Abstract
Multiple adult tissues are maintained by stem cells of restricted developmental potential which can only form a subset of lineages within the tissue. For instance, the two adult lung epithelial compartments (airways and alveoli) are separately maintained by distinct lineage-restricted stem cells. A challenge has been to obtain multipotent stem cells and/or progenitors that can generate all epithelial cell types of a given tissue. Here we show that mouse Sox9+ multipotent embryonic lung progenitors can be isolated and expanded long term in 3D culture. Cultured Sox9+ progenitors transcriptionally resemble their in vivo counterparts and generate both airway and alveolar cell types in vitro. Sox9+ progenitors that were transplanted into injured adult mouse lungs differentiated into all major airway and alveolar lineages in vivo in a region-appropriate fashion. We propose that a single expandable embryonic lung progenitor population with broader developmental competence may eventually be used as an alternative for region-restricted adult tissue stem cells in regenerative medicine.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kopp, J.L., Grompe, M. & Sander, M. Stem cells versus plasticity in liver and pancreas regeneration. Nat. Cell Biol. 18, 238–245 (2016).
Hogan, B.L. et al. Repair and regeneration of the respiratory system: complexity, plasticity, and mechanisms of lung stem cell function. Cell Stem Cell 15, 123–138 (2014).
Rock, J.R. & Hogan, B.L. Epithelial progenitor cells in lung development, maintenance, repair, and disease. Annu. Rev. Cell Dev. Biol. 27, 493–512 (2011).
Wansleeben, C., Barkauskas, C.E., Rock, J.R. & Hogan, B.L. Stem cells of the adult lung: their development and role in homeostasis, regeneration, and disease. Wiley Interdiscip. Rev. Dev. Biol. 2, 131–148 (2013).
Barkauskas, C.E. et al. Type 2 alveolar cells are stem cells in adult lung. J. Clin. Invest. 123, 3025–3036 (2013).
Rock, J.R. et al. Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc. Natl. Acad. Sci. USA 106, 12771–12775 (2009).
Desai, T.J., Brownfield, D.G. & Krasnow, M.A. Alveolar progenitor and stem cells in lung development, renewal and cancer. Nature 507, 190–194 (2014).
Rawlins, E.L. et al. The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell 4, 525–534 (2009).
Zuo, W. et al. p63(+)Krt5(+) distal airway stem cells are essential for lung regeneration. Nature 517, 616–620 (2015).
Vaughan, A.E. et al. Lineage-negative progenitors mobilize to regenerate lung epithelium after major injury. Nature 517, 621–625 (2015).
Rosen, C. et al. Preconditioning allows engraftment of mouse and human embryonic lung cells, enabling lung repair in mice. Nat. Med. 21, 869–879 (2015).
Rawlins, E.L., Clark, C.P., Xue, Y. & Hogan, B.L. The Id2+ distal tip lung epithelium contains individual multipotent embryonic progenitor cells. Development 136, 3741–3745 (2009).
Rockich, B.E. et al. Sox9 plays multiple roles in the lung epithelium during branching morphogenesis. Proc. Natl. Acad. Sci. USA 110, E4456–E4464 (2013).
Morrisey, E.E. & Hogan, B.L. Preparing for the first breath: genetic and cellular mechanisms in lung development. Dev. Cell 18, 8–23 (2010).
Chan, H.Y. et al. Comparison of IRES and F2A-based locus-specific multicistronic expression in stable mouse lines. PLoS One 6, e28885 (2011).
Cardoso, W.V. & Lü, J. Regulation of early lung morphogenesis: questions, facts and controversies. Development 133, 1611–1624 (2006).
Boroviak, T., Loos, R., Bertone, P., Smith, A. & Nichols, J. The ability of inner-cell-mass cells to self-renew as embryonic stem cells is acquired following epiblast specification. Nat. Cell Biol. 16, 516–528 (2014).
