Abstract
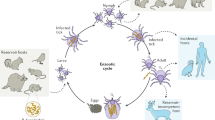
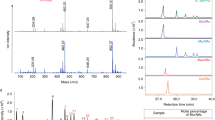
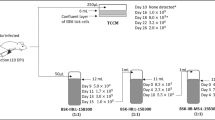
Thiamin pyrophosphate (ThDP), the active form of thiamin (vitamin B1), is believed to be an essential cofactor for all living organisms1,2. Here, we report the unprecedented result that thiamin is dispensable for the growth of the Lyme disease pathogen Borrelia burgdorferi (Bb)3. Bb lacks genes for thiamin biosynthesis and transport as well as known ThDP-dependent enzymes4, and we were unable to detect thiamin or its derivatives in Bb cells. We showed that eliminating thiamin in vitro and in vivo using BcmE, an enzyme that degrades thiamin, has no impact on Bb growth and survival during its enzootic infectious cycle. Finally, high-performance liquid chromatography analysis reveals that the level of thiamin and its derivatives in Ixodes scapularis ticks, the enzootic vector of Bb, is extremely low. These results suggest that by dispensing with use of thiamin, Borrelia, and perhaps other tick-transmitted bacterial pathogens, are uniquely adapted to survive in tick vectors before transmitting to mammalian hosts. To our knowledge, such a mechanism has not been reported previously in any living organisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jurgenson, C. T., Begley, T. P. & Ealick, S. E. The structural and biochemical foundations of thiamin biosynthesis. Annu. Rev. Biochem. 78, 569–603 (2009).
Begley, T. P. et al. Thiamin biosynthesis in prokaryotes. Arch. Microbiol. 171, 293–300 (1999).
Burgdorfer, W. et al. Lyme disease—a tick-borne spirochetosis? Science 216, 1317–1319 (1982).
Fraser, C. M. et al. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature 390, 580–586 (1997).
Downs, D. M. Understanding microbial metabolism. Annu. Rev. Microbiol. 60, 533–559 (2006).
Eijkman, C. [Anti-neuritis vitamin and beriberi. Nobel prize paper. 1929]. Ned. Tijdschr. Geneeskd. 134, 1654–1657 (1990).
Webb, E., Claas, K. & Downs, D. thiBPQ encodes an ABC transporter required for transport of thiamine and thiamine pyrophosphate in Salmonella typhimurium. J. Biol. Chem. 273, 8946–8950 (1998).
Jenkins, A. H., Schyns, G., Potot, S., Sun, G. & Begley, T. P. A new thiamin salvage pathway. Nat. Chem. Biol. 3, 492–497 (2007).
Bazurto, J. V., Farley, K. R. & Downs, D. M. An unexpected route to an essential cofactor: Escherichia coli relies on threonine for thiamine biosynthesis. MBio 7, e01840-15 (2016).
Radolf, J. D., Caimano, M. J., Stevenson, B. & Hu, L. T. Of ticks, mice and men: understanding the dual-host lifestyle of Lyme disease spirochaetes. Nat. Rev. Microbiol. 10, 87–99 (2012).
Bale, S., Rajashankar, K. R., Perry, K., Begley, T. P. & Ealick, S. E. HMP binding protein thiY and HMP-P synthase THI5 are structural homologues. Biochemistry 49, 8929–8936 (2010).
Button, D. K. Selective thiamine removal from culture media by ultraviolet irradiation. Appl. Microbiol. 16, 530–531 (1968).
Dwivedi, B. K. & Arnold, R. G. Chemistry of thiamine degradation in food products and model systems. Review. J. Agric. Food Chem. 21, 54–60 (1973).
Sikowitz, M. D., Shome, B., Zhang, Y., Begley, T. P. & Ealick, S. E. Structure of a Clostridium botulinum C143S thiaminase I/thiamin complex reveals active site architecture. Biochemistry 52, 7830–7839 (2013).
Bian, J., Shen, H., Tu, Y., Yu, A. & Li, C. The riboswitch regulates a thiamine pyrophosphate ABC transporter of the oral spirochete Treponema denticola. J. Bacteriol. 193, 3912–3922 (2011).
Caimano, M. J., Drecktrah, D., Kung, F. & Samuels, D. S. Interaction of the Lyme disease spirochete with its tick vector. Cell Microbiol. 18, 919–927 (2016).
Corona, A. & Schwartz, I. Borrelia burgdorferi: carbon metabolism and the tick–mammal enzootic cycle. Microbiol. Spectr. http://dx.doi.org/10.1128/microbiolspec.MBP-0011-2014 (2015).
Gherardini, F. C. in Borrelia: Molecular Biology, Host Interaction and Pathogenesis (eds Samuels, D. S. & Radolf, J. D. ) 103–138 (Caister Academic, 2010).
