Abstract
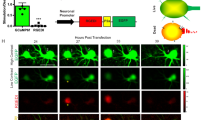
Apoptosis is of central importance to many areas of biological research, but there is a lack of methods that permit continuous monitoring of apoptosis or cell viability in a nontoxic and noninvasive manner. Here we report the development of a tool applicable to live-cell imaging that facilitates the visualization of real-time apoptotic changes without perturbing the cellular environment. We designed a polarity-sensitive annexin-based biosensor (pSIVA) with switchable fluorescence states, which allows detection only when bound to apoptotic cells. Using pSIVA with live-cell imaging, we observed dynamic local changes in individual rat neurons during degeneration in vitro and in vivo. Furthermore, we observed that pSIVA binding was reversible and clearly defined the critical period for neurons to be rescued. We anticipate pSIVA can be widely applied to address questions concerning spatiotemporal events in apoptotic processes, its reversibility and the general viability of cells in culture.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kerr, J.F., Wyllie, A.H. & Currie, A.R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer 26, 239–257 (1972).
Fadok, V.A. et al. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J. Immunol. 148, 2207–2216 (1992).
Koopman, G. et al. Annexin V for flow cytometric detection of phosphatidylserine expression on B cells undergoing apoptosis. Blood 84, 1415–1420 (1994).
Green, D.R. & Reed, J.C. Mitochondria and apoptosis. Science 281, 1309–1312 (1998).
Li, J. & Yuan, J. Caspases in apoptosis and beyond. Oncogene 27, 6194–6206 (2008).
Gavrieli, Y., Sherman, Y. & Ben-Sasson, S.A. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J. Cell Biol. 119, 493–501 (1992).
Martin, S.J. et al. Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: inhibition by overexpression of Bcl-2 and Abl. J. Exp. Med. 182, 1545–1556 (1995).
Bretscher, M.S. Asymmetrical lipid bilayer structure for biological membranes. Nat. New Biol. 236, 11–12 (1972).
Chan, A., Reiter, R., Wiese, S., Fertig, G. & Gold, R. Plasma membrane phospholipid asymmetry precedes DNA fragmentation in different apoptotic cell models. Histochem. Cell Biol. 110, 553–558 (1998).
Rimon, G., Bazenet, C.E., Philpott, K.L. & Rubin, L.L. Increased surface phosphatidylserine is an early marker of neuronal apoptosis. J. Neurosci. Res. 48, 563–570 (1997).
Seaton, B.A. (ed.) Annexins: Molecular Structure to Cellular Function. (R.G. Landes Company, Austin, Texas, USA, 1996).
Isas, J.M. et al. Calcium- and membrane-induced changes in the structure and dynamics of three helical hairpins in Annexin B12. Biochemistry 44, 16435–16444 (2005).
Isas, J.M., Langen, R., Hubbell, W.L. & Haigler, H.T. Structure and dynamics of a helical hairpin that mediates calcium-dependent membrane binding of Annexin B12. J. Biol. Chem. 279, 32492–32498 (2004).
Langen, R., Isas, J.M., Luecke, H., Haigler, H.T. & Hubbell, W.L. Membrane-mediated assembly of annexins studied by site-directed spin labeling. J. Biol. Chem. 273, 22453–22457 (1998).
Vermes, I., Haanen, C., Steffens-Nakken, H. & Reutelingsperger, C. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J. Immunol. Methods 184, 39–51 (1995).
Karpinich, N.O., Tafani, M., Rothman, R.J., Russo, M.A. & Farber, J.L. The course of etoposide-induced apoptosis from damage to DNA and p53 activation to mitochondrial release of cytochrome c. J. Biol. Chem. 277, 16547–16552 (2002).
Raff, M.C., Whitmore, A.V. & Finn, J.T. Axonal self-destruction and neurodegeneration. Science 296, 868–871 (2002).
Goldstein, J.C., Waterhouse, N.J., Juin, P., Evan, G.I. & Green, D.R. The coordinate release of cytochrome c during apoptosis is rapid, complete and kinetically invariant. Nat. Cell Biol. 2, 156–162 (2000).
Mirnikjoo, B., Balasubramanian, K. & Schroit, A.J. Mobilization of lysosomal calcium regulates the externalization of phosphatidylserine during apoptosis. J. Biol. Chem. 284, 6918–6923 (2009).
Saxena, S. & Caroni, P. Mechanisms of axon degeneration: from development to disease. Prog. Neurobiol. 83, 174–191 (2007).
Geske, F.J., Lieberman, R., Strange, R. & Gerschenson, L.E. Early stages of p53-induced apoptosis are reversible. Cell Death Differ. 8, 182–191 (2001).
Hammill, A.K., Uhr, J.W. & Scheuermann, R.H. Annexin V staining due to loss of membrane asymmetry can be reversible and precede commitment to apoptotic death. Exp. Cell Res. 251, 16–21 (1999).
Tang, H.L., Yuen, K.L., Tang, H.M. & Fung, M.C. Reversibility of apoptosis in cancer cells. Br. J. Cancer 100, 118–122 (2009).
