Abstract
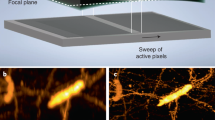
We present a technique called white-light diffraction tomography (WDT) for imaging microscopic transparent objects such as live unlabelled cells. The approach extends diffraction tomography to white-light illumination and imaging rather than scattering plane measurements. Our experiments were performed using a conventional phase contrast microscope upgraded with a module to measure quantitative phase images. The axial dimension of the object was reconstructed by scanning the focus through the object and acquiring a stack of phase-resolved images. We reconstructed the three-dimensional structures of live, unlabelled, red blood cells and compared the results with confocal and scanning electron microscopy images. The 350 nm transverse and 900 nm axial resolution achieved reveals subcellular structures at high resolution in Escherichia coli cells. The results establish WDT as a means for measuring three-dimensional subcellular structures in a non-invasive and label-free manner.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Als-Nielsen, J. & McMorrow, D. Elements of Modern X-ray Physics (Wiley, 2001).
Jiang, H. et al. Quantitative 3D imaging of whole, unstained cells by using X-ray diffraction microscopy. Proc. Natl Acad. Sci. USA 107, 11234–11239 (2010).
Raines, K. S. et al. Three-dimensional structure determination from a single view. Nature 463, 214–217 (2010).
Born, M. & Wolf, E. Principles of Optics: Electromagnetic Theory of Propagation, Interference and Diffraction of Light 7th expanded edn (Cambridge Univ. Press, 1999).
Devaney, A. J. Inverse-scattering theory within the Rytov approximation. Opt. Lett. 6, 374–376 (1981).
Wolf, E. in Advances in Imaging and Electron Physics Vol. 165 (ed. Hawkes, P. W.) Ch. 7 (Academic, 2011).
Wolf, E. Three-dimensional structure determination of semi-transparent objects from holographic data. Opt. Commun. 1, 153–156 (1969).
Gabor, D. A new microscopic principle. Nature 161, 777–778 (1948).
Popescu, G. Quantitative Phase Imaging of Cells and Tissues (McGraw-Hill, 2011).
Paganin, D. & Nugent, K. A. Noninterferometric phase imaging with partially coherent light. Phys. Rev. Lett. 80, 2586–2589 (1998).
Marquet, P. et al. Digital holographic microscopy: a noninvasive contrast imaging technique allowing quantitative visualization of living cells with subwavelength axial accuracy. Opt. Lett. 30, 468–470 (2005).
Chalut, K. J., Brown, W. J. & Wax, A. Quantitative phase microscopy with asynchronous digital holography. Opt. Express 15, 3047–3052 (2007).
Popescu, G. et al. Fourier phase microscopy for investigation of biological structures and dynamics. Opt. Lett. 29, 2503–2505 (2004).
Ikeda, T., Popescu, G., Dasari, R. R. & Feld, M. S. Hilbert phase microscopy for investigating fast dynamics in transparent systems. Opt. Lett. 30, 1165–1168 (2005).
Popescu, G., Ikeda, T., Dasari, R. R. & Feld, M. S. Diffraction phase microscopy for quantifying cell structure and dynamics. Opt. Lett. 31, 775–777 (2006).
Wang, Z. et al. Spatial light interference microscopy (SLIM). Opt. Express 19, 1016–1026 (2011).
Rappaz, B. et al. Comparative study of human erythrocytes by digital holographic microscopy, confocal microscopy, and impedance volume analyzer. Cytometry A 73A, 895–903 (2008).
Khmaladze, A., Kim, M. & Lo, C. M. Phase imaging of cells by simultaneous dual-wavelength reflection digital holography. Opt. Express 16, 10900–10911 (2008).
Shaked, N. T., Rinehart, M. T. & Wax, A. Dual-interference-channel quantitative-phase microscopy of live cell dynamics. Opt. Lett. 34, 767–769 (2009).
Park, Y. K. et al. Refractive index maps and membrane dynamics of human red blood cells parasitized by plasmodium falciparum. Proc. Natl Acad. Sci. USA 105, 13730–13735 (2008).
Park, Y. K. et al. Measurement of red blood cell mechanics during morphological changes. Proc. Natl Acad. Sci. USA 107, 6731–6736 (2010).
Mir, M. et al. Optical measurement of cycle-dependent cell growth. Proc. Natl Acad. Sci. USA 108, 13124–13129 (2011).
Pavillon, N. et al. Early cell death detection with digital holographic microscopy. PLoS ONE 7, e30912 (2012).
Chen, B. Q. & Stamnes, J. J. Validity of diffraction tomography based on the first Born and the first Rytov approximations. Appl. Opt. 37, 2996–3006 (1998).
Carney, P. S., Wolf, E. & Agarwal, G. S. Diffraction tomography using power extinction measurements. J. Opt. Soc. Am. A 16, 2643–2648 (1999).
Lauer, V. New approach to optical diffraction tomography yielding a vector equation of diffraction tomography and a novel tomographic microscope. J. Microsc. 205, 165–176 (2002).
Charriere, F. et al. Living specimen tomography by digital holographic microscopy: morphometry of testate amoeba. Opt. Express 14, 7005–7013 (2006).
Charriere, F. et al. Cell refractive index tomography by digital holographic microscopy. Opt. Lett. 31, 178–180 (2006).
Choi, W. et al. Tomographic phase microscopy. Nature Methods 4, 717–719 (2007).
Cotte, Y. et al. Marker-free phase nanoscopy. Nature Photon. 7, 113–117 (2013).
Choi, W. S., Fang-Yen, C., Badizadegan, K., Dasari, R. R. & Feld, M. S. Extended depth of focus in tomographic phase microscopy using a propagation algorithm. Opt. Lett. 33, 171–173 (2008).
