Abstract
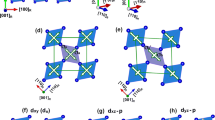
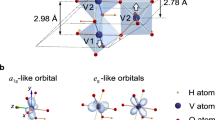
External control of the conductivity of correlated oxides is one of the most promising schemes for realizing energy-efficient electronic devices. Vanadium dioxide (VO2), an archetypal correlated oxide compound, undergoes a temperature-driven metal–insulator transition near room temperature with a concomitant change in crystal symmetry. Here, we show that the metal–insulator transition temperature of thin VO2(001) films can be changed continuously from ∼285 to ∼345 K by varying the thickness of the RuO2 buffer layer (resulting in different epitaxial strains). Using strain-, polarization- and temperature-dependent X-ray absorption spectroscopy, in combination with X-ray diffraction and electronic transport measurements, we demonstrate that the transition temperature and the structural distortion across the transition depend on the orbital occupancy in the metallic state. Our findings open up the possibility of controlling the conductivity in atomically thin VO2 layers by manipulating the orbital occupancy by, for example, heterostructural engineering.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Imada, M., Fujimori, A. & Tokura, Y. Metal–insulator transitions. Rev. Mod. Phys. 70, 1039–1263 (1998).
Bednorz, J. G. & Müller, K. A. Possible high T c superconductivity in the Ba–La–Cu–O system. Zeit. Für. Phys. 64, 189–193 (1986).
Torrance, J. B., Lacorre, P., Asavaroengchai, C. & Metzger, R. M. Why are some oxides metallic, while most are insulating? Physica C 182, 351–364 (1991).
Bocquet, A. E., Saitoh, T., Mizokawa, T. & Fujimori, A. Systematics in the electronic structure of 3d transition-metal compounds. Solid State Commun. 83, 11–15 (1992).
Jin, S. et al. Thousandfold change in resistivity in magnetoresistive La–Ca–Mn–O films. Science 264, 413–415 (1994).
Tokura, Y. & Nagaosa, N. Orbital physics in transition-metal oxides. Science 288, 462–468 (2000).
Zeches, R. J. et al. A strain-driven morphotropic phase boundary in BiFeO3 . Science 326, 977–980 (2009).
Morin, F. J. Oxides which show a metal-to-insulator transition at the Néel temperature. Phys. Rev. Lett. 3, 34–36 (1959).
Abbate, M. et al. Soft-x-ray-absorption studies of the electronic-structure changes through the VO2 phase transition. Phys. Rev. B 43, 7263–7266 (1991).
Biermann, S., Poteryaev, A., Lichtenstein, A. I. & Georges, A. Dynamical singlets and correlation-assisted Peierls transition in VO2 . Phys. Rev. Lett. 94, 026404 (2005).
Haverkort, M. W. et al. Orbital-assisted metal-insulator transition in VO2 . Phys. Rev. Lett. 95, 196404 (2005).
McWhan, D. B., Marezio, M., Remeika, J. P. & Dernier, P. D. X-ray diffraction study of metallic VO2 . Phys. Rev. B 10, 490–495 (1974).
Eyert, V. The metal-insulator transitions of VO2: A band theoretical approach. Ann. Phys. 11, 650–704 (2002).
Goodenough, J. B. The two components of the crystallographic transition in VO2 . J. Solid State. Chem. 3, 490–500 (1971).
Goodenough, J. B. Direct cation–cation interactions in several oxides. Phys. Rev. 117, 1442–1451 (1960).
Koethe, T. C. et al. Transfer of spectral weight and symmetry across the metal–insulator transition in VO2 . Phys. Rev. Lett. 97, 116402 (2006).
Tanaka, A. On the metal–insulator transitions in VO2 and Ti2O3 from a unified viewpoint. J. Phys. Soc. Jpn 73, 152–162 (2004).
Butler, S. R. & Gillson, J. L. Crystal growth, electrical resistivity and lattice parameters of RuO2 and IrO2 . Mater. Res. Bull. 6, 81–89 (1971).
Grant, F. A. Properties of rutile (titanium dioxide). Rev. Mod. Phys. 31, 646–674 (1959).
Muraoka, Y., Ueda, Y. & Hiroi, Z. Large modification of the metal–insulator transition temperature in strained VO2 films grown on TiO2 substrates. J. Phys. Chem. Solids 63, 965–967 (2002).