Boroviak, T. et al. Lineage-specific profiling delineates the emergence and progression of naive pluripotency in mammalian embryogenesis. Dev. Cell 35, 366–382 (2015).
You, Y., Richer, E.J., Huang, T. & Brody, S.L. Growth and differentiation of mouse tracheal epithelial cells: selection of a proliferative population. Am. J. Physiol. Lung Cell. Mol. Physiol. 283, L1315–L1321 (2002).
Nikolic´, M.Z. et al. Human embryonic lung epithelial tips are multipotent progenitors that can be expanded in vitro as long-term self-renewing organoids. eLife 6, e26575 (2017).
Nichane, M. et al. 3D culture of multipotent Sox9+ mouse embryonic lung progenitors: isolation, expansion and cryopreservation. Protocol Exchange https://doi.org/10.1038/protex.2017.106 (2017).
Picelli, S. et al. Full-length RNA-seq from single cells using Smart-seq2. Nat. Protoc. 9, 171–181 (2014).
Acknowledgements
We thank K. Chee, D.A. Silva and N. Talib (SIgN FACS Core); J. Cheng and H.Y. Ng (IMCB Histology Core); S. Lin and G. Wright (IMB Microscopy Unit); K.S. Keyau (Singapore General Hospital); and other A*STAR core facilities for infrastructure support. We also thank A. Bhinge, S.-C. Ng and Z. Wang for discussion; S. Namboori, E. Lim, S.H. Choo, S.P. Guan and J. Lee for technical assistance; and R. Ettikan for logistical support. This work was supported by the Singapore National Medical Research Council (NMRC/TCR/007-NCC/2013), the Singapore Agency for Science, Technology & Research (A*STAR) (1331AEG071; 1334I00052; 1334I00053; SPF 2012/001; H16/99/b0/003) (to B.L.) and the Stanford-UC Berkeley Siebel Stem Cell Institute (to K.M.L.).
Author information
Authors and Affiliations
Contributions
M.N. conceptualized this work; M.N. developed methodology; M.N. and M.G. carried out investigation; A.J. conducted formal analysis; A.J., V.S. and M.N. curated data; V.S., A.J. and M.N. carried out visualization; M.N. and K.M.L. wrote the original draft; M.N., A.J., V.S., K.M.L, L.T.A. and B.L. reviewed and edited the manuscript; B.L. and K.M.L. acquired funds; V.S., L.T.A., K.M.L., T.L. and P.K. provided resources; M.N. and B.L. supervised this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Isolation and characterization of Sox9+ embryonic lung progenitors.
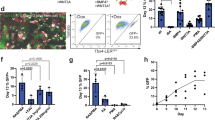
(a) FACS gating strategy used to isolate Sox9+ progenitors from E12.5 Sox9GFP embryonic lungs. Viable cells (DAPI−) from singlets were subjected to negative sorting in order to remove endothelial (Pecam1), hematopoietic (Ptprc) and red blood cells (Ter119) with e450 conjugated specific antibodies. Sox9GFP+ cells were further selected followed by Cdh1-e660 sorting in order to discriminate between epithelial and nonepithelial cells. Double positive Sox9GFP+ Cdh1+ cells are the distal tip lung progenitors while other populations reflect the nonprogenitor epithelial fraction (Sox9GFP− Cdh1+) or a mesenchymal cell subtype (Sox9GFP+ Cdh1−). (b) Growth curve of early (P2, >30 d) or late (P15, >180 d) passage in vitro-cultured Sox9+ lung progenitors from day 0 to day 10. Initial cell seeding: 1000. (c) Representative flow cytometry profile of in vitro-cultured Sox9+ progenitors derived from WT Cdh1+ or Sox9GFP+ Cdh1+ E12.5 embryonic lungs. (d) Quantification of Edu+ cells of in vitro-cultured Sox9+ progenitors (P2, >30 d; P10, >120 d and P15, >180 d). (e) Representative karyotype of Sox9+ progenitors after long-term culture (P10, >120 d). 3 independent lines were analyzed. Average of n = 4 independent experiments (b,d). Data are mean ± s.d. (b,d).
