Abstract
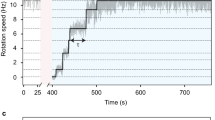
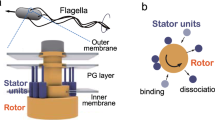
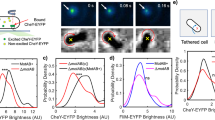
The switching mechanism of the flagellar motor provides the basis for the motile behaviour of flagellated bacteria. Its highly sensitive response has previously been understood in terms of equilibrium models, either the classical two-state concerted allosteric model, or more generally, the Ising-type conformation spread model. Here, we systematically study motor switching under various load conditions from high to zero load, under different proton motive force (pmf) conditions and varying the number of torque-generating units (stators). In doing so, we reveal the signature of a non-equilibrium effect. To consistently account for the motor-switching dependence on each those conditions, a previously neglected non-equilibrium effect—the energy input from the motor torque—has to be incorporated into models of the flagellar switch. We further show that this effect increases the sensitivity of the flagellar switch. Exploiting a very small fraction of the energy expense of the flagellar motor for functional regulation increases its sensitivity greatly. Similar mechanisms are expected to be found in other protein complexes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Berg, H. C. The rotary motor of bacterial flagella. Annu. Rev. Biochem. 72, 19–54 (2003).
Turner, L., Ryu, W. & Berg, H. C. Real-time imaging of fluorescent flagellar filaments. J. Bacteriol. 182, 2793–2801 (2000).
Welch, M., Oosawa, K., Aizawa, S.-I. & Eisenbach, M. Phosphorylation-dependent binding of a signal molecule to the flagellar switch of bacteria. Proc. Natl Acad. Sci. USA 90, 8787–8791 (1993).
Cluzel, P., Surette, M. & Leibler, S. An ultrasensitive bacterial motor revealed by monitoring signaling proteins in single cells. Science 287, 1652–1655 (2000).
Sourjik, V. & Berg, H. C. Binding of the Escherichia coli response regulator CheY to its target measured in vivo by fluorescence resonance energy transfer. Proc. Natl Acad. Sci. USA 99, 12669–12674 (2002).
Silverman, M. & Simon, M. Flagellar rotation and the mechanism of bacterial motility. Nature 249, 73–74 (1974).
Ryu, W. S., Berry, R. M. & Berg, H. C. Torque-generating units of the flagellar motor of Escherichia coli have a high duty ratio. Nature 403, 444–447 (2000).
Bai, F. et al. Conformational spread as a mechanism for cooperativity in the bacterial flagellar switch. Science 327, 685–689 (2010).
Korobkova, E., Emonet, T., Vilar, J. M. G., Shimizu, T. S. & Cluzel, P. From molecular noise to behavioral variability in a single bacterium. Nature 428, 574–578 (2004).
Korobkova, E. A., Emonet, T., Park, H. & Cluzel, P. Hidden stochastic nature of a single bacterial motor. Phys. Rev. Lett. 96, 058105 (2006).
Wang, F., Yuan, J. & Berg, H. C. Switching dynamics of the bacterial flagellar motor near zero load. Proc. Natl Acad. Sci. USA 111, 15752–15755 (2014).
Block, S. M., Segall, J. E. & Berg, H. C. Adaptation kinetics in bacterial chemotaxis. J. Bacteriol. 154, 312–323 (1983).
Tu, Y. The nonequilibrium mechanism for ultrasensitivity in a biological switch: sensing by Maxwell’s demons. Proc. Natl Acad. Sci. USA 105, 11737–11741 (2008).
Scharf, B. E., Fahrner, K. A., Turner, L. & Berg, H. C. Control of direction of flagellar rotation in bacterial chemotaxis. Proc. Natl Acad. Sci. USA 95, 201–206 (1998).
van Albada, S. B., Tanase-Nicola, S. & ten Wolde, P. R. The switching dynamics of the bacterial flagellar motor. Mol. Syst. Biol. 5, 316 (2009).
Yuan, J. & Berg, H. C. Resurrection of the flagellar motor near zero load. Proc. Natl Acad. Sci. USA 105, 1182–1185 (2008).
