Abstract
We combine the labeling of newly transcribed RNAs with 5-ethynyluridine with the characterization of bound proteins. This approach, named capture of the newly transcribed RNA interactome using click chemistry (RICK), systematically captures proteins bound to a wide range of RNAs, including nascent RNAs and traditionally neglected nonpolyadenylated RNAs. RICK has identified mitotic regulators amongst other novel RNA-binding proteins with preferential affinity for nonpolyadenylated RNAs, revealed a link between metabolic enzymes/factors and nascent RNAs, and expanded the known RNA-bound proteome of mouse embryonic stem cells. RICK will facilitate an in-depth interrogation of the total RNA-bound proteome in different cells and systems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Castello, A. et al. Insights into RNA biology from an atlas of mammalian mRNA-binding proteins. Cell 149, 1393–1406 (2012).
Baltz, A.G. et al. The mRNA-bound proteome and its global occupancy profile on protein-coding transcripts. Mol. Cell 46, 674–690 (2012).
Yang, L., Duff, M.O., Graveley, B.R., Carmichael, G.G. & Chen, L.L. Genomewide characterization of non-polyadenylated RNAs. Genome Biol. 12, R16 (2011).
Wu, Q. et al. Poly A- transcripts expressed in HeLa cells. PLoS One 3, e2803 (2008).
Cheng, J. et al. Transcriptional maps of 10 human chromosomes at 5-nucleotide resolution. Science 308, 1149–1154 (2005).
Jao, C.Y. & Salic, A. Exploring RNA transcription and turnover in vivo by using click chemistry. Proc. Natl. Acad. Sci. USA 105, 15779–15784 (2008).
Zhang, X.O. et al. Complementary sequence-mediated exon circularization. Cell 159, 134–147 (2014).
Core, L.J., Waterfall, J.J. & Lis, J.T. Nascent RNA sequencing reveals widespread pausing and divergent initiation at human promoters. Science 322, 1845–1848 (2008).
Rahl, P.B. et al. c-Myc regulates transcriptional pause release. Cell 141, 432–445 (2010).
Lai, F., Gardini, A., Zhang, A. & Shiekhattar, R. Integrator mediates the biogenesis of enhancer RNAs. Nature 525, 399–403 (2015).
Wang, D. et al. Reprogramming transcription by distinct classes of enhancers functionally defined by eRNA. Nature 474, 390–394 (2011).
Glažar, P., Papavasileiou, P. & Rajewsky, N. circBase: a database for circular RNAs. RNA 20, 1666–1670 (2014).
Memczak, S. et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 495, 333–338 (2013).
Liu, L. et al. Transcriptional pause release is a rate-limiting step for somatic cell reprogramming. Cell Stem Cell 15, 574–588 (2014).
Liu, P. et al. Release of positive transcription elongation factor b (P-TEFb) from 7SK small nuclear ribonucleoprotein (snRNP) activates hexamethylene bisacetamide-inducible protein (HEXIM1) transcription. J. Biol. Chem. 289, 9918–9925 (2014).
ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012).
Andersson, R. et al. An atlas of active enhancers across human cell types and tissues. Nature 507, 455–461 (2014).
Beckmann, B.M. et al. The RNA-binding proteomes from yeast to man harbour conserved enigmRBPs. Nat. Commun. 6, 10127 (2015).
Conrad, T. et al. Serial interactome capture of the human cell nucleus. Nat. Commun. 7, 11212 (2016).
Alexandrov, A., Martzen, M.R. & Phizicky, E.M. Two proteins that form a complex are required for 7-methylguanosine modification of yeast tRNA. RNA 8, 1253–1266 (2002).
Xiao, W. et al. Nuclear m(6)A reader YTHDC1 regulates mRNA splicing. Mol. Cell 61, 507–519 (2016).
Hafner, M. et al. Transcriptome-wide identification of RNA-binding protein and microRNA target sites by PAR-CLIP. Cell 141, 129–141 (2010).
Okamoto, H. et al. Substrate tRNA recognition mechanism of tRNA (m7G46) methyltransferase from Aquifex aeolicus. J. Biol. Chem. 279, 49151–49159 (2004).
Leulliot, N. et al. Structure of the yeast tRNA m7G methylation complex. Structure 16, 52–61 (2008).