Supplementary Figure 2 Gene expression profile of in vitro-cultured Sox9+ progenitors.
(a) Heatmap (qPCR) of selected airway, alveolar, endothelial and mesenchymal markers expression. Fold change (log2) relative to total E12.5 embryonic lungs control is shown. (b) Heatmap (qPCR) of selected markers from various adult and embryonic tissues. Fold change relative to whole E14.5 embryos is shown. Average of n = 4 (a) or n = 2 (b) independent experiments.
Supplementary Figure 3 Effect of LPM-3D medium components withdrawal.
(a) Growth curve of Sox9+ progenitors cultured in complete medium (LPM-3D) or in media where one component was removed (cell analyzed at P0, >10 d). Initial cell seeding: 1,000. (b-f) Gene expression profile (qPCR) of selected lung progenitor (b), airway (c,d), and alveolar (e,f) markers at P0 (>10 d; b,c,e) or prolonged culture (P3, >40 d; d,f) under the indicated culture conditions. Fold change relative to Sox9+ progenitors cultured with complete LPM-3D medium is shown. Average of n = 4 independent experiments (a-f). Data are mean ± s.d. (a-f). Adjusted P Value (Two-way ANOVA) *, P < 0.01; **, P < 0.02; ***, P < 0.05; n.s., not significant (b-f).
Supplementary Figure 4 Effect of cell density modulation on in vitro-cultured Sox9+ progenitors.
(a) Growth curve of Sox9+ progenitors with different initial cell seeding (300 to 6,000). (b) Relative total number of Sox9+ progenitor colonies per 1,000 cells seeded (seeding: 300 to 6,000). (c) Quantification of Sox9GFP+ cells in in vitro-cultured Sox9+ progenitors with variable cell density (seeding: 1,000 to 60,000). (d) Gene expression profile (qPCR) of selected lung progenitor markers and Cdh1 (seeding: 1,000 to 60,000). Fold change relative to total E12.5 embryonic lungs control is shown. (e) Gene expression profile (qPCR) of selected lung progenitor markers, Nkx2.1, Cdh1, GFP and tdTomato comparing in vitro-cultured lung progenitor colonies derived from wild-type (WT) Cdh1+, tdTomato+ Cdh1+, eGFP+ Cdh1+ and Sox9GFP+ Cdh1+ E12.5 embryonic lungs. Fold change relative to total E12.5 embryonic lungs control is shown. (f-h) Immunostaining of colonies derived from WT Cdh1+ (f), eGFP+ Cdh1+ (g) and tdTomato+ Cdh1+ (h) E12.5 embryonic lungs with Sox9 lung progenitor marker. DAPI with either eGFP (g) or tdTomato (h) fluorescence is shown in bottom-right insets. Scale bars = 10 μm. (i) Relative percentage of eGFP+ (green), tdTomato+ (red) or mixed (dark blue) colonies in which Cdh1+ cells from eGFP+ or tdTomato+ E12.5 embryonic lungs were mixed at the indicated ratios. Initial cell seeding: 3,000. Average of n = 4 independent experiments (a-e,i). Data are mean ± s.d. (a-e). Adjusted P Value (Two-way ANOVA) *, P < 0.01; ***, P < 0.05 (d,e).
Supplementary Figure 5 Transcriptome of in vitro-cultured Sox9+ progenitors.