Snyder, A. K., Deberry, J. W., Runyen-Janecky, L. & Rio, R. V. Nutrient provisioning facilitates homeostasis between tsetse fly (Diptera: Glossinidae) symbionts. Proc. Biol. Sci. 277, 2389–2397 (2010).
Rego, R. O., Bestor, A. & Rosa, P. A. Defining the plasmid-borne restriction-modification systems of the Lyme disease spirochete Borrelia burgdorferi. J. Bacteriol. 193, 1161–1171 (2011).
Smith, M. E. B., Kaulmann, U., Ward, J. M. & Hailes, H. C. A colorimetric assay for screening transketolase activity. Bioorg. Med. Chem. 14, 7062–7065 (2006).
Cázares, A. et al. Non-α-hydroxylated aldehydes with evolved transketolase enzymes. Org. Biomol. Chem. 8, 1301–1309 (2010).
Ge, Y. & Charon, N. W. Identification of a large motility operon in Borrelia burgdorferi by semi-random PCR chromosome walking. Gene 189, 195–201 (1997).
Elias, A. F. et al. New antibiotic resistance cassettes suitable for genetic studies in Borrelia burgdorferi. J. Mol. Microbiol. Biotechnol. 6, 29–40 (2003).
Samuels, D. S. Electrotransformation of the spirochete Borrelia burgdorferi. Methods Mol. Biol. 47, 253–259 (1995).
Elias, A. F. et al. Clonal polymorphism of Borrelia burgdorferi strain B31 MI: implications for mutagenesis in an infectious strain background. Infect. Immun. 70, 2139–2150 (2002).
Sze, C. W., Zhang, K., Kariu, T., Pal, U. & Li, C. Borrelia burgdorferi needs chemotaxis to establish infection in mammals and to accomplish its enzootic cycle. Infect. Immun. 80, 2485–2492 (2012).
Dunning Hotopp, J. C. et al. Comparative genomics of emerging human ehrlichiosis agents. PLoS Genet. 2, e21 (2006).
Wu, M. & Eisen, J. A. A simple, fast, and accurate method of phylogenomic inference. Genome Biol. 9, R151 (2008).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Stajich, J. E. et al. The Bioperl toolkit: Perl modules for the life sciences. Genome Res. 12, 1611–1618 (2002).
Enright, A. J., Van Dongen, S. & Ouzounis, C. A. An efficient algorithm for large-scale detection of protein families. Nucleic Acids Res. 30, 1575–1584 (2002).
Camacho, C. et al. BLAST+: architecture and applications. BMC Bioinformatics 10, 421 (2009).
Costelloe, S. J., Ward, J. M. & Dalby, P. A. Evolutionary analysis of the TPP-dependent enzyme family. J. Mol. Evol. 66, 36–49 (2008).
Popescu, A.-A., Huber, K. T. & Paradis, E. ape 3.0: New tools for distance-based phylogenetics and evolutionary analysis in R. Bioinformatics 28, 1536–1537 (2012).
Acknowledgements
This work was supported by the National Institutes of Health (grants DE023080 and AI078958 to C.H.L., AI107955 to W.G.Q., T34GM007823 to R.E.N., AI080615 to U.P. and DK67081 to S.E.E.). The authors thank L. Kinsland and L. Di for assistance in manuscript and figure preparations and J. Leadbetter for assistance with the discussion.
Author information
Authors and Affiliations
Contributions
K.Z., J.B., Y.D., A.S., R.E.N., M.B.L., U.P. and A.Y. conducted the experiments and data analyses. W.Q., S.E.E., and C.L. designed the experiments and prepared the manuscript. All authors contributed to the interpretation of the results and writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Figures 1 and 2, Supplementary Tables 1-4 (PDF 456 kb)
Rights and permissions
About this article
Cite this article
Zhang, K., Bian, J., Deng, Y. et al. Lyme disease spirochaete Borrelia burgdorferi does not require thiamin. Nat Microbiol 2, 16213 (2017). https://doi.org/10.1038/nmicrobiol.2016.213
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.213
This article is cited by
-
Dietary factors potentially impacting thiaminase I-mediated thiamine deficiency
Scientific Reports (2023)
-
Genetic basis of thiaminase I activity in a vertebrate, zebrafish Danio rerio
Scientific Reports (2023)
-
The unusual cell wall of the Lyme disease spirochaete Borrelia burgdorferi is shaped by a tick sugar
Nature Microbiology (2021)
-
Interactions between Borrelia burgdorferi and ticks
Nature Reviews Microbiology (2020)
-
Ixodes scapularis does not harbor a stable midgut microbiome
The ISME Journal (2018)