Yang, E. et al. Decay rates of human mRNAs: correlation with functional characteristics and sequence attributes. Genome Res. 13, 1863–1872 (2003).
Bao, X. et al. The p53-induced lincRNA-p21 derails somatic cell reprogramming by sustaining H3K9me3 and CpG methylation at pluripotency gene promoters. Cell Res. 25, 80–92 (2015).
Mews, P. et al. Acetyl-CoA synthetase regulates histone acetylation and hippocampal memory. Nature 546, 381–386 (2017).
Roundtree, I.A., Evans, M.E., Pan, T. & He, C. Dynamic rna modifications in gene expression regulation. Cell 169, 1187–1200 (2017).
Kwon, S.C. et al. The RNA-binding protein repertoire of embryonic stem cells. Nat. Struct. Mol. Biol. 20, 1122–1130 (2013).
Wong, D.J. et al. Module map of stem cell genes guides creation of epithelial cancer stem cells. Cell Stem Cell 2, 333–344 (2008).
Ben-Porath, I. et al. An embryonic stem cell-like gene expression signature in poorly differentiated aggressive human tumors. Nat. Genet. 40, 499–507 (2008).
Bhattacharya, B. et al. Gene expression in human embryonic stem cell lines: unique molecular signature. Blood 103, 2956–2964 (2004).
Ivanova, N. et al. Dissecting self-renewal in stem cells with RNA interference. Nature 442, 533–538 (2006).
Lu, X. et al. SON connects the splicing-regulatory network with pluripotency in human embryonic stem cells. Nat. Cell Biol. 15, 1141–1152 (2013).
Ho, L. et al. An embryonic stem cell chromatin remodeling complex, esBAF, is essential for embryonic stem cell self-renewal and pluripotency. Proc. Natl. Acad. Sci. USA 106, 5181–5186 (2009).
Hu, G. et al. A genome-wide RNAi screen identifies a new transcriptional module required for self-renewal. Genes Dev. 23, 837–848 (2009).
Kanakkanthara, A. et al. Cyclin A2 is an RNA binding protein that controls Mre11 mRNA translation. Science 353, 1549–1552 (2016).
Brannan, K.W. et al. SONAR discovers RNA-binding proteins from analysis of large-scale protein-protein interactomes. Mol. Cell 64, 282–293 (2016).
Liao, Y. et al. ThecCardiomyocyte RNA-binding proteome: links to intermediary metabolism and heart disease. Cell Rep. 16, 1456–1469 (2016).
He, C. et al. High-resolution mapping of RNA-binding regions in the nuclear proteome of embryonic stem cells. Mol. Cell 64, 416–430 (2016).
Ying, Q.L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008).
Langmead, B. & Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Hutchins, A.P., Jauch, R., Dyla, M. & Miranda-Saavedra, D. glbase: a framework for combining, analyzing and displaying heterogeneous genomic and high-throughput sequencing data. Cell Regen. (Lond.) 3, 1 (2014).
Trapnell, C., Pachter, L. & Salzberg, S.L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009).
Corcoran, D.L. et al. PARalyzer: definition of RNA binding sites from PAR-CLIP short-read sequence data. Genome Biol. 12, R79 (2011).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 38, 576–589 (2010).