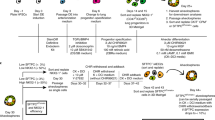
(a) Schematic of the experimental strategy used for whole-transcriptome comparison by Microarray. Unsupervised hierarchical clustering of in vitro-cultured Sox9+ lung progenitors at early (P2) and late (P10) passages with in vivo Sox9+ embryonic lung progenitors, adult lung epithelial (Cdh1+ and EpCAM+) and non-epithelial (Cdh1− and EpCAM−) cells is shown. (b) Three-dimensional t-SNE plot of RNA-seq data. Different colors and different shapes represent in vitro-cultured Sox9+ progenitors (blue squares), in vivo E12.5 embryonic lung progenitors (derived from Sox9GFP+ Cdh1+ Pecam1− Ptrprc− Ter119−, green circles), adult lung epithelial cells (red triangles) and adult lung non-epithelial cells (yellow triangles). (c,d) Pairwise correlation plots from RNA-seq data comparing all the genes detected between in vitro-cultured Sox9+ progenitors with either adult lung Cdh1+/EpCAM+ epithelial (c) or Cdh1−/EpCAM− non-epithelial (d) samples. Raw data values were normalized and are shown as rlog (see Online Methods). Solid red line indicates x = y and dashed line ≥2-fold change in either direction. Representative genes showing differential expression are shown. Range of correlation value (R) from Fig. 3b are indicated.
Supplementary Figure 6 In vitro differentiation of Sox9+ progenitors.
(a) Gene expression profile (qPCR) of selected lung progenitor markers comparing Sox9+ progenitors grown in the indicated conditions. Fold change relative to Sox9+ progenitors cultured in LPM-3D is shown. (b) Quantification of Sox9GFP+ cells in Sox9+ progenitors grown in the indicated conditions. (c) Immunostaining of Sox9+ progenitors grown in LPM-3D that shows the absence of Krt5 staining. Inset: DAPI. (d) Total number of differentiated alveolo- and bronchosphere observed on day 16 in transwell assay from early (P3) and late (P11) passage in vitro-cultured Sox9+ progenitors. Initial cell seeding: 20,000. (e-g) Immunostaining of differentiated Sox9+ progenitors colonies in transwell assay for markers of type II alveolar (Sftpb+, e) and type I alveolar cells (Hopx+, f; Pdpn+, g). (h) Immunostaining of ALI-differentiated cells for marker of goblet cells (Muc5ac+). (i-k) Gene expression profile (qPCR) of selected airway (i), alveolar (j) and lung progenitor (k) markers comparing Sox9+ progenitors grown in LPM-3D or in sphere- and ALI-differentiation assay from either early (P3, >30 d) or late (P11, >120 d) passage in vitro Sox9+ progenitors. Fold change relative to Sox9+ progenitors cultured in LPM-3D is shown. Scale bars = 10 μm (c, e-g) and 20 μm (h). Average of n = 4 (a,d), n = 2 (b) or n = 3 (i-k) independent experiments. Data are mean ± s.d. (a,b,d and i-k). Adjusted P Value (Two-way ANOVA) *, P < 0.01; **, P < 0.02; ***, P < 0.05; n.s., not significant (a,i-k). One-tailed unpaired t-test *, P < 0.01; **, P < 0.02 (b).
Supplementary Figure 7 Transplantation of Sox9+ progenitors into uninjured lungs.
Cryosections of adult lungs with transplanted Sox9+ tdTomato+ progenitors and stained for the indicated markers. Transplanted Sox9+ tdTomato+ progenitors fail to engraft the uninjured mouse lung. Representative images (n = 3 uninjured lungs) of airway markers (ciliated (AcTubulin+, a,b); club (Scgb1A1+, c,d); neuroendocrine (Calca+, e); goblet (Muc5ac+, f)) and alveolar markers (Type II alveolar (Sftpc+, g,h) and Type I alveolar (Pdpn+, i,j; Hopx+, k,l)) are shown. Scale bars = 40 μm.
Supplementary Figure 8 Transplantation of Sox9+ progenitors into the naphthalene-injured mouse lung.