Nikolaev, A., McLaughlin, T., O'Leary, D.D. & Tessier-Lavigne, M. APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature 457, 981–989 (2009).
Luecke, H., Chang, B.T., Mailliard, W.S., Schlaepfer, D.D. & Haigler, H.T. Crystal structure of the annexin XII hexamer and implications for bilayer insertion. Nature 378, 512–515 (1995).
Mailliard, W.S., Luecke, H. & Haigler, H.T. Annexin XII forms calcium-dependent multimers in solution and on phospholipid bilayers: a chemical cross-linking study. Biochemistry 36, 9045–9050 (1997).
Fischer, T., Lu, L., Haigler, H.T. & Langen, R. Annexin B12 is a sensor of membrane curvature and undergoes major curvature-dependent structural changes. J. Biol. Chem. 282, 9996–10004 (2007).
Rosenberg, S.S., Kelland, E.E., Tokar, E., De la Torre, A.R. & Chan, J.R. The geometric and spatial constraints of the microenvironment induce oligodendrocyte differentiation. Proc. Natl. Acad. Sci. USA 105, 14662–14667 (2008).
Chan, J.R. et al. NGF controls axonal receptivity to myelination by Schwann cells or oligodendrocytes. Neuron 43, 183–191 (2004).
Acknowledgements
We thank the members of the Chan lab for technical assistance and M. Cayouette for commenting on the manuscript. This work was partially supported by the US National Multiple Sclerosis Society Career Transition Award (J.R.C.), EY12155 (J.C.). J.R.C. is a Harry Weaver Neuroscience Scholar (TA 3008A/T, JF 2142-A-2).
Author information
Authors and Affiliations
Contributions
R.L. conceived the study and, together with J.C. and J.R.C., supervised the project. Y.E.K. designed and performed the experiments and, together with J.R.C., designed and performed the experiments involving neurons. R.L., J.C., J.R.C. and Y.E.K. analyzed the data. Y.E.K. wrote the paper.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Table 1 (PDF 7788 kb)
Supplementary Video 1
Time-lapse imaging of DRG neurons undergoing apoptosis (axon terminals). pSIVA staining of PS exposure on degenerating axons is shown in green, and PI staining of nuclei after loss of membrane integrity is shown in red. Images were taken 9 h after initial NGF deprivation, and the movie was compiled at a speed of 6 frames per second, with a 20 min interval between frames (images). (MOV 383 kb)
Supplementary Video 2
Time-lapse imaging of DRG neurons undergoing apoptosis. pSIVA staining of PS exposure is shown in green, and PI staining of nuclei after loss of membrane integrity is shown in red. Images were taken 9 h after initial NGF deprivation, and the movie was compiled at a speed of 6 frames per second, with a 20 min interval between frames (images). (MOV 702 kb)
Supplementary Video 3
Recovery of DRG neurons from 10 h of NGF deprivation. Neurons were deprived of NGF for 10 h to induce apoptosis before NGF was subsequently added back to the culture medium to induce rescue. pSIVA staining of PS exposure on degenerating axons is shown in green, and PI staining of nuclei after loss of membrane integrity is shown in red. Images were taken 7.5 h after initial NGF deprivation, and the movie was compiled at a speed of 4 frames per second, with a 30 min interval between frames (images). (MOV 710 kb)
Supplementary Video 4
Recovery of DRG neurons from 15 h of NGF deprivation. Neurons were deprived of NGF for 15 h to induce apoptosis before NGF was subsequently added back to the culture medium to induce rescue. pSIVA staining of PS exposure on degenerating axons is shown in green, and PI staining of nuclei after loss of membrane integrity is shown in red. Images were taken 7.5 h after initial NGF deprivation, and the movie was compiled at a speed of 4 frames per second, with a 30 min interval between frames (images). Boxes highlight specific areas where PS exposure occurs on the axon and reverses. (MOV 636 kb)
Rights and permissions
About this article
Cite this article
Kim, Y., Chen, J., Chan, J. et al. Engineering a polarity-sensitive biosensor for time-lapse imaging of apoptotic processes and degeneration. Nat Methods 7, 67–73 (2010). https://doi.org/10.1038/nmeth.1405
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1405
This article is cited by
-
Neutral sphingomyelinase inhibition promotes local and network degeneration in vitro and in vivo
Cell Communication and Signaling (2023)
-
Phosphatidylserine in the Nervous System: Cytoplasmic Regulator of the AKT and PKC Signaling Pathways and Extracellular “Eat-Me” Signal in Microglial Phagocytosis
Molecular Neurobiology (2023)
-
A mitotic chromatin phase transition prevents perforation by microtubules
Nature (2022)
-
A fluorogenic cyclic peptide for imaging and quantification of drug-induced apoptosis
Nature Communications (2020)
-
PTEN activation contributes to neuronal and synaptic engulfment by microglia in tauopathy
Acta Neuropathologica (2020)