Acknowledgements
We thank all other members of the Laboratory of RNA, Chromatin, and Human Disease for their support, and we also thank R. Johnson (University of Bern, Switzerland), X. Zhang (Institute of Biophysics, Chinese Academy of Sciences, Beijing), L.-L. Chen (Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences, Shanghai), Y. Zhou (Wuhan University, Wuhan), M. Tortorella, and Q. Zheng (Guangzhou Institutes of Biomedicine and Health, Guangzhou), Y. Xu (Sun Yat-sen University, Guangzhou), and P. Liu (Anhui University, Hefei) for their expert comments and technical support. C.W. is supported by Zhujiang Talent-Overseas Postdoc Funding Grant, M.J.K. and S.K. are supported by CAS President's International Fellowships, and M.T. is supported by CAS-TWAS President's PhD Fellowship. This work was supported by the National Key Research and Development Program of China (2016YFA0100701 to X.B., 2016YFA0100102 to M.A.E., 2016YFA0100301 to B.Q., and 2017YFA0504400 to J.Y.), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA16030502 to M.A.E.), the Pearl River Science and Technology Nova Program of Guangzhou (201610010107 to X.B., 201610010053 to W.W.), the Youth Innovation Promotion Association of the Chinese Academy of Sciences (2015294) to X.B.; the Cooperation Program of the Research Grants Council (RGC) of Hong Kong and the National Natural Science Foundation of China (NSFC; 81261160506 to M.A.E.); the NSFC (31371513 and 31671537 to M.A.E., 91440115 to B.Z., 81401909 to M.Y.), and 91440110 to J.Y., the General Research Funds from the RGC of Hong Kong (14116014 to H.W. 14113514 to H.S.), the Focused Innovations Scheme (Scheme B: 1907307 to H.S.), the RGC Collaborative Research Fund from RGC (C6015-14G to H.S. and H.W.), the Bureau of Science, Technology and Information of Guangzhou Municipality (2012J5100040 and 201508030027 to M.A.E.); the Natural Science Foundation of Guangdong Province (2014A030312001 to M.A.E., 2016A050503037 and 2015A030308007 to B.Q.); the Strategic Emerging Industry Key Technology Project of Guangdong Province (2012A080800006 to B.Z.), the Shenzhen Peacock Team Project (KQTD2015033117210153 to Nan L.); the Introduced Innovative R&D Team Program of Guangdong Province (201001Y0104789252 to B.Z.), the NIH Grant (R00CA175290) and the Cancer Prevention Research Institute of Texas First-Time Tenure-Track Faculty Recruitment Grant (RR140071) to Y.C. The Guangdong Provincial Key Laboratory of Stem Cell and Regenerative Medicine is supported by the Science and Technology Planning Project of Guangdong Province, China (2017B030314056).
Author information
Authors and Affiliations
Contributions
X.B. and M.A.E. conceived the idea, and X.G. contributed to the idea; X.B. and X.G. performed most of the experiments; M.Y. contributed to the experiments; M.T., Yiwei L., S.K., J.Z., Na L., Yuan L., C.P.-Q., X.W., L.J., M.J.K., X.Z., Z.L., C.S., D.-H.L., X.L., W.W., M.H., Y.-L.L., C.W., T.W., G.Z., D.W., J.Y., Y.C., C.Z., R.J., Y.-G.Y., Y.W., B.Q., M.-L.A., A.P.H., H.S., and X.-D.F. provided technical support, relevant advice, or performed computational analyses; Nan L., B.Q., H.W., and B.Z. provided infrastructural support; X.B., B.Z., and M.A.E. provided most of the financial support and supervised the project; X.B., X.G., and M.A.E. wrote the manuscript. X.B., B.Z., and M.A.E. approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 EU labeling and protein capture using RICK.
a, Schematic showing the conjugation of a biotin-azide to the nucleoside analog EU (incorporated into RNA) by click chemistry; Cu2+ and sodium L-ascorbate were used as catalysts, and tris(3-hydroxypropyltriazolylmethyl)amine (THPTA) as ligand. b, Visualization of EU incorporation (indicated by horseradish peroxidase activity using DAB staining) in HeLa cells. RNase A and different concentrations of biotin-azide were added as indicated. Scale bar, 100 μm; 3 biologically independent experiments were performed. (c-d), Silver staining (c) and Western blotting (d) of proteins eluted from a representative RICK experiment of HeLa cells labeled with EU for 16 hours. β-ACTIN and β-TUBULIN were used as negative controls, and POLR2A, DDX5, HNRNPK, and PTBP1 as positive controls; 3 biologically independent experiments were performed.
Supplementary Figure 2 Analysis of circRNAs isolated by RICK.
a, RNA species isolated by RICK. The percentage of total known RNAs in each class was calculated according to Ensemble annotations. b, Workflow showing the methodology used for circRNA identification. c, Representative candidate circRNAs identified by RICK; 2 biologically independent experiments were performed. d, Sanger sequencing of a representative circRNA to confirm its sequence; 2 biologically independent experiments were performed.
