Abstract
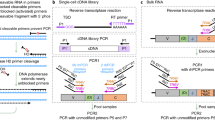
Many studies require the measurement of nucleic acid sequence diversity. Here we describe a method, called AmpliCot, that measures the sequence diversity of PCR products on the basis of DNA hybridization kinetics, thereby avoiding the time, expense and biases associated with cloning and sequencing. SYBR Green dye is used to measure DNA hybridization kinetics in a homogeneous, automated fashion. PCR products are prepared in wholly double-stranded homoduplex form for a baseline measurement of DNA concentration. The DNA is melted and then reannealed under stringent conditions that allow only homoduplexes to form. The sequence diversity of a sample is proportional to the product of its concentration and the time required for it to anneal. After validating AmpliCot with a library of diverse sequences, we use it to measure the diversity of expressed rearrangements of the gene encoding the T-cell antigen receptor (TCR) β chain. AmpliCot measurements are in good agreement with previous estimates of murine TCR repertoire diversity that required extensive cloning and sequencing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Britten, R.J. & Kohne, D.E. Repeated sequences in DNA. Science 161, 529–540 (1968).
Casrouge, A. et al. Size estimate of the αβ TCR repertoire of naive mouse splenocytes. J. Immunol. 164, 5782–5787 (2000).
Thompson, J.R., Marcelino, L.A. & Polz, M.F. Heteroduplexes in mixed-template amplifications: formation, consequence and elimination by 'reconditioning PCR'. Nucleic Acids Res. 30, 2083–2088 (2002).
Pannetier, C. et al. The sizes of the CDR3 hypervariable regions of the murine T-cell receptor β chains vary as a function of the recombined germ-line segments. Proc. Natl. Acad. Sci. USA 90, 4319–4323 (1993).
Pannetier, C., Even, J. & Kourilsky, P. T-cell repertoire diversity and clonal expansions in normal and clinical samples. Immunol. Today 16, 176–181 (1995).
Arstila, T.P. et al. A direct estimate of the human αβ T cell receptor diversity. Science 286, 958–961 (1999).
Garboczi, D.N. et al. Structure of the complex between human T-cell receptor, viral peptide and HLA-A2. Nature 384, 134–141 (1996).
Garcia, K.C. et al. An αβ T cell receptor structure at 2.5 Å and its orientation in the TCR-MHC complex. Science 274, 209–219 (1996).
Suzuki, M.T. & Giovannoni, S.J. Bias caused by template annealing in the amplification of mixtures of 16S rRNA genes by PCR. Appl. Environ. Microbiol. 62, 625–630 (1996).
Mathieu-Daude, F., Welsh, J., Vogt, T. & McClelland, M. DNA rehybridization during PCR: the 'Cot effect' and its consequences. Nucleic Acids Res. 24, 2080–2086 (1996).
Ririe, K.M., Rasmussen, R.P. & Wittwer, C.T. Product differentiation by analysis of DNA melting curves during the polymerase chain reaction. Anal. Biochem. 245, 154–160 (1997).
Shen, D.F., Doukhan, L., Kalams, S. & Delwart, E. High-resolution analysis of T-cell receptor β-chain repertoires using DNA heteroduplex tracking. J. Immunol. Methods 215, 113–121 (1998).
Yguerabide, J. & Ceballos, A. Quantitative fluorescence method for continuous measurement of DNA hybridization kinetics using a fluorescent intercalator. Anal. Biochem. 228, 208–220 (1995).
Britten, R.J. & Davidson, E.H. Hybridization strategy. In Nucleic Acid Hybridisation: a Practical Approach (eds. Hames, B.D. & Higgins, S.J.) 3–15 (IRL, Oxford, 1985).
Gilfillan, S., Dierich, A., Lemeur, M., Benoist, C. & Mathis, D. Mice lacking TdT: mature animals with an immature lymphocyte repertoire. Science 261, 1175–1178 (1993).
Cabaniols, J.P., Fazilleau, N., Casrouge, A., Kourilsky, P. & Kanellopoulos, J.M. Most α/β T cell receptor diversity is due to terminal deoxynucleotidyl transferase. J. Exp. Med. 194, 1385–1390 (2001).
Arstila, T.P. et al. Diversity of human αβ T cell receptors. Science 288, 1135 (2000).
Soen, Y., Chen, D.S., Kraft, D.L., Davis, M.M. & Brown, P.O. Detection and characterization of cellular immune responses using peptide-MHC microarrays. PLoS Biol. 1, 429–438 (2003).
Wagner, U.G., Koetz, K., Weyand, C.M. & Goronzy, J.J. Perturbation of the T cell repertoire in rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 95, 14447–14452 (1998).
Naylor, K. et al. The influence of age on T cell generation and TCR diversity. J. Immunol. 174, 7446–7452 (2005).
Ogle, B.M. et al. Direct measurement of lymphocyte receptor diversity. Nucleic Acids Res. 31, e139 (2003).
Cohn, M. & Langman, R.E. The immune system: a look from a distance. Front. Biosci. 1, 318–323 (1996).
Halpin, D.R. & Harbury, P.B. DNA display I. Sequence-encoded routing of DNA populations. PLoS Biol. 2, E173 (2004).
Ellington, A.D. & Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990).
Adleman, L.M. Molecular computation of solutions to combinatorial problems. Science 266, 1021–1024 (1994).
Glusman, G. et al. Comparative genomics of the human and mouse T cell receptor loci. Immunity 15, 337–349 (2001).
Acknowledgements
We thank C. Stoddart, T. Liegler and K. Shokat for use of equipment; G. Chkhenkeli and N. Sapitan for assistance with mice; P. Harbury, M. Chamberlin, L. Hood, R. Britten and members of The J. David Gladstone Institutes for discussions; and J. Oksenberg, E. Delwart, C. Barker and laboratory colleagues for comments on the manuscript. The work was supported in part by grants from the US National Institutes of Health (including AI47062 and AI43864 to J.M.M.), and was conducted while P.B. was a Pfizer Postdoctoral Fellow. J.M.M. is a recipient of the Burroughs Wellcome Fund Clinical Scientist Award in Translational Research and the NIH Director's Pioneer Award Program, part of the NIH Roadmap for Medical Research (funded through grant number DPI OD00329).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
T-cell receptor mRNA expression. (PDF 357 kb)
Supplementary Fig. 2
Interassay variability. (PDF 226 kb)
Rights and permissions
About this article
Cite this article
Baum, P., McCune, J. Direct measurement of T-cell receptor repertoire diversity with AmpliCot. Nat Methods 3, 895–901 (2006). https://doi.org/10.1038/nmeth949
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth949
This article is cited by
-
Longitudinal analysis of peripheral blood T cell receptor diversity in patients with systemic lupus erythematosus by next-generation sequencing
Arthritis Research & Therapy (2015)
-
Methods for diversity and overlap analysis in T-cell receptor populations
Journal of Mathematical Biology (2013)
-
Estimating diversity, the easy way
Nature Methods (2006)