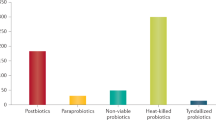
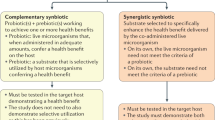
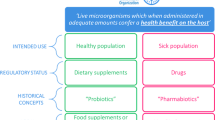
Abstract
The leading probiotics currently available to consumers are generally drawn from a narrow range of organisms. Knowledge of the gut microbiota and its constituent actors is changing this paradigm, particularly given the phylogenetic range and relatively unknown characteristics of the organisms under investigation as novel therapeutics. For this reason, and because their development is likely to be more amenable to a pharmaceutical than a food delivery route, these organisms are often operationally referred to as next-generation probiotics, a concept that overlaps with the emerging concept of live biotherapeutic products. The latter is a class of organisms developed exclusively for pharmaceutical application. In this Perspective, we discuss what lessons have been learned from working with traditional probiotics, explore the kinds of organisms that are likely to be used as novel microbial therapeutics, discuss the regulatory framework required, and propose how scientists may meet this challenge.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hill, C. et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 11, 506–514 (2014).
EFSA. Statement on the update of the list of QPS-recommended biological agents intentionally added to food or feed as notified to EFSA. 2: suitability of taxonomic units notified to EFSA until March 2015. EFSA J. 13, 4138 (2015).
Sun, X., Fiala, J. L. & Lowery, D. Patent watch: modulating the human microbiome with live biotherapeutic products: intellectual property landscape. Nat. Rev. Drug. Discov. 15, 224–225 (2016).
Deng, H. M. et al. A novel strain of Bacteroides fragilis enhances phagocytosis and polarises M1 macrophages. Sci. Rep. 6, 29401 (2016).
Moncrief, J. S., Duncan, A. J., Wright, R. L., Barroso, L. A. & Wilkins, T. D. Molecular characterization of the fragilysin pathogenicity islet of enterotoxigenic Bacteroides fragilis. Infect. Immun. 66, 1735–1739 (1998).
Obiso, R. J. Jr., Azghani, A. O. & Wilkins, T. D. The Bacteroides fragilis toxin fragilysin disrupts the paracellular barrier of epithelial cells. Infect. Immun. 65, 1431–1439 (1997).
Shiryaev, S. A. et al. Substrate cleavage profiling suggests a distinct function of Bacteroides fragilis metalloproteinases (fragilysin and metalloproteinase II) at the microbiome-inflammation-cancer interface. J. Biol. Chem. 288, 34956–34967 (2013).
Mazmanian, S. K., Liu, C. H., Tzianabos, A. O. & Kasper, D. L. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122, 107–118 (2005).
Ulsemer, P., Toutounian, K., Schmidt, J., Karsten, U. & Goletz, S. Preliminary safety evaluation of a new Bacteroides xylanisolvens isolate. Appl. Environ. Microbiol. 78, 528–535 (2012).
Ulsemer, P. et al. Impact of oral consumption of heat-treated Bacteroides xylanisolvens DSM 23964 on the level of natural TFα-specific antibodies in human adults. Benef. Microbes 7, 485–500 (2016).
Yanagibashi, T. et al. IgA production in the large intestine is modulated by a different mechanism than in the small intestine: Bacteroides acidifaciens promotes IgA production in the large intestine by inducing germinal center formation and increasing the number of IgA+ B cells. Immunobiology 218, 645–651 (2013).
Woo, T. D. et al. Inhibition of the cytotoxic effect of Clostridium difficile in vitro by Clostridium butyricum MIYAIRI 588 strain. J. Med. Microbiol. 60, 1617–1625 (2011).
Shimbo, I. et al. Effect of Clostridium butyricum on fecal flora in Helicobacter pylori eradication therapy. World J. Gastroenterol. 11, 7520–7524 (2005).
Kobashi, K., Takeda, Y., Itoh, H. & Hase, J. Cholesterol-lowering effect of Clostridium butyricum in cholesterol-fed rats. Digestion 26, 173–178 (1983).
Takeda, Y., Itoh, H. & Kobashi, K. Effect of Clostridium butyricum on the formation and dissolution of gallstones in experimental cholesterol cholelithiasis. Life Sci. 32, 541–546 (1983).
Shinnoh, M. et al. Clostridium butyricum MIYAIRI 588 shows antitumor effects by enhancing the release of TRAIL from neutrophils through MMP-8. Int. J. Oncol. 42, 903–911 (2013).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 105, 16731–16736 (2008).
Rossi, O. et al. Faecalibacterium prausnitzii A2–165 has a high capacity to induce IL-10 in human and murine dendritic cells and modulates T cell responses. Sci. Rep. 6, 18507 (2016).