Yuan, J., Fahrner, K. A. & Berg, H. C. Switching of the bacterial flagellar motor near zero load. J. Mol. Biol. 390, 394–400 (2009).
Yuan, J. & Berg, H. C. Thermal and solvent-isotope effects on the flagellar rotary motor near zero load. Biophys. J. 98, 2121–2126 (2010).
Walter, J. M., Greenfield, D., Bustamante, C. & Liphardt, J. Light-powering Escherichia coli with proteorhodopsin. Proc. Natl Acad. Sci. USA 104, 2408–2412 (2007).
Blair, D. F. & Berg, H. C. Restoration of torque in defective flagellar motors. Science 242, 1678–1681 (1988).
Tipping, M. J., Steel, B. C., Delalez, N. J., Berry, R. M. & Armitage, J. P. Quantification of flagellar motor stator dynamics through in vivo proton-motive force control. Mol. Microbiol. 87, 338–347 (2013).
Lele, L. P., Hosu, B. G. & Berg, H. C. Dynamics of mechanosensing in the bacterial flagellar motor. Proc. Natl Acad. Sci. USA 110, 11839–11844 (2013).
Tipping, M. J., Delalez, N. J., Lim, R., Berry, R. M. & Armitage, J. P. Load-dependent assembly of the bacterial flagellar motor. mBio 4, e00551 (2013).
Duke, T. A. J., Le Novere, N. & Bray, D. Conformational spread in a ring of proteins: a stochastic approach to allostery. J. Mol. Biol. 308, 541–553 (2001).
Fahrner, K. A., Ryu, W. S. & Berg, H. C. Bacterial flagellar switching under load. Nature 423, 938 (2003).
Bai, F., Minamino, T., Wu, Z., Namba, K. & Xing, J. Coupling between switching regulation and torque generation in bacterial flagellar motor. Phys. Rev. Lett. 108, 178105 (2012).
Xie, L., Altindal, T., Chattopadhyay, S. & Wu, X.-L. Bacterial flagellum as a propeller and as a rudder for efficient chemotaxis. Proc. Natl Acad. Sci. USA 108, 2246–2251 (2011).
Morse, M., Bell, J., Li, G. & Tang, J. X. Flagellar motor switching in Caulobacter crescentus obeys first passage time statistics. Phys. Rev. Lett. 115, 198103 (2015).
Guzman, L.-M., Belin, D., Carson, M. J. & Beckwith, J. Tight regulation, modulation, and high-level expression by vectors containing the arabinose PBAD promoter. J. Bacteriol. 177, 4121–4130 (1995).
Acknowledgements
We thank H. Berg, J. Tang, and Y. Tu for comments. This work was supported by National Natural Science Foundation of China Grants 11374282, 21573214 (to J.Y.) and 11402265 (to R.Z.), Fundamental Research Funds for the Central Universities (WK2030020023, to J.Y.), and Anhui Natural Science Foundation Grant 1408085MA10 (to R.Z.). J.Y. and R.Z. are supported by the Chinese Government ‘1000 Youth Talent Program’.
Author information
Authors and Affiliations
Contributions
J.Y. and R.Z. planned the work; F.W., H.S. and R.H. performed the measurements and the simulation with help from R.W.; J.Y. proposed the non-equilibrium model; J.Y., R.Z. and F.W. wrote the paper with inputs from other authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 946 kb)
Rights and permissions
About this article
Cite this article
Wang, F., Shi, H., He, R. et al. Non-equilibrium effect in the allosteric regulation of the bacterial flagellar switch. Nature Phys 13, 710–714 (2017). https://doi.org/10.1038/nphys4081
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphys4081
This article is cited by
-
Size limits the sensitivity of kinetic schemes
Nature Communications (2023)
-
Mechanosensitive recruitment of stator units promotes binding of the response regulator CheY-P to the flagellar motor
Nature Communications (2021)
-
High pressure inhibits signaling protein binding to the flagellar motor and bacterial chemotaxis through enhanced hydration
Scientific Reports (2020)
-
Topology, landscapes, and biomolecular energy transport
Nature Communications (2019)
-
An early peak in ion channel research
Nature Physics (2018)