Supplementary Figure 3 Analysis of ppRNAs and eRNAs isolated by RICK.
a, Schematic showing the calculation used to determine the Pol II TR. b, Normalized gene body and promoter density of either RICK or oligo(dT) RNA-seq peaks1; Pol II occupancy16 was used to categorize different gene classes based on their TR, TR>4 (n=5,889) and TR<4 (n=6,838). The central line is the median, the boxes indicate the upper and lower quartiles, the whiskers indicate 1.5 × interquartile range. c, Pol II ChIP-seq peaks and RICK/oligo(dT) RNA-seq peaks for the ACOX1 gene locus. d, Metagene representation of Pol II (black) occupancy and RICK (green)/oligo(dT) (blue) RNA-seq intensity at loci producing eRNAs identified by FANTOM5 database17; n=2,189. e, H3K27ac and H3K4me140, Pol II ChIP-seq peaks, and RICK/oligo(dT) RNA-seq peaks at a specific enhancer site. One representative experiment is shown; n=2 biologically independent RICK samples were sequenced and analyzed. f, RT-qPCR analysis of ppRNAs and elongated RNAs for 2 representative genes with paused Pol II in both RICK and oligo(dT) capture samples. Relative RNA levels were first normalized to the input and then to the coding DNA sequence; n=3 biologically independent experiments and data are shown as the mean ± SD. PP indicates promoter-proximal and CDS coding DNA sequence, respectively. P value is shown (Student’s t-test, two tailed).
Supplementary Figure 4 Proteins identified by RICK.
a, Correlation analysis of LC-MS/MS in 3 independent RICK experiments. ‘Correlation’ indicates the pairwise Pearson correlation of all 1,353 identified proteins, ‘high-confidence’ represents the 720 high-confidence proteins, and ‘low-confidence’ the 633 low-confidence proteins. Both X-axis and Y-axis in scatter plots are ion intensities. Curves show the intensity distribution of ‘high-confidence proteins’ (cyan) and ‘low-confidence proteins’ (red) in each experiment. Solid straight cyan and solid straight red trend lines represent linear regression of high-confidence and low-confidence proteins, respectively. b, Scatter plot showing the comparison of unique peptide counts in both control and RICK experiment groups; the circles indicate ‘high-confidence proteins’, the triangles indicate ‘low-confidence proteins’, and the squares indicate ‘background proteins’. c, Venn diagram comparing the high-confidence proteins identified by RICK, the ‘HeLa mRNA interactome’1, and our own oligo(dT) capture without EU treatment. d, Venn diagram comparing the proteins identified by oligo(dT) capture of HeLa cells cultured with or without EU for 16 hours. e, Representative Western blot detecting the capture of selected proteins identified by RICK and oligo(dT) capture with or without EU. β-ACTIN was used as a negative control, and HNRNPK, HNRNPU, PTBP1, and DDX5 as positive controls; 3 biologically independent experiments were performed.
Supplementary Figure 5 Functional characterization of proteins overlapping in RICK and oligo(dT) capture.
a, GO analysis for the 425 proteins identified by RICK and present in different oligo(dT) capture datasets1,2,18,19; the top 10 GO terms with the smallest P value in the indicated categories are shown. The number of proteins for each individual term is also shown. P value is also shown (Fisher’s exact test, one tailed). b, KEGG pathway analysis of the 425 proteins identified by RICK and present in different oligo(dT) capture datasets. The number of proteins for each individual pathway is also shown. The numbers in the parentheses are P values (Fisher’s exact test, one tailed). c, GO terms belonging to the molecular functions (MF) category that are related to transcriptional regulation in the 295 RICK-unique RBPs. The number of proteins for each individual term is also shown. d, Schematic representation of the proteasome complex, the subunits identified by RICK are shown in red (high-confidence group) or blue (low-confidence group). e, RNA binding activity of selected proteins from the 344 RICK-exclusive RBPs was detected by PAR-CLIP-biotin chemiluminescent nucleic acid detection. Proteins related to mitosis, epigenetic regulation, and others were selected. Antibodies against FLAG were used to confirm the protein size and equal loading of samples. GFP was used as a negative control, SND1 and HNRNPK as positive controls. We also used SMC2, SMC1A, SQSTM1 and CCT3 (in green), identified by our own or other oligo(dT) capture study by Baltz et al.2, as additional positive controls. Arrowheads indicate specific bands. Representative gels are shown, 3 biologically independent experiments were performed. f, Representative PAR-CLIP-biotin chemiluminescent nucleic acid detection showing the RNA binding activities of CDK1, CDK4, and HNRNPK (positive control) in the presence of different RNase T1 concentrations. Western blotting was used to confirm protein size and comparable loading of samples. Arrowheads indicate specific bands; 3 biologically independent experiments were performed.