Zhang, M. et al. Faecalibacterium prausnitzii inhibits interleukin-17 to ameliorate colorectal colitis in rats. PLoS ONE 9, e109146 (2014).
Motta, J. P. et al. Food-grade bacteria expressing elafin protect against inflammation and restore colon homeostasis. Sci. Transl. Med. 4, 158ra144 (2012).
Frossard, C. P., Steidler, L. & Eigenmann, P. A. Oral administration of an IL-10-secreting Lactococcus lactis strain prevents food-induced IgE sensitization. J. Allergy Clin. Immunol. 119, 952–959 (2007).
Caluwaerts, S. et al. AG013, a mouth rinse formulation of Lactococcus lactis secreting human trefoil factor 1, provides a safe and efficacious therapeutic tool for treating oral mucositis. Oral Oncol. 46, 564–570 (2010).
Robert, S. & Steidler, L. Recombinant Lactococcus lactis can make the difference in antigen-specific immune tolerance induction, the type 1 diabetes case. Microb. Cell Fact. 13, (Suppl 1), S11 (2014).
Farrar, M. D. et al. Engineering of the gut commensal bacterium Bacteroides ovatus to produce and secrete biologically active murine interleukin-2 in response to xylan. J. Appl. Microbiol. 98, 1191–1197 (2005).
Hamady, Z. Z. et al. Xylan-regulated delivery of human keratinocyte growth factor-2 to the inflamed colon by the human anaerobic commensal bacterium Bacteroides ovatus. Gut 59, 461–469 (2010).
Hamady, Z. Z. et al. Treatment of colitis with a commensal gut bacterium engineered to secrete human TGF-β1 under the control of dietary xylan 1. Inflamm. Bowel Dis. 17, 1925–1935 (2011).
Hamady, Z. Z. et al. Identification and use of the putative Bacteroides ovatus xylanase promoter for the inducible production of recombinant human proteins. Microbiology 154, 3165–3174 (2008).
EC. Regulation (EC) no 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods. OJ L. 404, 9–25 (2013).
Substantiation for Structure/Function Claims Made in Infant Formula Labels and Labeling: Guidance for Industry (FDA, 2016).
Dietary Supplements: New Dietary Ingredient Notifications and Related Issues: Guidance for Industry (FDA, 2016).
Early Clinical Trials with Live Biotherapeutic Products: Chemistry, Manufacturing, and Control Information: Guidance for Industry (FDA, 2016).
Sanders, M. E., Shane, A. L. & Merenstein, D. J. Advancing probiotic research in humans in the United States: challenges and strategies. Gut Microbes 7, 97–100 (2016).
Investigational New Drug Applications — Determining Whether Human Research Studies Can Be Conducted Without an Investigational New Drug Application: Guidance for Clinical Investigators, Sponsors, and Institutional Review Boards; Partial Stay and Republication of Guidance (FDA, 2015).
Olle, B. Medicines from microbiota. Nat. Biotechnol. 31, 309–315 (2013).
van Nood, E. et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N. Engl. J. Med. 368, 407–415 (2013).
Petrof, E. O. & Khoruts, A. From stool transplants to next-generation microbiota therapeutics. Gastroenterol. 146, 1573–1582 (2014).
Ulsemer, P. et al. Specific humoral immune response to the Thomsen-Friedenreich tumor antigen (CD176) in mice after vaccination with the commensal bacterium Bacteroides ovatus D-6. Cancer Immunol. Immunother. 62, 875–887 (2013).
Gerard, P. et al. Bacteroides sp. strain D8, the first cholesterol-reducing bacterium isolated from human feces. Appl. Environ. Microbiol. 73, 5742–5749 (2007).
Chen, J. C., Lee, W. J., Tsou, J. J., Liu, T. P. & Tsai, P. L. Effect of probiotics on postoperative quality of gastric bypass surgeries: a prospective randomized trial. Surg. Obes. Relat. Dis. 12, 57–61 (2016).
Hosomi, M., Tanida, N. & Shimoyama, T. The role of intestinal bacteria in gallstone formation in animal model. A study on biliary lipid composition and bile acid profiles in bile, small intestinal contents and feces of clostridium butyricum MIYAIRI no. 588 monocontaminated mice. Gastroenterol. Jpn. 17, 316–323 (1982).
Isa, K. et al. Safety assessment of the Clostridium butyricum MIYAIRI 588(R) probiotic strain including evaluation of antimicrobial sensitivity and presence of Clostridium toxin genes in vitro and teratogenicity in vivo. Hum. Exp. Toxicol. 35, 818–832 (2016).
