Abstract
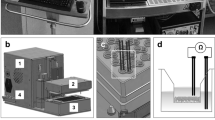
Here we describe the development of a human lung 'small airway-on-a-chip' containing a differentiated, mucociliary bronchiolar epithelium and an underlying microvascular endothelium that experiences fluid flow, which allows for analysis of organ-level lung pathophysiology in vitro. Exposure of the epithelium to interleukin-13 (IL-13) reconstituted the goblet cell hyperplasia, cytokine hypersecretion and decreased ciliary function of asthmatics. Small airway chips lined with epithelial cells from individuals with chronic obstructive pulmonary disease recapitulated features of the disease such as selective cytokine hypersecretion, increased neutrophil recruitment and clinical exacerbation by exposure to viral and bacterial infections. With this robust in vitro method for modeling human lung inflammatory disorders, it is possible to detect synergistic effects of lung endothelium and epithelium on cytokine secretion, identify new biomarkers of disease exacerbation and measure responses to anti-inflammatory compounds that inhibit cytokine-induced recruitment of circulating neutrophils under flow.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
World Health Organization. Top 10 causes of death http://www.who.int/mediacentre/factsheets/fs310/en/ (2014).
Bafadhel, M. . et al. Acute exacerbations of chronic obstructive pulmonary disease: identification of biologic clusters and their biomarkers. Am. J. Respir. Crit. Care Med. 184, 662–671 (2011).
Jackson, D.J., Sykes, A., Mallia, P. & Johnston, S.L. Asthma exacerbations: origin, effect, and prevention. J. Allergy Clin. Immunol. 128, 1165–1174 (2011).
Hyde, D.M., Hamid, Q. & Irvin, C.G. Anatomy, pathology, and physiology of the tracheobronchial tree: emphasis on the distal airways. J. Allergy Clin. Immunol. 124 (suppl. 6), S72–S77 (2009).
Kolaczkowska, E. & Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 13, 159–175 (2013).
Wright, J.L., Cosio, M. & Churg, A. Animal models of chronic obstructive pulmonary disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 295, L1–L15 (2008).
Di Stefano, A. et al. Severity of airflow limitation is associated with severity of airway inflammation in smokers. Am. J. Respir. Crit. Care Med. 158, 1277–1285 (1998).
Fahy, J.V. Eosinophilic and neutrophilic inflammation in asthma: insights from clinical studies. Proc. Am. Thorac. Soc. 6, 256–259 (2009).
Hogg, J.C. et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 350, 2645–2653 (2004).
Bueters, T., Ploeger, B.A. & Visser, S.A. The virtue of translational PKPD modeling in drug discovery: selecting the right clinical candidate while sparing animal lives. Drug Discov. Today 18, 853–862 (2013).
Fulcher, M.L., Gabriel, S., Burns, K.A., Yankaskas, J.R. & Randell, S.H. Well-differentiated human airway epithelial cell cultures. Methods Mol. Med. 107, 183–206 (2005).
Ugonna, K., Bingle, C.D., Plant, K., Wilson, K. & Everard, M.L. Macrophages are required for dendritic cell uptake of respiratory syncytial virus from an infected epithelium. PLoS ONE 9, e91855 (2014).
Didierlaurent, A. et al. Sustained desensitization to bacterial Toll-like receptor ligands after resolution of respiratory influenza infection. J. Exp. Med. 205, 323–329 (2008).
Hammad, H. et al. House dust mite allergen induces asthma via Toll-like receptor 4 triggering of airway structural cells. Nat. Med. 15, 410–416 (2009).
Teijaro, J.R. et al. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell 146, 980–991 (2011).
Langer, H.F. & Chavakis, T. Leukocyte-endothelial interactions in inflammation. J. Cell. Mol. Med. 13, 1211–1220 (2009).
Bhatia, S.N. & Ingber, D.E. Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014).
Esch, E.W., Bahinski, A. & Huh, D. Organs-on-chips at the frontiers of drug discovery. Nat. Rev. Drug Discov. 14, 248–260 (2015).
Huh, D. et al. A human disease model of drug toxicity-induced pulmonary edema in a lung-on-a-chip microdevice. Sci. Transl. Med. 4, 159ra147 (2012).
Huh, D. et al. Reconstituting organ-level lung functions on a chip. Science 328, 1662–1668 (2010).
Wanner, A., Salathe, M. & O'Riordan, T.G. Mucociliary clearance in the airways. Am. J. Respir. Crit. Care Med. 154, 1868–1902 (1996).
Fliegauf, M., Benzing, T. & Omran, H. When cilia go bad: cilia defects and ciliopathies. Nat. Rev. Mol. Cell Biol. 8, 880–893 (2007).
