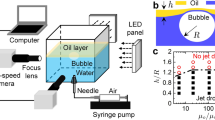
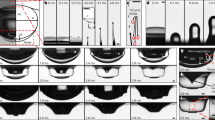
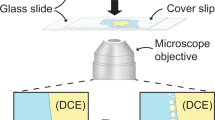
Abstract
Bursting of bubbles at an air/liquid interface is a familiar occurrence relevant to foam stability, cell cultures in bioreactors and ocean–atmosphere mass transfer. In the latter case, bubble-bursting leads to the dispersal of sea-water aerosols in the surrounding air. Here we show that bubbles bursting at a compound air/oil/water-with-surfactant interface can disperse submicrometre oil droplets in water. Dispersal results from the detachment of an oil spray from the bottom of the bubble towards water during bubble collapse. We provide evidence that droplet size is selected by physicochemical interactions between oil molecules and the surfactants rather than by hydrodynamics. We demonstrate the unrecognized role that this dispersal mechanism may play in the fate of the sea surface microlayer and of pollutant spills by dispersing petroleum in the water column. Finally, our system provides an energy-efficient route, with potential upscalability, for applications in drug delivery, food production and materials science.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Blanchard, D. C. & Woodcock, A. H. Bubble formation and modification in the sea and its meteorological significance. Tellus 9, 145–158 (1957).
Tong, M., Cole, K. & Neethling, S. J. Drainage and stability of 2D foams: Foam behaviour in vertical Hele–Shaw cells. Colloids Surf. A 382, 42–49 (2011).
Moheimani, N. R., Isdepsky, A., Lisec, J., Raes, E. & Borowitzka, M. A. Coccolithophorid algae culture in closed photobioreactors. Biotechnol. Bioeng. 108, 2078–2087 (2011).
Wu, J. Evidence of sea spray produced by bursting bubbles. Science 212, 324–326 (1981).
Schmitt-Kopplin, P. et al. Dissolved organic matter in sea spray: a transfer study from marine surface water to aerosols. Biogeosciences 9, 1571–1582 (2012).
Duchemin, L., Popinet, S., Josserand, C. & Zaleski, S. Jet formation in bubbles bursting at a free surface. Phys. Fluids 14, 3000–3008 (2002).
Bird, J. C., de Ruiter, R., Courbin, L. & Stone, H. A. Daughter bubble cascades produced by folding of ruptured thin films. Nature 465, 759–762 (2010).
Lhuissier, H. & Villermaux, E. Bursting bubble aerosols. J. Fluid Mech. 696, 5–44 (2012).
Uemura, T., Ueda, Y. & Iguchi, M. Ripples on a rising bubble through an immiscible two-liquid interface generate numerous micro droplets. Europhys. Lett. 92, 34004 (2010).
Mukerjee, P. & Mysels, K. J. Critical Micelle Concentrations of Aqueous Surfactant Systems (US National Bureau of Standards; for sale by the Supt of Docs, US Govt Print Off, 1971).
Boulton-Stone, J. M. & Blake, J. R. Gas-bubbles bursting at a free surface. J. Fluid Mech. 254, 437–466 (1993).
Eggers, J. & Villermaux, E. Physics of liquid jets. Rep. Prog. Phys. 71, 036601 (2008).
Walstra, P. Principles of emulsion formation. Chem. Eng. Sci. 48, 333–349 (1993).
Marmottant, P. H. & Villermaux, E. On spray formation. J. Fluid Mech. 498, 73–111 (2004).
Wilkinson, K. M., Bain, C. D., Matsubara, H. & Aratono, M. Wetting of surfactant solutions by alkanes. ChemPhysChem 6, 547–555 (2005).
Ash, P. A., Bain, C. D. & Matsubara, H. Wetting in oil/water/surfactant systems. Curr. Opin. Colloid Interf. 17, 196–204 (2012).
Kellay, H., Meunier, J. & Binks, B. P. Wetting properties of normal-alkanes on AOT monolayers at the brine–air interface. Phys. Rev. Lett. 69, 1220–1223 (1992).
Cheng, Y., Ye, X., Huang, X. D. & Ma, H. R. Reentrant wetting transition on surfactant solution surfaces. J. Chem. Phys. 125, 164709 (2006).
Boulton-Stone, J. M. The effect of surfactant on bursting gas-bubbles. J. Fluid Mech. 302, 231–257 (1995).
Israelachvili, J. N. Intermolecular and Surface Forces 2nd edn (Academic, 1991).
Bertrand, E. et al. First-order and critical wetting of alkanes on water. Phys. Rev. Lett. 85, 1282–1285 (2000).
Matsubara, H., Aratono, A., Wilkinson, K. M. & Bain, C. D. Lattice model for the wetting transition of alkanes on aqueous surfactant solutions. Langmuir 22, 982–988 (2006).
Liu, T. & Peter Sheng, Y. Three dimensional simulation of transport and fate of oil spill under wave induced circulation. Mar. Pollut. Bull. 80, 148–159 (2014).