Cryosections of adult lungs with transplanted Sox9+ tdTomato+ progenitors after naphthalene injury and stained for the indicated markers. Transplanted Sox9+ tdTomato+ progenitors differentiate into basal (Ngfr+, a; Krt5+, b), suprabasal (Krt8+, b) cells, goblet cells (Muc5ac+, c,d) or neuroendocrine cells (Calca+, e). Representative images of n = 5 (a,b), n = 3 (c-e) transplants. Insets (a, middle subpanel; c-e) show higher magnification of dashed boxed region. Bottom-right subpanels show voxels demonstrating colocalization (white) from 3D imaris analysis of z-stacks from insets (a,e) or from dashed boxed region (b). The numeric value is the percentage of tdTomato+ volume above the threshold (percent red voxels) that is colocalized with either Ngfr (a), Krt5 and Krt8 (b) or Calca (e) signal (percent green voxels) above the threshold. White arrowheads show representative transplanted tdTomato+ Sox9+ progenitors cells coexpressing specific airway markers. Scale bars = 40 μm.
Supplementary Figure 9 Transplantation of Sox9+ progenitors into the bleomycin-injured mouse lung.
Cryosections of adult lungs with transplanted Sox9+ tdTomato+ progenitors after bleomycin injury and stained for the indicated markers. Transplanted Sox9+ tdTomato+ progenitors differentiate into Type II alveolar (Sftpb+, a) and Type I alveolar (Pdpn+, b,c and Aqp5+, d) cells. Representative images of n = 5 (a), n = 3 (b,c,d) transplants. Insets (a-d) show higher magnification of dashed boxed region. Bottom-right subpanels show voxels demonstrating colocalization (white) from 3D imaris analysis of z-stacks in a,b,d (bottom-right sub-panels). The numeric value is the percentage of tdTomato+ volume above the threshold (percent red voxels) that is colocalized with either Sftpb (a), Pdpn (b) or Aqp5 (d) signal (percent green voxels) above the threshold. White arrowheads show representative transplanted tdTomato+ Sox9+ progenitors cells coexpressing specific alveolar markers. Scale bars = 40 μm.
Supplementary Figure 10 Three-dimensional reconstruction of engrafted Sox9+ progenitors in alveolar area after bleomycin injury.
Cryosections of adult lungs with transplanted Sox9+ tdTomato+ progenitors after bleomycin injury and stained for the Type I alveolar marker Hopx. Three-dimensional reconstruction of z-stack shown in Fig. 6f at lower magnification. tdTomato (a), Hopx antibody staining (b), merge (c) and colocalized (d) channels are shown. Scale bars = 70 μm.
Supplementary Figure 11 Transplanted Sox9+ progenitors contribute exclusively to lung epithelial lineages.
Cryosections of adult lungs with transplanted Sox9+ tdTomato+ progenitors after bleomycin injury and stained for the indicated markers. Transplanted Sox9+ tdTomato+ progenitors were never colocalized with the nonepithelial markers Pecam1 (endothelial, a), Ptprc (hematopoietic, b), Acta2 (smooth muscle, c) and Vimentin (mesenchyme, d). Representative images of n = 3 transplants. Insets (a-d) show higher magnification of dashed boxed region. Bottom-right subpanel show voxels demonstrating colocalization (white) from 3D imaris analysis of z-stacks in a. The numeric value is the percentage of tdTomato+ volume above the threshold (percent red voxels) that is colocalized with Pecam1 signal (percent green voxels) above the threshold. Only 2.91% of tdTomato+ voxels coexpress Pecam1 (compared with the higher percentage of colocalization observed for Type II and Type I alveolar markers, see Fig. 6e,f and Supplementary Fig. 9). The areas of colocalization are typically observed to occur at the interface between tdTomato+ cells and neighboring Pecam1+ cells, an interface that the software cannot clearly distinguish given limitations of image resolution. Scale bars = 40 μm.
Supplementary Figure 12 Single cell qPCR of transplanted Sox9+ progenitors.