Livraghi, A. & Randell, S.H. Cystic fibrosis and other respiratory diseases of impaired mucus clearance. Toxicol. Pathol. 35, 116–129 (2007).
Wills-Karp, M. Interleukin-13 in asthma pathogenesis. Immunol. Rev. 202, 175–190 (2004).
Kuperman, D.A. et al. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat. Med. 8, 885–889 (2002).
Danahay, H. et al. Notch2 is required for inflammatory cytokine-driven goblet cell metaplasia in the lung. Cell Rep. 10, 239–252 (2015).
Sousa, A.R., Poston, R.N., Lane, S.J., Nakhosteen, J.A. & Lee, T.H. Detection of GM-CSF in asthmatic bronchial epithelium and decrease by inhaled corticosteroids. Am. Rev. Respir. Dis. 147, 1557–1561 (1993).
Ordoñez, C.L. et al. Mild and moderate asthma is associated with airway goblet cell hyperplasia and abnormalities in mucin gene expression. Am. J. Respir. Crit. Care Med. 163, 517–523 (2001).
Thomas, B. et al. Ciliary dysfunction and ultrastructural abnormalities are features of severe asthma. J. Allergy Clin. Immunol. 126, 722–729 (2010).
Hewson, C.A., Jardine, A., Edwards, M.R., Laza-Stanca, V. & Johnston, S.L. Toll-like receptor 3 is induced by and mediates antiviral activity against rhinovirus infection of human bronchial epithelial cells. J. Virol. 79, 12273–12279 (2005).
Curradi, G. et al. Airway basal cell vascular endothelial growth factor-mediated cross-talk regulates endothelial cell-dependent growth support of human airway basal cells. Cell. Mol. Life Sci. 69, 2217–2231 (2012).
Ding, B.S., Gomi, K., Rafii, S., Crystal, R.G. & Walters, M.S. Endothelial MMP14 is required for endothelial-dependent growth support of human airway basal cells. J. Cell Sci. 128, 2983–2988 (2015).
Lawrence, M.B. & Springer, T.A. Neutrophils roll on E-selectin. J. Immunol. 151, 6338–6346 (1993).
Mayer, A.K. et al. Differential recognition of TLR-dependent microbial ligands in human bronchial epithelial cells. J. Immunol. 178, 3134–3142 (2007).
O'Shaughnessy, T.C., Ansari, T.W., Barnes, N.C. & Jeffery, P.K. Inflammation in bronchial biopsies of subjects with chronic bronchitis: inverse relationship of CD8+ T lymphocytes with FEV1. Am. J. Respir. Crit. Care Med. 155, 852–857 (1997).
Papi, A. et al. Infections and airway inflammation in chronic obstructive pulmonary disease severe exacerbations. Am. J. Respir. Crit. Care Med. 173, 1114–1121 (2006).
Deslee, G. et al. Bronchial epithelial spheroids: an alternative culture model to investigate epithelium inflammation-mediated COPD. Respir. Res. 8, 86 (2007).
Fleischmann, R. et al. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N. Engl. J. Med. 367, 495–507 (2012).
Bush, A. et al. Severe childhood asthma: a common international approach? Lancet 372, 1019–1021 (2008).
Kudlacz, E., Conklyn, M., Andresen, C., Whitney-Pickett, C. & Changelian, P. The JAK-3 inhibitor CP-690550 is a potent anti-inflammatory agent in a murine model of pulmonary eosinophilia. Eur. J. Pharmacol. 582, 154–161 (2008).
Ammit, A.J. Glucocorticoid insensitivity as a source of drug targets for respiratory disease. Curr. Opin. Pharmacol. 13, 370–376 (2013).
Wu, J. et al. Design and chemoproteomic functional characterization of a chemical probe targeted to bromodomains of BET family proteins. Medchemcomm 5, 1871–1878 (2014).
Belkina, A.C., Nikolajczyk, B.S. & Denis, G.V. BET protein function is required for inflammation: Brd2 genetic disruption and BET inhibitor JQ1 impair mouse macrophage inflammatory responses. J. Immunol. 190, 3670–3678 (2013).
Abbassi, O., Kishimoto, T.K., McIntire, L.V., Anderson, D.C. & Smith, C.W. E-selectin supports neutrophil rolling in vitro under conditions of flow. J. Clin. Invest. 92, 2719–2730 (1993).
Gomez-Cambronero, J., Horn, J., Paul, C.C. & Baumann, M.A. Granulocyte-macrophage colony-stimulating factor is a chemoattractant cytokine for human neutrophils: involvement of the ribosomal p70 S6 kinase signaling pathway. J. Immunol. 171, 6846–6855 (2003).