Pouget, J. P., Launois, H., D’Haenens, J. P., Merenda, P. & Rice, T. M. Electron localization induced by uniaxial stress in pure VO2 . Phys. Rev. Lett. 35, 873–875 (1975).
Ferrari, C., Buffagni, E. & Rossi, F. in Characterization of Semiconductor Heterostructures and Nanostructures (eds Agostini, G. & Lamberti, C.) Ch. 3 (Elsevier, 2013).
De Groot, F. M. F. X-ray absorption and dichroism of transition metals and their compounds. J. Electron Spectrosc. Relat. Phenom. 67, 529–622 (1994).
De Groot, F. M. F. et al. Oxygen 1s X-ray-absorption edges of transition-metal oxides. Phys. Rev. B 40, 5715–5723 (1989).
De Groot, F. High-resolution X-ray emission and X-ray absorption spectroscopy. Chem. Rev. 101, 1779–1808 (2001).
Park, J. H. et al. Spin and orbital occupation and phase transitions in V2O3 . Phys. Rev. B 61, 11506–11509 (2000).
Sawatzky, G. A. & Post, D. X-ray photoelectron and Auger spectroscopy study of some vanadium oxides. Phys. Rev. B 20, 1546–1555 (1979).
Zaanen, J., Sawatzky, G. A. & Allen, J. W. Band gaps and electronic structure of transition-metal compounds. Phys. Rev. Lett. 55, 418–421 (1985).
Khomskii, D. I. & Mizokawa, T. Orbitally induced Peierls state in spinels. Phys. Rev. Lett. 94, 156402 (2005).
Senn, M. S., Wright, J. P. & Attfield, J. P. Charge order and three-site distortions in the Verwey structure of magnetite. Nature 481, 173–176 (2012).
Chakhalian, J. et al. Orbital reconstruction and covalent bonding at an oxide interface. Science 318, 1114–1117 (2007).
Pardo, V. & Pickett, W. E. Half-metallic semi-Dirac point generated by quantum confinement in TiO2/VO2 nanostructures. Phys. Rev. Lett. 102, 166803 (2009).
Acknowledgements
The authors thank S. Yang, X. Jiang and A. Pushp for useful discussions and J. Jeong for help with VO2 deposition. Research at Stanford is supported by the US Department of Energy, Office of Basic Energy Sciences, Materials Sciences and Engineering Division under contract DE-AC02-76SF00515. The Advanced Light Source is supported by the Director, Office of Science, Office of Basic Energy Sciences, US Department of Energy under Contract No. DE-AC02-05CH11231. Part of this research was supported by the Stanford Synchrotron Radiation Lightsource, a national user facility operated by Stanford University on behalf of the US Department of Energy, Office of Basic Energy Sciences.
Author information
Authors and Affiliations
Contributions
N.B.A., M.G.S. and S.S.P.P. designed the study. N.B.A. deposited VO2 films and L.G. deposited RuO2 films. X-ray diffraction and electrical transport measurements were performed and analysed by N.B.A., M.D. and M.C. with help from L.G. A.X.G., N.B.A., A.H.R., R.K. and H.O. carried out and analysed the X-ray absorption measurements, with assistance from C.A.J. and E.A. and under the supervision of H.A.D. M.G.S. helped with X-ray absorption data analysis and along with K.P.R. provided thin-film deposition expertise. N.B.A. and S.S.P.P. wrote the paper with contributions from A.X.G., H.A.D. and M.G.S. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2553 kb)
Rights and permissions
About this article
Cite this article
Aetukuri, N., Gray, A., Drouard, M. et al. Control of the metal–insulator transition in vanadium dioxide by modifying orbital occupancy. Nature Phys 9, 661–666 (2013). https://doi.org/10.1038/nphys2733
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphys2733
This article is cited by
-
Modulation-doping a correlated electron insulator
Nature Communications (2023)
-
A neuromorphic physiological signal processing system based on VO2 memristor for next-generation human-machine interface
Nature Communications (2023)
-
Tuning orbital-selective phase transitions in a two-dimensional Hund’s correlated system
Nature Communications (2023)
-
Photo-induced high-temperature ferromagnetism in YTiO3
Nature (2023)
-
Microstructure effects on the phase transition behavior of a prototypical quantum material
Scientific Reports (2022)