(a) Representative flow cytometry profile of adult lung transplanted with Sox9+ tdTomato+ progenitors in naphthalene and bleomycin injury models. (b) Quantification of tdTomato+ cells per adult lung after naphthalene (n = 5 transplants) and bleomycin (n = 5 transplants) injury. No tdTomato+ cells were ever observed in noninjured lungs (n = 3 transplants). One-tailed unpaired t-test *, P ≤ 0.01; ***, P < 0.05. More tdTomato+ cells per adult lung were found in bleomycin model probably due to the specific accessibility (conducting airways or alveolar areas) upon injury. (c) Heatmap showing the unsupervised hierarchical clustering of single-cell gene expression signatures from control adult lungs epithelial and nonepithelial single-cells (from wild-type (WT) or tdTomato mouse lungs) (in green), pre-transplant in vitro-cultured Sox9+ lung progenitors (in blue) and FACS-sorted tdTomato+ single-cells from transplanted lungs with Sox9+ tdTomato+ progenitors after either bleomycin (in yellow) or naphthalene (in red). Each column corresponds to a particular single-cell and each row corresponds to a specific gene. Genes representative of each differentiated lung epithelial cell type including embryonic lung progenitors as well as lung mesenchymal and endothelial cells were tested. Red to light red suggest high to middle expression (Ct value range: 5-15), whereas white to grey suggest low to no expression (Ct value range: 15-28). Single-cells pooled from n = 7 transplanted lungs, n = 2 control adult lung epithelial cells and n = 2 in vitro-cultured Sox9+ progenitors (see also Online Methods and source file Supplementary fig. 12 for more details). (d) t-SNE of single-cells analyzed in c distinguishes between major adult airway and alveolar cell lineages as well as lung nonepithelial lineages. Transplanted Sox9+ tdTomato+ progenitors are found in the Type II alveolar, club and basal cells clusters and a closely related cluster of differentiating airway cells while pre-transplant in vitro cultured Sox9+ progenitors are found in a separate cluster. No transplanted Sox9+ tdTomato+ progenitors cluster with either lung mesenchymal or endothelial cells.
Supplementary Figure 13 Engraftment specificity and nontumorigenicity of Sox9+ progenitors.
(a) Representative flow cytometry profile of adult esophagus, heart and liver from mice intratracheally transplanted with Sox9+ tdTomato+ progenitors in naphthalene and bleomycin injury models. No tdTomato+ cells were detected in these three organs (n = 3 for each tissue). (b) In vitro-cultured Sox9+ progenitors do not form tumor when injected subcutaneously. 1 million cells were injected in the right flank. n = 6 mice analyzed after 4 and 12 weeks. A representative image of a mouse with arrows indicating the injected and control sides is shown.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14, Supplementary Note 1 and Supplementary Tables 1–5 (PDF 4434 kb)
Life Sciences Reporting Summary
Life Sciences Reporting Summary (PDF 207 kb)
Supplementary Protocol
3D culture of multipotent Sox9+ mouse embryonic lung progenitors: Isolation, Expansion and Cryopreservation (PDF 1021 kb)
Source data
Rights and permissions
About this article
Cite this article
Nichane, M., Javed, A., Sivakamasundari, V. et al. Isolation and 3D expansion of multipotent Sox9+ mouse lung progenitors. Nat Methods 14, 1205–1212 (2017). https://doi.org/10.1038/nmeth.4498
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.4498
This article is cited by
-
KRAS(G12D) drives lepidic adenocarcinoma through stem-cell reprogramming
Nature (2023)
-
A Maverick Review of Common Stem/Progenitor Markers in Lung Development
Stem Cell Reviews and Reports (2022)
-
Sox9-expressing cells promote regeneration after radiation-induced lung injury via the PI3K/AKT pathway
Stem Cell Research & Therapy (2021)
-
In vitro models of fetal lung development to enhance research into congenital lung diseases
Pediatric Surgery International (2021)
-
Engraftment and proliferation potential of embryonic lung tissue cells in irradiated mice with emphysema
Scientific Reports (2019)