Abstract
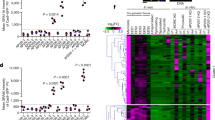
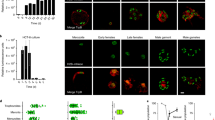
We established a conditional site-specific recombination system based on dimerizable Cre recombinase−mediated recombination in the apicomplexan parasite Toxoplasma gondii. Using a new single-vector strategy that allows ligand-dependent, efficient removal of a gene of interest, we generated three knockouts of apicomplexan genes considered essential for host-cell invasion. Our findings uncovered the existence of an alternative invasion pathway in apicomplexan parasites.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Herm-Götz, A. et al. Nat. Methods 4, 1003–1005 (2007).
Meissner, M., Breinich, M.S., Gilson, P.R. & Crabb, B.S. Curr. Opin. Microbiol. 10, 349–356 (2007).
Meissner, M., Schluter, D. & Soldati, D. Science 298, 837–840 (2002).
Buguliskis, J.S., Brossier, F., Shuman, J. & Sibley, L.D. PLoS Pathog. 6, e1000858 (2010).
Starnes, G.L., Coincon, M., Sygusch, J. & Sibley, L.D. Cell Host Microbe 5, 353–364 (2009).
Daher, W. & Soldati-Favre, D. Curr. Opin. Microbiol. 12, 408–414 (2009).
Plattner, F. et al. Cell Host Microbe 3, 77–87 (2008).
Huynh, M.H. & Carruthers, V.B. PLoS Pathog. 2, e84 (2006).
Jullien, N., Sampieri, F., Enjalbert, A. & Herman, J.P. Nucleic Acids Res. 31, e131 (2003).
Bulina, M.E. et al. Nat. Protoc. 1, 947–953 (2006).
Skillman, K.M. et al. PLoS Pathog. 7, e1002280 (2011).
Soldati, D.B. Mol. Cell. Biol. 15, 87–93 (1995).
Shaw, M.K., Compton, H.L., Roos, D.S. & Tilney, L.G. J. Cell Sci. 113, 1241–1254 (2000).
Fichera, M.E. & Roos, D.S. Nature 390, 407–409 (1997).
Hettmann, C. et al. Mol. Biol. Cell 11, 1385–1400 (2000).
Donald, R.G. & Roos, D.S. Proc. Natl. Acad. Sci. USA 90, 11703–11707 (1993).
Brecht, S., Erdhart, H., Soete, M. & Soldati, D. Gene 234, 239–247 (1999).
Huynh, M.H. & Carruthers, V.B. Eukaryot. Cell 8, 530–539 (2009).
Donald, R.G., Carter, D., Ullman, B. & Roos, D.S. J. Biol. Chem. 271, 14010–14019 (1996).
Kim, K., Soldati, D. & Boothroyd, J.C. Science 262, 911–914 (1993).
Black, M.W., Arrizabalaga, G. & Boothroyd, J.C. Mol. Cell. Biol. 20, 9399–9408 (2000).
Acknowledgements
We thank D. Soldati-Favre (University of Geneva), C.J. Beckers (University of North Carolina, Chapel Hill), A. Scherf (Pasteur Institute, Paris), J.F. Dubremetz (University of Montpellier), V. Carruthers (University of Michigan, Ann Arbor), B. Striepen (University of Georgia, Athens) and D.L. Sibley (Washington University) for sharing reagents, and members of the FACS facility of the Institute of Infection, Immunity and Inflammation at the University of Glasgow for their support. This work was supported by the Wellcome Trust. M.M. is funded by a Wellcome Trust Senior Fellowship (087582/Z/08/Z). N.A. is supported by an EviMalaR (European FP7/2007-2013, grant number 242095) PhD fellowship, and S.E. was funded via Signalling in life cycle stages of malaria parasites (MALSIG) (European FP7/2009-2012, grant number 223044). The Wellcome Trust Centre for Molecular Parasitology is supported by core funding from the Wellcome Trust (085349).
Author information
Authors and Affiliations
Contributions
N.A. established the DiCre system and generated and analyzed the myoA knockout parasites. A.J.J. generated and analyzed the mic2 knockout. S.E. generated and analyzed the conditional act1 knockout. N.J. and J.-P.H. shared confidential information for the establishment of the DiCre system. M.M. initiated and guided this study. M.M., A.J.J., N.A. and S.E. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Table 1, Supplementary Discussion, Supplementary Note (PDF 12842 kb)
Rights and permissions
About this article
Cite this article
Andenmatten, N., Egarter, S., Jackson, A. et al. Conditional genome engineering in Toxoplasma gondii uncovers alternative invasion mechanisms. Nat Methods 10, 125–127 (2013). https://doi.org/10.1038/nmeth.2301
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2301
This article is cited by
-
A splitCas9 phenotypic screen in Toxoplasma gondii identifies proteins involved in host cell egress and invasion
Nature Microbiology (2022)
-
Screening the Toxoplasma kinome with high-throughput tagging identifies a regulator of invasion and egress
Nature Microbiology (2022)
-
Phosphatidylinositol synthesis, its selective salvage, and inter-regulation of anionic phospholipids in Toxoplasma gondii
Communications Biology (2020)
-
A highly dynamic F-actin network regulates transport and recycling of micronemes in Toxoplasma gondii vacuoles
Nature Communications (2019)