Wurl, O., Wurl, E., Miller, L., Johnson, K. & Vagle, S. Formation and global distribution of sea-surface microlayers. Biogeosciences 8, 121–135 (2011).
Cunliffe, M. et al. Sea surface microlayers: A unified physicochemical and biological perspective of the air–ocean interface. Prog. Oceanogr. 109, 104–116 (2012).
Brock, C. A., Murphy, D. M., Bahreini, R. & Middlebrook, A. M. Formation and growth of organic aerosols downwind of the Deepwater Horizon oil spill. Geophys. Res. Lett. 38, L17805 (2011).
Sellegri, K., O’Dowd, C. D., Yoon, Y. J., Jennings, S. G. & de Leeuw, G. Surfactants and submicron sea spray generation. J. Geophys. Res. 111, D22215 (2006).
Arnaudov, L. N., Stoyanov, S. D. & Stuart, M. A. C. Colloid fabrication by co-extrusion. Colloids Surf. A 323, 94–98 (2008).
Talsma, H., Vansteenbergen, M. J., Borchert, J. C. H. & Crommelin, D. J. A. A novel technique for the one-step preparation of liposomes and nonionic surfactant vesicles without the use of organic solvents. Liposome formation in a continuous gas-stream: The bubble method. J. Pharm. Sci. 83, 276–280 (1994).
Wang, L. J., Dong, J. F., Chen, J., Eastoe, J. & Li, X. F. Design and optimization of a new self-nanoemulsifying drug delivery system. J. Colloid Interface Sci. 330, 443–448 (2009).
Rao, J. & McClements, D. J. Food-grade microemulsions and nanoemulsions: Role of oil phase composition on formation and stability. Food Hydrocolloids 29, 326–334 (2012).
Mason, T. G., Wilking, J. N., Meleson, K., Chang, C. B. & Graves, S. M. Nanoemulsions: Formation, structure, and physical properties. J. Phys. Condens. Matter 18, R635–R666 (2006).
Solans, C. & Sole, I. Nano-emulsions: Formation by low-energy methods. Curr. Opin. Colloid Interf. 17, 246–254 (2012).
Rosen, M. J. & Kunjappu, J. T. Surfactants and Interfacial Phenomena 4th edn (Wiley, 2012).
Matyjaszewski, K. & Davis, T. P. Handbook of Radical Polymerization 1st edn (Wiley, 2002).
Russev, S. C. & Arguirov, T. V. Rotating analyzer–fixed analyzer ellipsometer based on null type ellipsometer. Rev. Sci. Instrum. 70, 3077–3082 (1999).
Acknowledgements
We acknowledge the contribution of S. C. Russev from Department of Solid State Physics & Microelectronics, Sofia University, Bulgaria, who helped us with the interpretation of the ellipsometric data and R. D. Stanimirova from Department of Chemical Engineering, Sofia University, Bulgaria, who performed measurements in a Langmuir trough and some spreading experiments. We also acknowledge R. K. Prud’homme from Department of Chemical and Biological Engineering (Princeton University) for the use of the Malvern Zetasizer. T.D.G. and S.D.S. acknowledge the financial support of EU project FP7-REGPOT-2011-1, ‘Beyond Everest’. M.R. acknowledges D. Langevin for fruitful discussions. This research was made possible in part by the CMEDS grant from BP/The Gulf of Mexico Research Initiative.
Author information
Authors and Affiliations
Contributions
J.F., M.R., L.N.A., S.D.S. and H.A.S. conceived of and planned the experiments. J.F. executed the experimental work. J.F., M.R. and H.A.S. wrote the manuscript. J.F. and D.V. analysed the DLS data. J.F., M.R., L.N.A., S.D.S., T.D.G. and H.A.S. analysed and interpreted the experimental results. T.D.G. and G.G.T. performed ellipsometry measurements. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2276 kb)
Supplementary Movie
Supplementary Movie 1 (AVI 296 kb)
Supplementary Movie
Supplementary Movie 2 (AVI 421 kb)
Supplementary Movie
Supplementary Movie 3 (AVI 19344 kb)
Supplementary Movie
Supplementary Movie 4 (AVI 4710 kb)
Rights and permissions
About this article
Cite this article
Feng, J., Roché, M., Vigolo, D. et al. Nanoemulsions obtained via bubble-bursting at a compound interface. Nature Phys 10, 606–612 (2014). https://doi.org/10.1038/nphys3003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphys3003
This article is cited by
-
Bubble-Bursting Atomization for Minimum Quantity Lubrication Using the TRIZ Method and Computational Fluid Dynamics Simulations
Journal of Materials Engineering and Performance (2024)
-
Gas-assisted microfluidic step-emulsification for generating micron- and submicron-sized droplets
Microsystems & Nanoengineering (2023)
-
Suppression of hollow droplet rebound on super-repellent surfaces
Nature Communications (2023)
-
Enhanced singular jet formation in oil-coated bubble bursting
Nature Physics (2023)
-
The Preparation of Nanoemulsion Formulation from Pelargonic Acid in Lignin Carrier and Its Application to Weeds
Journal of The Institution of Engineers (India): Series E (2023)