Zhou, R., Edwards, C., Arbabi, A., Popescu, G. & Goddard, L. L. Detecting 20 nm wide defects in large area nanopatterns using optical interferometric microscopy. Nano Lett. 13, 3716–3721 (2013).
Goodman, J. W. Speckle Phenomena in Optics: Theory and Applications (Roberts & Co., 2007).
Mir, M. et al. Visualizing Escherichia coli sub-cellular structure using sparse deconvolution spatial light interference tomography. PLoS ONE 7, e39816 (2012).
Wang, Z. et al. Spatial light interference tomography (SLIT). Opt. Express 19, 19907–19918 (2011).
Bon, P., Aknoun, S., Savatier, J., Wattellier, B. & Monneret, S. in Three-Dimensional and Multidimensional Microscopy: Image Acquisition and Processing XX (eds Cogswell, C. J., Brown, T. G., Conchello, J.-A. & Wilson, T.) 858–918 SPIE (2013).
Pawley, J. B. Handbook of Biological Confocal Microscopy 3rd edn (Springer, 2006).
Ding, H. F., Wang, Z., Nguyen, F., Boppart, S. A. & Popescu, G. Fourier transform light scattering of inhomogeneous and dynamic structures. Phys. Rev. Lett. 101, 238102 (2008).
Chew, W. C. Waves and Fields in Inhomogeneous Media (IEEE Press, 1995).
Kim, T., Sridharan, S. & Popescu, G. in Handbook of Coherent-Domain Optical Methods Vol. 1 (ed. Tuchin, V. V.) Ch. 7, 259–290 (Springer, 2013).
Huang, D. et al. Optical coherence tomography. Science 254, 1178–1181 (1991).
Ralston, T. S., Marks, D. L., Carney, P. S. & Boppart, S. A. Interferometric synthetic aperture microscopy. Nature Phys. 3, 129–134 (2007).
Bain, B. J. A Beginner's Guide to Blood Cells 2nd edn (Blackwell, 2004).
Khairy, K., Foo, J. & Howard, J. Shapes of red blood cells: comparison of 3D confocal images with the bilayer-couple model. Cell. Mol. Bioeng. 1, 173–181 (2008).
Babacan, S. D., Wang, Z., Do, M. & Popescu, G. Cell imaging beyond the diffraction limit using sparse deconvolution spatial light interference microscopy. Biomed. Opt. Exp. 2, 1815–1827 (2011).
Donachie, W. D. Co-ordinate regulation of the Escherichia coli cell cycle or the cloud of unknowing. Mol. Microbiol. 40, 779–785 (2001).
Raskin, D. M. & De Boer, P. A. J. Rapid pole-to-pole oscillation of a protein required for directing division to the middle of Escherichia coli. Proc. Natl Acad. Sci. USA 96, 4971–4976 (1999).
Ghosh, A. S. & Young, K. D. Helical disposition of proteins and lipopolysaccharide in the outer membrane of Escherichia coli. J. Bacteriol. 187, 1913–1922 (2005).
Wiener, N. Generalized harmonic analysis. Acta Mathematica 55, 117–258 (1930).
Khintchine, A. Eine verschärfung des poincaréschen ‘wiederkehrsatzes’. Comp. Math 1, 177–179 (1935).
Acknowledgements
This research was supported in part by the National Science Foundation (grants CBET-1040462 MRI, CBET 08-46660 CAREER) and the Science and Technology Center for Emergent Behaviors of Integrated Cellular Systems (EBICS, CBET-0939511). The authors thank R. Bashir and K. Park for providing HT29 cells, I. Golding and M. Bednarz for providing E. coli cells and S. Robinson for assistance with SEM imaging of RBCs. The authors also thank J. Howard, K. Khairy and J.-J. Foo for providing confocal images of RBCs. R.Z. acknowledges support from the Beckman Foundation through a Beckman Graduate Fellowship. For more information, visit http://light.ece.illinois.edu
Author information
Authors and Affiliations
Contributions
G.P., R.Z. and S.D.B. proposed the idea. G.P., R.Z., T.K. and P.S.C. developed the theoretical description of the method. T.K. and R.Z. performed three-dimensional PSF calculations. T.K. and M.M. performed quantitative phase imaging. S.D.B. and M.M. developed the sparse deconvolution method. T.K. and R.Z. performed data analysis and three-dimensional reconstruction. G.P. and L.L.G. supervised the research. All authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.P. has financial interest in Phi Optics, Inc., a company developing quantitative phase imaging technology for materials and life science applications, which, however, did not sponsor the research.
Supplementary information
Supplementary information
Supplementary information (PDF 2058 kb)
Supplementary information
Supplementary movie (MP4 867 kb)
Supplementary information
Supplementary movie (MP4 1164 kb)
Supplementary information
Supplementary movie (MP4 1115 kb)
Rights and permissions
About this article
Cite this article
Kim, T., Zhou, R., Mir, M. et al. White-light diffraction tomography of unlabelled live cells. Nature Photon 8, 256–263 (2014). https://doi.org/10.1038/nphoton.2013.350
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphoton.2013.350
This article is cited by
-
Characterizing dry mass and volume changes in human multiple myeloma cells upon treatment with proteotoxic and genotoxic drugs
Clinical and Experimental Medicine (2023)
-
Live-dead assay on unlabeled cells using phase imaging with computational specificity
Nature Communications (2022)
-
In memory of Gabriel Popescu
Nature Photonics (2022)
-
Adaptive dynamic range shift (ADRIFT) quantitative phase imaging
Light: Science & Applications (2021)
-
Label-free SARS-CoV-2 detection and classification using phase imaging with computational specificity
Light: Science & Applications (2021)