator for COPD30—for example, one-third of the USA population presents respiratory symptoms.31 A cohort study of 3,955 individuals in the USA showed that 83% reported at least one respiratory symptom and 65% reported multiple respiratory symptoms, and among the 2,917 smokers, 86% reported at least one symptom and 70% had multiple symptoms.32 Another study in Spain showed a low specificity for respiratory symptoms: 8.6% of individuals with chronic cough, 6.2% with dyspnoea and 30.7% with wheezing had normal spirometry, and among individuals with restrictive disease 10.6% had cough, 17.2% had dyspnoea and 45.5% had wheezing.33
Strengths and limitations of this study
Our study has some limitations. We used an FEV1/FVC ratio below 0.7 to define COPD, which decreases with age due to loss of lung elasticity and recoil, resulting in overdiagnosis of COPD in the elderly population.34 Nonetheless, this definition is currently the most accepted one owing to its practicality and it has been used in the majority of studies related to COPD prevalence published lately.11,25 Another limitation was the utilisation of a self-reported questionnaire for assessment of symptoms. The ideal would be a more detailed and individualised evaluation as most individuals in our population were lay people and they may not comprehend the definition of symptoms.
Conclusions and implications for future research, policy and practice
We conclude that criteria for spirometry screening for COPD that only consider age or isolated symptoms have low specificity and may require a higher number of spirometries to be performed. Our GOLD-modified criteria may recruit a smaller proportion of individuals for spirometry but they are the criteria with the largest sum of sensitivity and specificity and the largest proportion of COPD patients diagnosed. In addition to incentives for spirometry performance in primary care practice, the insufficient use of spirometric testing for COPD detection shown in this study suggests the need to implement more specific educational programmes. Aditionally, it is necessary to increase the availability of spirometry tests in primary care settings in order to increase the chances of identifying COPD patients.
References
Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management and Prevention of COPD 2011. Available at: http://www.goldcopd.org/uploads/users/files/GOLD_Report_2011_Feb21.pdf
Celli BR, MacNee W, Force AET . Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur Respir J 2004; 23: 932–946.
Qaseem A, Snow V, Shekelle P, Sherif K, Wilt TJ, Weinberger S et al. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline from the American College of Physicians. Ann Intern Med 2007; 147: 633–638.
The National Lung Health Education Program Executive Committee. Strategies in preserving lung health and preventing COPD and associated diseases. The National Lung Health Education Program (NLHEP). Chest 1998; 113: 123S–163SS.
Natt RS, Earis JE, Swift AC . Chronic cough: a multidisciplinary approach. J Laryngol Otol. 2012; 126: 441–444.
Morice AH, Kastelik JA . Cough. 1: chronic cough in adults. Thorax 2003; 58: 901–907.
Bende M, Millqvist E . Prevalence of chronic cough in relation to upper and lower airway symptoms; the Skovde population-based study. Front Physiol 2012; 3: 251.
King PT, Holdsworth SR, Farmer M, Freezer N, Villanueva E, Holmes PW . Phenotypes of adult bronchiectasis: onset of productive cough in childhood and adulthood. COPD 2009; 6: 130–136.
Wahls SA . Causes and evaluation of chronic dyspnea. Am Fam Physician 2012; 86: 173–182.
Smoller JW, Pollack MH, Otto MW, Rosenbaum JF, Kradin RL . Panic anxiety, dyspnea, and respiratory disease. Theoretical and clinical considerations. Am J Respir Crit Care Med 1996; 154: 6–17.
Menezes AM, Perez-Padilla R, Jardim JR, Muino A, Lopez MV, Valdivia G et al. Chronic obstructive pulmonary disease in five Latin American cities (the PLATINO study): a prevalence study. Lancet 2005; 366: 1875–1881.
Shahab L, Jarvis MJ, Britton J, West R . Prevalence, diagnosis and relation to tobacco dependence of chronic obstructive pulmonary disease in a nationally representative population sample. Thorax 2006; 61: 1043–1047.
Ferris BG . Epidemiology Standardization Project (American Thoracic Society). Am Rev Respir Dis 1978; 118: 1–120.
European Community Respiratory Health Survey IISC. The European Community Respiratory Health Survey II. Eur Respir J 2002; 20: 1071–1079.
BC Cancer Research Centre. Lung Health Study Questionnaire. BC Cancer Research Centre: Vancouver, 2004.
Ware JE, Kosinski M, Keller SD . How to Score the SF12 Physical and Mental Health Summary Scales. The Health Institute, New England Medical Center: Boston, 1995.
American Thoracic Society. Standardization of Spirometry, 1994 Update. Am J Respir Crit Care Med 1995; 152: 1107–1136.
Bourbeau J, van der Palen J . Promoting effective self-management programmes to improve COPD. Eur Respir J 2009; 33: 461–463.
Hill K, Goldstein RS, Guyatt GH, Blouin M, Tan WC, Davis LL et al. Prevalence and underdiagnosis of chronic obstructive pulmonary disease among patients at risk in primary care. CMAJ 2010; 182: 673–678.
