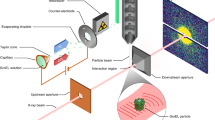
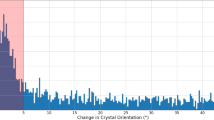
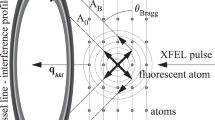
Abstract
X-ray free-electron lasers have enabled new approaches to the structural determination of protein crystals that are too small or radiation-sensitive for conventional analysis1. For sufficiently short pulses, diffraction is collected before significant changes occur to the sample, and it has been predicted that pulses as short as 10 fs may be required to acquire atomic-resolution structural information1,2,3,4. Here, we describe a mechanism unique to ultrafast, ultra-intense X-ray experiments that allows structural information to be collected from crystalline samples using high radiation doses without the requirement for the pulse to terminate before the onset of sample damage. Instead, the diffracted X-rays are gated by a rapid loss of crystalline periodicity, producing apparent pulse lengths significantly shorter than the duration of the incident pulse. The shortest apparent pulse lengths occur at the highest resolution, and our measurements indicate that current X-ray free-electron laser technology5 should enable structural determination from submicrometre protein crystals with atomic resolution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chapman, H. N. et al. Femtosecond X-ray protein nanocrystallography. Nature 470, 73–77 (2011).
Neutze, R. et.al. Potential for biomolecular imaging with femtosecond X-ray pulses. Nature 406, 752–757 (2000).
Chapman, H. N. et al. Femtosecond time-delay X-ray holography. Nature 448, 676–679 (2007).
Hau-Riege, S. P. et al. Subnanometer-scale measurements of the interaction of ultrafast soft X-ray free-electron-laser pulses with matter. Phys. Rev. Lett. 98, 145502 (2007).
Emma, P. et al. First lasing and operation of an Ångstrom wavelength free-electron laser. Nature Photon. 4, 641–647 (2010).
Bergh, M., Timneanu, N. & van der Spoel, D. Model for the dynamics of a water cluster in an X-ray free electron laser beam. Phys. Rev. E 70, 051904 (2004).
Caleman, C. et al. On the feasibility of nanocrystal imaging using intense and ultrashort X-ray pulses. ACS Nano 5, 139–146 (2011).
Hau-Riege, S. P., London, R. A., Chapman, H. N. & Bergh, M. Soft-X-ray free-electron-laser interaction with materials. Phys. Rev. E 76, 046403 (2007).
Hau-Riege, S. P., London, R. A. & Szoke, A. Dynamics of biological molecules irradiated by short X-ray pulses. Phys. Rev. E 69, 051906 (2002).
Gnodtke, C., Saalmann, U. & Rost, J. M. Ionization and charge migration through strong internal fields in clusters exposed to intense X-ray pulses. Phys. Rev. A 79, 041201 (2009).
Bergh, M., Huldt, G., Timneanu, N., Maia, F. R. N. C. & Hajdu, J. Feasibility of imaging living cells at subnanometer resolutions by ultrafast X-ray diffraction. Q. Rev. Biophys. 41, 181–204 (2008).
Jordan, P. et al. Three-dimensional structure of cyanobacterial photosystem I at 2.5 Å resolution. Nature 411, 909–917 (2001).
Owen, R. L., Rudino-Pinera, E. & Garman, E. F. Experimental determination of the radiation dose limit for cryocooled protein crystals. Proc. Natl Acad. Sci. USA 103, 4912–4917 (2006).
Henderson, R. The potential and limitations of neutrons, electrons and X-rays for atomic resolution microscopy of unstained biological molecules. Q. Rev. Biophys. 28, 171–193 (1995).
Holton, J. M. A beginner's guide to radiation damage. J. Synchr. Rad. 16, 133–142 (2009).
Hau-Riege, S. P. X-ray atomic scattering factors of low-z ions with a core hole. Phys. Rev. A 76, 042511 (2007).
Quiney, H. M. & Nugent, K. A. Biomolecular imaging and electronic damage using X-ray free-electron lasers. Nature Phys. 7, 142–146 (2011).
Warren, B. E. X-Ray Diffraction 229 (Dover, 1990).
Scott, H. A. Cretin—a radiative transfer capability for laboratory plasmas. J. Quant. Spect. Radiat. Transf. 71, 689–701 (2001).
Young, L. et al. Femtosecond electronic response of atoms to ultra intense X-rays. Nature 466, 56–61 (2010).
Bozek, J. D. AMO instrumentation for the LCLS X-ray FEL. Euro. Phys. J. Special Topics 169, 129–132 (2009).