Hubeau, C., Kubera, J.E., Masek-Hammerman, K. & Williams, C.M. Interleukin-6 neutralization alleviates pulmonary inflammation in mice exposed to cigarette smoke and poly(I:C). Clin. Sci. (Lond.) 125, 483–493 (2013).
Sellgren, K.L., Butala, E.J., Gilmour, B.P., Randell, S.H. & Grego, S. A biomimetic multicellular model of the airways using primary human cells. Lab Chip 14, 3349–3358 (2014).
Boers, J.E., Ambergen, A.W. & Thunnissen, F.B. Number and proliferation of clara cells in normal human airway epithelium. Am. J. Respir. Crit. Care Med. 159, 1585–1591 (1999).
Mercer, R.R., Russell, M.L., Roggli, V.L. & Crapo, J.D. Cell number and distribution in human and rat airways. Am. J. Respir. Cell Mol. Biol. 10, 613–624 (1994).
Villenave, R. et al. In vitro modeling of respiratory syncytial virus infection of pediatric bronchial epithelium, the primary target of infection in vivo. Proc. Natl. Acad. Sci. USA 109, 5040–5045 (2012).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25, 402–408 (2001).
Benam, K.H., Kok, W.L., McMichael, A.J. & Ho, L.P. Alternative spliced CD1d transcripts in human bronchial epithelial cells. PLoS ONE 6, e22726 (2011).
Kitagawa, S., Takaku, F. & Sakamoto, S. Evidence that proteases are involved in superoxide production by human polymorphonuclear leukocytes and monocytes. J. Clin. Invest. 65, 74–81 (1980).
Acknowledgements
Funding was provided by Pfizer, Merck, Wyss Institute for Biologically Inspired Engineering at Harvard University and the Defense Advanced Research Projects Agency (DARPA) under Cooperative Agreement Number W911NF-12-2-0036. We thank K. Karalis for helpful discussions, and B. Hassell and M. Mazur for technical assistance.
Author information
Authors and Affiliations
Contributions
K.H.B., R.V., C.H., H.-H.L., S.E.A., M.S., G.A.H. and D.E.I. designed the research; K.H.B. and R.V. developed the basic small airway chip model; R.V. and A.V. conducted the asthma work; K.H.B. and C.L. conducted the COPD studies; K.H.B. optimized and performed leukocyte-recruitment studies; J.C.W. performed scanning electron microscopy imaging; T.C.F. helped with confocal microscopy imaging; K.H.B. and R.V. prepared the manuscript; G.A.H. and A.B. commented on the manuscript; and D.E.I. critically revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
D.E.I. and G.A.H. are founders and hold equity in Emulate, Inc., and D.E.I. chairs its scientific advisory board.
Integrated supplementary information
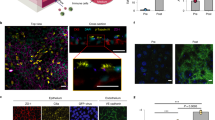
Supplementary Figure 1 Reconstitution of a differentiated human bronchiolar epithelium and pulmonary barrier on-chip.
(a) Well-differentiated human airway epithelium formed on-chip using hAECs derived from COPD donors; ciliated cells were labeled for β-tubulin IV (green), and goblet cells were stained for MUC5AC (magenta; scale bar, 20 μm; representative image from three independent stainings). (b) A confocal immunofluorescence image of club cells in bronchiolar epithelial cells differentiated on-chip (green, club cell secretory protein 10; yellow, F-actin; scale bar, 20 µm; representative image from two independent stainings). (c) Epithelial barrier function was assessed by flowing inulin-FITC (~4 kDa), dextran–Cascade blue (10 kDa) or dextran–Texas red (70 kDa) (100 µg ml−1 – 60 µL h−1) for 24h through the epithelial side of the small airway-on-a-chip containing endothelial cells alone or cocultured with well-differentiated hAECs or no cells, and measuring fluorescence in the effluent from the top and bottom channels. Barrier permeability is presented as apparent permeability (Papp; data from 1–2 independent biological replicates from 2 different donors are presented). (d) Transmission electron micrographic views of cilia formed on the apical surface of human airway epithelial cells grown in the small airway-on-a-chip; white arrows indicate two cilia (scale bar, 500 nm); inset shows a cross-section of an axoneme at higher magnification, highlighting the typical 9+2 structure (scale bar, 100 nm; representative image of 4 independent experiments performed using 4 different donors).
Supplementary Figure 2 Analysis of effects of the viral mimic poly(I:C) on interactions between epithelium and endothelium in the human small airway chip.
