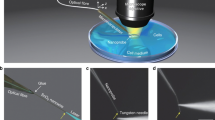
Abstract
Since their invention some 50 years ago1, lasers have made a tremendous impact on modern science and technology. Nevertheless, lasing has so far relied on artificial or engineered optical gain materials, such as doped crystals, semiconductors, synthetic dyes and purified gases2,3. Here, we show that fluorescent proteins4,5 in cells are a viable gain medium for optical amplification, and report the first successful realization of biological cell lasers based on green fluorescent protein (GFP). We demonstrate in vitro protein lasers using recombinant GFP solutions and introduce a laser based on single live cells expressing GFP. On optical pumping with nanojoule/nanosecond pulses, individual cells in a high-Q microcavity produce bright, directional and narrowband laser emission, with characteristic longitudinal and transverse modes. Lasing cells remained alive even after prolonged lasing action. Light amplification and lasing from and within biological systems pave the way to new forms of intracellular sensing, cytometry and imaging.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Maiman, T. H. Stimulated optical radiation in ruby. Nature 187, 493–494 (1960).
Weber, M. J. Handbook of Laser Wavelengths (CRC Press, 1999).
Townes, C. H. How the Laser Happened: Adventures of a Scientist (Oxford Univ. Press, 1999).
Shaner, N. C., Steinbach, P. A. & Tsien, R. Y. A guide to choosing fluorescent proteins. Nature Methods 2, 905–909 (2005).
Giepmans, B. N. G., Adams, S. R., Ellisman, M. H. & Tsien, R. Y. Review—the fluorescent toolbox for assessing protein location and function. Science 312, 217–224 (2006).
Shimomura, O., Johnson, F. H. & Saiga, Y. Extraction, purification and properties of aequorin, a bioluminescent protein from luminous hydromedusan, Aequorea. J. Cell. Comp. Physiol. 59, 223–239 (1962).
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W. W. & Prasher, D. C. Green fluorescent protein as a marker for gene-expression. Science 263, 802–805 (1994).
Heim, R., Cubitt, A. B. & Tsien, R. Y. Improved green fluorescence. Nature 373, 663–664 (1995).
Cormack, B. P., Valdivia, R. H. & Falkow, S. Facs-optimized mutants of the green fluorescent protein (gfp). Gene 173, 33–38 (1996).
Shaner, N. C. et al. Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein. Nature Biotechnol. 22, 1567–1572 (2004).
Merzlyak, E. M. et al. Bright monomeric red fluorescent protein with an extended fluorescence lifetime. Nature Methods 4, 555–557 (2007).
Patterson, G. H., Knobel, S. M., Sharif, W. D., Kain, S. R. & Piston, D. W. Use of the green fluorescent protein and its mutants in quantitative fluorescence microscopy. Biophys. J. 73, 2782–2790 (1997).
Pikas, D. J. et al. Nonlinear saturation and lasing characteristics of green fluorescent protein. J. Phys. Chem. B 106, 4831–4837 (2002).
Siegman, A. E. Lasers (University Science Books, 1986).
Chen, Y., Wei, L. N. & Muller, J. D. Probing protein oligomerization in living cells with fluorescence fluctuation spectroscopy. Proc. Natl Acad. Sci. USA 100, 15492–15497 (2003).
Lu, P., Vogel, C., Wang, R., Yao, X. & Marcotte, E. M. Absolute protein expression profiling estimates the relative contributions of transcriptional and translational regulation. Nature Biotechnol. 25, 117–124 (2007).
Kim, H. & Seed, B. The transcription factor MafB antagonizes antiviral responses by blocking recruitment of coactivators to the transcription factor IRF3. Nature Immunol. 11, 743–750 (2010).
Kemper, B. et al. Integral refractive index determination of living suspension cells by multifocus digital holographic phase contrast microscopy. J. Biomed. Opt. 12, 054009 (2007).
Svelto, O. Principles of Lasers (Springer-Verlag, 2009).
Bandres, M. A. & Gutiérrez-Vega, J. C. Ince-Gaussian modes of the paraxial wave equation and stable resonators. J. Opt. Soc. Am. A 21, 873–880 (2004).
Rose, A., Zhu, Z. G., Madigan, C. F., Swager, T. M. & Bulovic, V. Sensitivity gains in chemosensing by lasing action in organic polymers. Nature 434, 876–879 (2005).
Park, Y. et al. Refractive index maps and membrane dynamics of human red blood cells parasitized by Plasmodium falciparum. Proc. Natl Acad. Sci. USA 105, 13730–13735 (2008).
Klar, T. A., Jakobs, S., Dyba, M., Egner, A. & Hell, S. W. Fluorescence microscopy with diffraction resolution barrier broken by stimulated emission. Proc. Natl Acad. Sci. USA 97, 8206–8210 (2000).
Westphal, V. et al. Video-rate far-field optical nanoscopy dissects synaptic vesicle movement. Science 320, 246–249 (2008).
Min, W. et al. Imaging chromophores with undetectable fluorescence by stimulated emission microscopy. Nature 461, 1105–1109 (2009).
Saar, B. G. et al. Video-rate molecular imaging in vivo with stimulated Raman scattering. Science 330, 1368–1370 (2010).
Noginov, M. A. et al. Demonstration of a spaser-based nanolaser. Nature 460, 1110–1113 (2009).
Ma, R.-M., Oulton, R. F., Sorger, V. J., Bartal, G. & Zhang, X. Room-temperature sub-diffraction-limited plasmon laser by total internal reflection. Nature Mater. 10, 110–113 (2011).
Acknowledgements
The authors thank Ji-Joon Song (KAIST, Korea) for providing recombinant eGFP solutions, S. Sassi and B. Seed (Harvard Medical School) for the donation of 293ETN cells and support with eGFP transfection, U. Shama for initial testing of a fluorescent protein and W. Farinelli for setting up the OPO system. This work was supported in part by the US National Science Foundation (ECCS-1101947) and the Korea National Research Foundation (R31-2008-000-10071-0). M.C.G. acknowledges financial support from the Bullock-Wellman Fellowship.
Author information
Authors and Affiliations
Contributions
M.C.G. designed and performed the experiments. S.H.Y conceived and supervised the project. Both authors discussed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Gather, M., Yun, S. Single-cell biological lasers. Nature Photon 5, 406–410 (2011). https://doi.org/10.1038/nphoton.2011.99
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphoton.2011.99
This article is cited by
-
Generalized Laguerre–Gaussian expansion for propagating Ince–Gaussian beams through paraxial optical system
Optical and Quantum Electronics (2024)
-
Ince–Gaussian beams propagation through turbulent atmospheric medium
Optical and Quantum Electronics (2024)
-
Ultra-small low-threshold mid-infrared plasmonic nanowire lasers based on n-doped GaN
Discover Nano (2023)
-
Highly nonlinear optic nucleic acid thin-solid film to generate short pulse laser
Scientific Reports (2023)
-
Multiplexed spatially-focused localization of light in adipose biological tissues
Scientific Reports (2022)