Strüder, L. et al. Large-format, high-speed, X-ray pnCCDs combined with electron and ion imaging spectrometers in a multipurpose chamber for experiments at 4th generation light sources. Nucl. Instrum. Meth. Phys. Res. A 614, 483–496 (2010).
DePonte, D. P. et al. Gas dynamic virtual nozzle for generation of microscopic droplet streams. J. Phys. D 41, 195505 (2008).
Kirian, R. et al. Femtosecond protein nanocrystallography—data analysis methods. Opt. Express 18, 5713–5723 (2010).
Caleman, C. et al.: Simulations of radiation damage in biomolecular nanocrystals induced by femtosecond X-ray pulses. J. Mod. Opt. 58, 1486–1497 (2011).
Andreasson, J. et al. Saturated ablation in metal hydrides and acceleration of protons and deuterons to keV energies with a soft-X-ray laser. Phys. Rev. E 83, 016403 (2011).
Acknowledgements
Experiments were carried out at the Linac Coherent Light Source national user facilities operated by Stanford University on behalf of the US Department of Energy (DOE), Office of Basic Energy Sciences. The authors acknowledge support from the Helmholtz Association, the Max Planck Society for funding the development and operation of the CAMP instrument within the ASG at CFEL, the DOE through the PULSE Institute at the SLAC National Accelerator Laboratory, and the Lawrence Livermore National Laboratory (contract DE-AC52-07NA27344), the US National Science Foundation (awards 0417142 and MCB-1021557), the US National Institutes of Health (awards 1R01GM095583-01 (ROADMAP) and 1U54GM094625-01 (PSI:Biology)), the Joachim Herz Stiftung and the Swedish Research Council. The authors also thank the staff of the LCLS for their support in carrying out these experiments, and D. van der Spoel for providing computational resources.
Author information
Authors and Affiliations
Contributions
H.N.C., J.C.H.S., A.B. and P.F. conceived the experiment, which was designed with T.A.W., R.A.K., J.S., D.D.P., U.W., R.B.D., S.Bo., M.J.B., D.S., I.S., S.M. and J.H. The CAMP instrument was the responsibility of S.W.E., R.H., D.R., A.R., L.F., N.K., P.H., B.R., B.E., A.H., Ch.R., G.W., L.S., G.H., H.G., J.U., I.S., H.So., H.H., L.G., H.G. and C.W., who operated the pnCCD detectors. C.B., J.B. and M.M. set up and aligned the beamline. P.F., M.S.H. and I.G. prepared samples. R.B.D., D.D.P., U.W., J.C.H.S., P.F., L.L. and R.L.S. developed and operated the sample delivery system. H.N.C., A.B., A.A., J.S., D.P.P., U.W., R.B.D., S.Ba., M.J.B., L.G., J.H., M.M.S., N.T., J.A., S.St. and J.C.H.S. developed diffraction instrumentation. M.B., M.L., A.B. and K.N. designed and/or fabricated calibration samples. H.N.C., J.C.H.S., P.F., A.B., T.A.W., R.A.K., C.C., A.A., L.L., J.S., D.P.D., U.W., R.B.D., I.S., N.C., R.L.S., M.S.H., M.B., S.W.E., R.H., D.R., A.R., S.K., T.E., M.L., C.B., J.U., L.F., J.D.B., M.M., M.F., C.Y.H., R.G.S., G.J.W., A.R., M.S., O.J., I.A. and J.H. carried out the experiment. A.B, C.C., N.T. and H.N.C. developed the theory, and analysed the data with L.L., T.A.W., I.S. and T.R.M.B. N.T., C.C. and H.S. carried out the Cretin simulations. A.B., C.C. and H.N.C. wrote the manuscript with discussion and improvements from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1655 kb)
Supplementary information
Supplementary Movie (MOV 162913 kb)
Rights and permissions
About this article
Cite this article
Barty, A., Caleman, C., Aquila, A. et al. Self-terminating diffraction gates femtosecond X-ray nanocrystallography measurements. Nature Photon 6, 35–40 (2012). https://doi.org/10.1038/nphoton.2011.297
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nphoton.2011.297
This article is cited by
-
Direct high-resolution X-ray imaging exploiting pseudorandomness
Light: Science & Applications (2023)
-
Water layer and radiation damage effects on the orientation recovery of proteins in single-particle imaging at an X-ray free-electron laser
Scientific Reports (2023)
-
Density functional tight binding approach utilized to study X-ray-induced transitions in solid materials
Scientific Reports (2022)
-
Few-fs resolution of a photoactive protein traversing a conical intersection
Nature (2021)
-
Viscosity-adjustable grease matrices for serial nanocrystallography
Scientific Reports (2020)