Dales RE, Vandemheen KL, Clinch J, Aaron SD . Spirometry in the primary care setting: influence on clinical diagnosis and management of airflow obstruction. Chest 2005; 128: 2443–2447.
Soriano JB, Zielinski J, Price D . Screening for and early detection of chronic obstructive pulmonary disease. Lancet 2009; 374: 721–732.
Enright PL, Crapo RO . Controversies in the use of spirometry for early recognition and diagnosis of chronic obstructive pulmonary disease in cigarette smokers. Clin Chest Med 2000; 21: 645–652.
Nascimento OA, Camelier A, Rosa FW, Menezes AM, Perez-Padilla R, Jardim JR et al. Chronic obstructive pulmonary disease is underdiagnosed and undertreated in Sao Paulo (Brazil): results of the PLATINO study. Braz J Med Biol Res 2007; 40: 887–895.
Pena VS, Miravitlles M, Gabriel R, Jimenez-Ruiz CA, Villasante C, Masa JF et al. Geographic variations in prevalence and underdiagnosis of COPD: results of the IBERPOC multicentre epidemiological study. Chest 2000; 118: 981–989.
Ancochea J, Badiola C, Duran-Tauleria E, Garcia Rio F, Miravitlles M, Munoz L et al. [The EPI-SCAN survey to assess the prevalence of chronic obstructive pulmonary disease in Spanish 40-to-80-year-olds: protocol summary]. Arch Bronconeumol 2009; 45: 41–47.
Joo MJ, Au DH, Fitzgibbon ML, McKell J, Lee TA . Determinants of spirometry use and accuracy of COPD diagnosis in primary care. J Gen Intern Med 2011; 26: 1272–1277.
Caramori G, Bettoncelli G, Tosatto R, Arpinelli F, Visona G, Invernizzi G et al. Underuse of spirometry by general practitioners for the diagnosis of COPD in Italy. Monaldi Arch Chest Dis 2005; 63: 6–12.
Fukuhara S, Nishimura M, Nordyke RJ, Zaher CA, Peabody JW . Patterns of care for COPD by Japanese physicians. Respirology 2005; 10: 341–348.
Buffels J, Degryse J, Heyrman J, Decramer M, Study D . Office spirometry significantly improves early detection of COPD in general practice: the DIDASCO Study. Chest 2004; 125: 1394–1399.
Medbo A, Melbye H . What role may symptoms play in the diagnosis of airflow limitation? A study in an elderly population. Scand J Prim Health Care 2008; 26: 92–98.
Wilt TJ, Niewoehner D, Kim C, Kane RL, Linabery A, Tacklind J et al. Use of spirometry for case finding, diagnosis, and management of chronic obstructive pulmonary disease (COPD). Summary, Evidence Report/Technology Assessment: Number 121. AHRQ Publication Number 05-E017-1. Agency for Healthcare Research and Quality: Rockville, MD, USA. http://www.ahrq.gov/clinic/epcsums/spirosum.htm. August 2005.
Ohar JA, Sadeghnejad A, Meyers DA, Donohue JF, Bleecker ER . Do symptoms predict COPD in smokers? Chest 2010; 137: 1345–1353.
Soriano JB, Miravitlles M, Garcia-Rio F, Munoz L, Sanchez G, Sobradillo V et al. Spirometrically-defined restrictive ventilatory defect: population variability and individual determinants. Prim Care Respir J 2012; 21: 187–193.
Hardie JA, Buist AS, Vollmer WM, Ellingsen I, Bakke PS, Morkve O . Risk of over-diagnosis of COPD in asymptomatic elderly never-smokers. Eur Respir J 2002; 20: 1117–1122.
Acknowledgements
We acknowledge Carolina Aguiar PT for the translation and editing of the text.
Author information
Authors and Affiliations
Consortia
Contributions
All authors participared in designing the Study and reviewed the text. Other functions: Ana M Menezes, coordinator of PLATINO Study; Rogelio Perez-Padilla, rewier of spirometries and quality control; Adriana Muiño and Maria Victorina, investigators in Montevideu; Valdivia and Carmen Lisboa, investigators in Santiago; Oliver A Nascimento and José R Jardim, investigators in São Paulo.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Luize, A., Menezes, A., Perez-Padilla, R. et al. Assessment of five different guideline indication criteria for spirometry, including modified GOLD criteria, in order to detect COPD: data from 5,315 subjects in the PLATINO study. npj Prim Care Resp Med 24, 14075 (2014). https://doi.org/10.1038/npjpcrm.2014.75
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/npjpcrm.2014.75
This article is cited by
-
Respiratory illness and air pollution from the steel industry: the case of Piquiá de Baixo, Brazil (Preliminary report)
Multidisciplinary Respiratory Medicine (2016)
-
Working in harmony with Nature: highlights from 2014, and a look to the future
npj Primary Care Respiratory Medicine (2015)