Kohiruimaki, M. et al. Effects of active egg white product/Clostridium butyricum MIYAIRI 588 additive on peripheral leukocyte populations in periparturient dairy cows. J. Vet. Med. Sci. 70, 321–323 (2008).
Kuroiwa, T., Kobari, K. & Iwanaga, M. Inhibition of enteropathogens by Clostridium butyricum MIYAIRI 588. Kansenshogaku Zasshi 64, 257–263 (1990).
Murayama, T. et al. Effects of orally administered Clostridium butyricum MIYAIRI 588 on mucosal immunity in mice. Vet. Immunol. Immunopathol. 48, 333–342 (1995).
Nakanishi, S. & Tanaka, M. Sequence analysis of a bacteriocinogenic plasmid of Clostridium butyricum and expression of the bacteriocin gene in Escherichia coli. Anaerobe 16, 253–257 (2010).
Sato, S., Nagai, H. & Igarashi, Y. Effect of probiotics on serum bile acids in patients with ulcerative colitis. Hepatogastroenterology 59, 1804–1808 (2012).
Seki, H. et al. Prevention of antibiotic-associated diarrhea in children by Clostridium butyricum MIYAIRI. Pediatr. Int. 45, 86–90 (2003).
Seo, M. et al. Clostridium butyricum MIYAIRI 588 improves high-fat diet-induced non-alcoholic fatty liver disease in rats. Dig. Dis. Sci. 58, 3534–3544 (2013).
Takahashi, M. et al. The effect of probiotic treatment with Clostridium butyricum on enterohemorrhagic Escherichia coli O157:H7 infection in mice. FEMS Immunol. Med. Microbiol. 41, 219–226 (2004).
Weng, H., Endo, K., Li, J., Kito, N. & Iwai, N. Induction of peroxisomes by butyrate-producing probiotics. PLoS ONE 10, e0117851 (2015).
Yasueda, A. et al. The effect of Clostridium butyricum MIYAIRI on the prevention of pouchitis and alteration of the microbiota profile in patients with ulcerative colitis. Surg. Today 46, 939–949 (2016).
Simonyte Sjodin, K., Vidman, L., Ryden, P. & West, C. E. Emerging evidence of the role of gut microbiota in the development of allergic diseases. Curr. Opin. Allergy Clin. Immunol. 16, 390–395 (2016).
Song, H., Yoo, Y., Hwang, J., Na, Y. C. & Kim, H. S. Faecalibacterium prausnitzii subspecies-level dysbiosis in the human gut microbiome underlying atopic dermatitis. J. Allergy Clin. Immunol. 137, 852–860 (2016)
Acknowledgements
We thank the panel members of the ISAPP 2016 meeting for stimulating discussions, and M. E. Sanders for reviewing the section on regulations. The opinions in this article are those of the authors only, and do not represent a consensus of the ISAPP convened panel.
Author information
Authors and Affiliations
Contributions
P.W.O.T. and C.H. are funded in part by Science Foundation Ireland (APC/SFI/12/RC/2273) in the form of a research centre which is/has recently been in receipt of research grants from the following companies: Cremo, Mead Johnson Nutrition, Kerry, General Mills, GE Healthcare, Friesland Campina, Sigmoid, Alimentary Health, Second Genome, Nutricia, Danone, Janssen, AbbVie, Suntory Morinaga Milk Industry Ltd, Pfizer Consumer Health, Radisens, 4D Pharma, Crucell, Adare Pharma, Artugen Therapeutics, Caelus. P.W.O.T. is a founder shareholder of Tucana Health Ltd. C.H. is a founder shareholder in Artugen therapeutics. These relationships with industry have no bearing on the present work and neither influenced nor constrained it. J.R.M. has consulted and received payment from Cultech Ltd, Takeda Pharmaceuticals and Unilever.
Corresponding author
Rights and permissions
About this article
Cite this article
O’Toole, P., Marchesi, J. & Hill, C. Next-generation probiotics: the spectrum from probiotics to live biotherapeutics. Nat Microbiol 2, 17057 (2017). https://doi.org/10.1038/nmicrobiol.2017.57
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2017.57
This article is cited by
-
Case report: Aberrant fecal microbiota composition of an infant diagnosed with prolonged intestinal botulism
Gut Pathogens (2024)
-
Pitting the olive seed microbiome
Environmental Microbiome (2024)
-
Utilization of the microbiome in personalized medicine
Nature Reviews Microbiology (2024)
-
Clostridium butyricum MIYAIRI 588 contributes to the maintenance of intestinal microbiota diversity early after haematopoietic cell transplantation
Bone Marrow Transplantation (2024)
-
Synbiotics as potent functional food: recent updates on therapeutic potential and mechanistic insight
Journal of Food Science and Technology (2024)