Supplementary Figure 6 PolyA-depleted RICK identifies proteins with preferential binding to non-polyA RNAs
a, Schematic showing the polyA-depleted RICK protocol. b, RT-qPCR to confirm the polyA RNA removal efficiency. ‘Control’ indicates samples after incubation with control beads (no oligo(dT)-coating), ‘EU+/-’ indicate samples (with/without EU) after incubation with oligo(dT) beads. 18S rRNA was used as a non-polyA RNA control; n=3 biologically independent experiments and data are shown as the mean ± SD. P value is shown (Student’s t-test, two tailed). c, Representative Western blots showing the isolation of selected proteins (CDK1, CDK4, INTS9, and METTL1) by RICK and polyA-depleted RICK, but not by RICK with RNase treatment, oligo(dT) capture, or RICK without EU treatment (negative control); 3 biologically independent experiments were performed.
Supplementary Figure 7 Identification of METTL1-bound RNAs.
a, Venn diagram comparing the transcripts identified by 2 independent METTL1 PAR-CLIP sequencing experiments. b, Venn diagram comparing the transcripts identified with METTL1 PAR-CLIP sequencing and RICK RNA-seq. c, Track view of METTL1 PAR-CLIP sequencing data showing regions at the VTRNA1-2 and VTRNA1-3 loci, the EIF4G2 locus, the regions proximal to ARL5C and the regions proximal to the LOC40124240 locus. H3K27ac and Pol II16 signals are also shown; n=2 biologically independent PAR-CLIP sequencing were analyzed. d, Comparison of the efficiency of reverse transcription of the selected oligo(dT)-captured mRNAs using random hexamers or oligo(dT) primers; 2 pairs of amplification primers for each individual gene were used for qPCR; n=3 biologically independent experiments and data are shown as the mean ± SD. e, Semi-quantitative PCR analysis of selected METTL1-bound RNAs. Random hexamers or oligo(dT) primers were used as indicated for the reverse transcription; n=2 biologically independent experiments were performed. f, METTL1 binding motifs on non-tRNA (n=25,751 and 20,367 for Rep1 and Rep2, respectively) or tRNA groups (n=1,022 and 858 for Rep1 and Rep2, respectively) identified in 2 independent PAR-CLIP sequencing experiments. P value is shown (Fisher’s exact test, one tailed). g, Representative METTL1-bound tRNAs are shown; the ‘UUCG’ core sequence on the T arm are marked with circles.
Supplementary Figure 8 Identification of CDK1-binding RNAs.
a, Distribution of RNA species identified by CDK1 PAR-CLIP sequencing. The average of n=2 biologically independent experiments is shown. b, Venn diagram comparing the transcripts identified with CDK1 PAR-CLIP sequencing and RICK RNA-seq. c, Track view of CDK1 PAR-CLIP sequencing data showing regions proximal to AGBL5 (left panel) and at an intergenic locus (right panel); n=2 biologically independent PAR-CLIP sequencing were analyzed. d, RIP-qPCR analysis of different RNAs binding to CDK1 identified in the PAR-CLIP sequencing. The enrichments were normalized to input; GFP was used as negative control. Random hexamers or oligo(dT) primers were used for the reverse transcription as indicated. CDK1-interacting mRNAs are marked in grey as opposed to intergenic RNAs; n=3 biologically independent experiments and data are shown as the mean ± SD. P value is shown (Student’s t-test, two tailed).