(a) GRO-α and IL-8 levels measured in basal secretions collected within the vascular effluent 24 h after fully differentiated hAECs cultured with (+) or without (–) endothelial cells in the presence (+) or absence (–) of 10 µg ml−1 poly(I:C) (unpaired Student’s t-test; *P < 0.05, **P < 0.01, ***P < 0.001; data represent mean ± s.e.m (compared to unstimulated epithelium only) from 3 healthy donors, with 1 biological replicate per donor; n = 3). (b) Quantitative RT-PCR analysis of the effect of poly(I:C) (10 μg ml−1) stimulation of the small airway chip for 6 h on expression of endothelial genes encoding the cell-adhesion molecules VCAM-1 and E-selectin (unpaired Student’s t-test; data represent mean ± s.e.m. (compared to unstimulated) from one donor, with 3 biological replicates per condition; n = 3). (c) Comparison of poly(I:C) induced cytokine secretion from bronchiolar and bronchial epithelial cells on-chip. Note that comparable results were found for two key proinflammatory cytokines (unpaired Student’s t-test; *P < 0.05, **P < 0.01, n.s., not significant; data represent mean ± s.e.m (compared to unstimulated) from 3–5 different healthy donors, with 1–4 biological replicates per donor; n = 4–7).
Supplementary Figure 3 Modulation of cytokine and chemokine gene expression in COPD small airway chips using a BRD4 inhibitor.
Quantitative real-time PCR analysis of the effects of budesonide and BRD4 inhibitor (applied as described in Fig. 5) on expression of IL-8, MCP-1, IL-6 and GRO-α genes in lung blood microvascular endothelial cells lysed in situ within the microvascular channel of the chips (unpaired Student’s t-test; *P < 0.05, **P < 0.01, ***P < 0.001; data represent mean ± s.e.m (compared to untreated) from two of the donors studied in Fig. 5, with 1–2 biological replicates per condition; n = 3).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Tables 1 and 2, and Supplementary Discussion (PDF 855 kb)
3D visualization of the lung small airway epithelium and endothelium reconstituted on-chip.
A movie showing various confocal fluorescence microscopic 3D views of a fully differentiated, pseudostratified, human small airway epithelium cultured at an air-liquid interface, formed from primary hAECs (F-actin, green) and cocultured with primary human pulmonary microvascular endothelial cells (F-actin, red) in the top and lower channels of the small airway chip device, respectively (DAPI-stained nuclei, blue). (MOV 339 kb)
Z-stack reconstructed 3D visualization of the mucociliary small airway epithelium on-chip.
A video showing multiple confocal immunofluorescence 3D views of a polarized, mucociliary, human bronchiolar epithelium grown in the small airway chip showing cilia stained with anti−β-tubulin (cyan) and goblet cells labeled with anti-MUC5AC (magenta). (MOV 612 kb)
Active ciliary beating of the differentiated human airway epithelium on-chip.
Time-lapse video of phase-contrast views of the human small airway epithelium with apical cilia beating actively on-chip. The video is slowed down to enable analysis of cilia beating frequencies in the region of interest (square at top left). (MOV 757 kb)
Visualization of human airway epithelial mucociliary transport on-chip.
Real-time fluorescence microscopic imaging of mucociliary transport by the differentiated human airway epithelium cultured on-chip when exposed to fluorescent 1-μm-diameter microbeads (white; scale bar, 50 μm). (MOV 503 kb)
Recruitment and adhesion of circulating human leukocytes in the human small airway chip.
Real-time fluorescence microscopic imaging showing how freshly isolated, CellTracker red−labeled human neutrophils adhere to the endothelium and roll over its surface (a single adherent neutrophil is shown in this high-magnification view) when these leukocytes are flowed under physiological conditions (shear stress, 1 dyn cm−2) through the microvascular (endothelium-lined) channel of a human small airway chip that was stimulated by the addition of poly(I:C) to the epithelium in its upper channel. (AVI 1388 kb)
Rights and permissions
About this article
Cite this article
Benam, K., Villenave, R., Lucchesi, C. et al. Small airway-on-a-chip enables analysis of human lung inflammation and drug responses in vitro. Nat Methods 13, 151–157 (2016). https://doi.org/10.1038/nmeth.3697
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.3697
This article is cited by
-
Electronic cigarette menthol flavoring is associated with increased inhaled micro and sub-micron particles and worse lung function in combustion cigarette smokers
Respiratory Research (2023)
-
Organoids and organs-on-chips: insights into predicting the efficacy of systemic treatment in colorectal cancer
Cell Death Discovery (2023)
-
Development of a novel air–liquid interface airway tissue equivalent model for in vitro respiratory modeling studies
Scientific Reports (2023)
-
Portable and integrated microfluidic flow control system using off-the-shelf components towards organs-on-chip applications
Biomedical Microdevices (2023)
-
Organ-On-A-Chip Technology: An In-depth Review of Recent Advancements and Future of Whole Body-on-chip
BioChip Journal (2023)