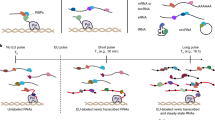
Supplementary Figure 9 Capture of the nascent-enriched RNA interactome in HeLa cells.
a, Schematic diagram showing procedures of 16-hour RICK and short labeling RICK. b, Silver staining of proteins eluted from representative RICK experiments of HeLa cells with short EU labeling times; n=3 biologically independent experiments were performed. c, Relative levels of RNA and protein isolated by RICK with short EU labeling time points compared to 16-hour RICK; n=3 biologically independent experiments and data are shown as the mean ± SD. d, Distribution of RNA species isolated in RICK experiments with short EU labeling times (0.5, 1, and 2 h); One experiment was performed; h indicates hour. e, Representative Western blot confirming the capture of 2 selected proteins identified by RICK with short EU labeling times in contrast to oligo(dT) capture with or without EU. PTBP1 and HNRNPK were detected in both short labeling RICK and oligo(dT) capture samples. β-ACTIN was used as a negative control; 3 biologically independent experiments were performed. f, Venn diagram comparing the 208 nascent-enriched RBPs with ‘HeLa mRNA interactome’ and our oligo(dT) capture in HeLa with or without EU (left); Venn diagram comparing the 43 nascent-enriched RBPs with the 295 RICK-unique RBPs (right). g, GO analysis (biological process) for the 43 nascent-specific RBPs; P value for the indicated categories is shown. (Fisher’s exact test, one tailed). h, Metabolic enzymes/factors isolated by short labeling RICK and absent in the ‘HeLa mRNA interactome’1 or our own oligo(dT) capture dataset.
Supplementary Figure 10 Application of RICK to mESCs.
a, Visualization of EU incorporation (indicated by horseradish peroxidase activity) in mESCs. Scale bar, 100 μm; n=2 biologically independent experiments were performed. b, Venn diagram comparing the RICK-identified RBPs in mESCs with the ‘mESC mRNA interactome’. c, GO analysis of the RICK-exclusive mESC RBPs; P value in the indicated categories is shown (Fisher’s exact test, one tailed). d, Venn diagram comparing the RICK-exclusive mESC RBPs with the ‘ESC-specific gene sets’29–32; The ‘ESC-specific gene sets’ is a combination of different gene sets in human embryonic stem cell and mESC studies. e, Venn diagram comparing the RICK-exclusive mESC RBPs with RBR-ID-identified proteins.
Supplementary Figure 11 Raw gels for all figures in the manuscript.
All the gels used in this manuscript have been repeated independently at least 3 times in our laboratory. This figure shows the original scans for all of the representative gels.
Supplementary Figure 12 Raw gels for all figures in the manuscript.
All the gels used in this manuscript have been repeated independently at least 3 times in our laboratory. This figure shows the original scans for all of the representative gels.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12, Supplementary Note 1 and Supplementary Table 7 (PDF 3184 kb)
Supplementary Protocol
Capture of the newly transcribed RNA interactome using click chemistry (PDF 278 kb)
Supplementary Table 1
Distribution of different types of RNA captured by RICK (XLSX 15 kb)
Supplementary Table 2
CircRNAs identified by RICK and oligo(dT) capture (XLSX 308 kb)
Supplementary Table 3
Characterization of proteins isolated by RICK (XLSX 298 kb)
Supplementary Table 4
Analysis of RICK-identified RBPs (XLSX 1280 kb)
Supplementary Table 5
Proteins identified by polyA-depleted RICK (XLSX 645 kb)
Supplementary Table 6
Short-labeling RICK time course and 16-hour RICK in mESCs (XLSX 870 kb)
Supplementary Table 8
List of primers and antibodies (XLSX 17 kb)
Supplementary Table 9
Statistics source data for Main Figures (XLSX 13 kb)
Supplementary Table 10
Statistics source data for Supplementary Figures (XLSX 19 kb)
Rights and permissions
About this article
Cite this article
Bao, X., Guo, X., Yin, M. et al. Capturing the interactome of newly transcribed RNA. Nat Methods 15, 213–220 (2018). https://doi.org/10.1038/nmeth.4595
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.4595
This article is cited by
-
Structure-based prediction and characterization of photo-crosslinking in native protein–RNA complexes
Nature Communications (2024)
-
RNA-binding proteins in cardiovascular biology and disease: the beat goes on
Nature Reviews Cardiology (2024)
-
RBP–RNA interactions in the control of autoimmunity and autoinflammation
Cell Research (2023)
-
A widely applicable and cost-effective method for specific RNA–protein complex isolation
Scientific Reports (2023)
-
The RNA-binding protein landscapes differ between mammalian organs and cultured cells
Nature Communications (2023)