Abstract
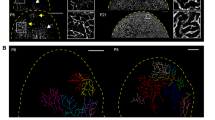
A lack of appropriate in vitro models of three-dimensional lymph vessel growth hampers the study of lymphangiogenesis. We developed a lymphatic ring assay—a potent, reproducible and quantifiable three-dimensional culture system for lymphatic endothelial cells that reproduces spreading of endothelial cells from a pre-existing vessel, cell proliferation, migration and differentiation into capillaries. In the assay, mouse thoracic duct fragments are embedded in a collagen gel, leading to the formation of lumen-containing lymphatic capillaries, which we assessed by electron microscopy and immunostaining. We developed a computerized method to quantify the lymphatic network. By applying this model to gene-deficient mice, we found evidence for involvement of the matrix metalloproteinase, MMP-2, in lymphangiogenesis. The lymphatic ring assay bridges the gap between two-dimensional in vitro models and in vivo models of lymphangiogenesis, can be used to exploit the potential of existing transgenic mouse models, and rapidly identify regulators of lymphangiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Karpanen, T. & Alitalo, K. Molecular biology and pathology of lymphangiogenesis. Annu. Rev. Pathol. 3, 367–397 (2008).
Tammela, T., Petrova, T.V. & Alitalo, K. Molecular lymphangiogenesis: new players. Trends Cell Biol. 15, 434–441 (2005).
Skobe, M. et al. Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis. Nat. Med. 7, 192–198 (2001).
Mandriota, S.J. et al. Vascular endothelial growth factor-C-mediated lymphangiogenesis promotes tumour metastasis. EMBO J. 20, 672–682 (2001).
Mancardi, S. et al. Lymphatic endothelial tumors induced by intraperitoneal injection of incomplete Freund's adjuvant. Exp. Cell Res. 246, 368–375 (1999).
Nakamura, E.S., Koizumi, K., Kobayashi, M. & Saiki, I. Inhibition of lymphangiogenesis-related properties of murine lymphatic endothelial cells and lymph node metastasis of lung cancer by the matrix metalloproteinase inhibitor MMI270. Cancer Sci. 95, 25–31 (2004).
Rutkowski, J.M., Boardman, K.C. & Swartz, M.A. Characterization of lymphangiogenesis in a model of adult skin regeneration. Am. J. Physiol. Heart Circ. Physiol. 291, H1402–H1410 (2006).
Tabibiazar, R. et al. Inflammatory manifestations of experimental lymphatic insufficiency. PLoS Med. 3, e254 (2006).
Cao, R. et al. PDGF-BB induces intratumoral lymphangiogenesis and promotes lymphatic metastasis. Cancer Cell 6, 333–345 (2004).
Ny, A., Autiero, M. & Carmeliet, P. Zebrafish and Xenopus tadpoles: Small animal models to study angiogenesis and lymphangiogenesis. Exp. Cell Res. 312, 684–693 (2006).
Kriehuber, E. et al. Isolation and characterization of dermal lymphatic and blood endothelial cells reveal stable and functionally specialized cell lineages. J. Exp. Med. 194, 797–808 (2001).
Nisato, R.E. et al. Generation and characterization of telomerase-transfected human lymphatic endothelial cells with an extended life span. Am. J. Pathol. 165, 11–24 (2004).
Pepper, M.S., Wasi, S., Ferrara, N., Orci, L. & Montesano, R. In vitro angiogenic and proteolytic properties of bovine lymphatic endothelial cells. Exp. Cell Res. 210, 298–305 (1994).
Mizuno, R., Yokoyama, Y., Ono, N., Ikomi, F. & Ohhashi, T. Establishment of rat lymphatic endothelial cell line. Microcirculation 10, 127–131 (2003).
Liersch, R., Nay, F., Lu, L. & Detmar, M. Induction of lymphatic endothelial cell differentiation in embryoid bodies. Blood 107, 1214–1216 (2006).
Nicosia, R.F. & Ottinetti, A. Growth of microvessels in serum-free matrix culture of rat aorta. A quantitative assay of angiogenesis in vitro. Lab. Invest. 63, 115–122 (1990).
Masson, V. et al. Mouse aortic ring assay: a new approach of the molecular genetics of angiogenesis. Biol. Proced. Online 4, 24–31 (2002).
Maquoi, E. et al. Anti-invasive, antitumoral, and antiangiogenic efficacy of a pyrimidine-2,4,6-trione derivative, an orally active and selective matrix metalloproteinases inhibitor. Clin. Cancer Res. 10, 4038–4047 (2004).
Masson, V. et al. Contribution of host MMP-2 and MMP-9 to promote tumor vascularization and invasion of malignant keratinocytes. FASEB J. 19, 234–236 (2005).
Leak, L.V. Studies on the permeability of lymphatic capillaries. J. Cell Biol. 50, 300–323 (1971).
Kirkin, V. et al. Characterization of indolinones which preferentially inhibit VEGF-C- and VEGF-D-induced activation of VEGFR-3 rather than VEGFR-2. Eur. J. Biochem. 268, 5530–5540 (2001).
Kathir, K.M., Kumar, T.K. & Yu, C. Understanding the mechanism of the antimitogenic activity of suramin. Biochemistry 45, 899–906 (2006).
Vincent, L. & Rafii, S. Vascular frontiers without borders: multifaceted roles of platelet-derived growth factor (PDGF) in supporting postnatal angiogenesis and lymphangiogenesis. Cancer Cell 6, 307–309 (2004).
Devy, L. et al. The pro- or antiangiogenic effect of plasminogen activator inhibitor 1 is dose dependent. FASEB J. 16, 147–154 (2002).
Lijnen, H.R. et al. Adipose tissue expression of gelatinases in mouse models of obesity. Thromb. Haemost. 85, 1111–1116 (2001).
Davis, G.E., Bayless, K.J. & Mavila, A. Molecular basis of endothelial cell morphogenesis in three-dimensional extracellular matrices. Anat. Rec. 268, 252–275 (2002).
Blacher, S. et al. Improved quantification of angiogenesis in the rat aortic ring assay. Angiogenesis 4, 133–142 (2001).
Fischer, C. et al. Anti-PlGF inhibits growth of VEGF(R)-inhibitor-resistant tumors without affecting healthy vessels. Cell 131, 463–475 (2007).
Kubo, H. et al. Blockade of vascular endothelial growth factor receptor-3 signaling inhibits fibroblast growth factor-2-induced lymphangiogenesis in mouse cornea. Proc. Natl. Acad. Sci. USA 99, 8868–8873 (2002).
Nicosia, R.F. Angiogenesis and the formation of lymphaticlike channels in cultures of thoracic duct. In Vitro Cell. Dev. Biol. 23, 167–174 (1987).
Acknowledgements
We thank S. Itohara (RIKEN brain Science Institute) for providing MMP-2–deficient mice; A. Bredan for scientific advice; and M.L. Alvarez, I. Dasoul, E. Feyereisen, L. Poma, P. Gavitelli and F. Olivier for technical assistance. This work was supported by grants from the EU Framework Program projects (FP6 and FP7), the Fonds de la Recherche Scientifique Médicale, the Fonds National de la Recherche Scientifique (FNRS, Belgium), the Fondation contre le Cancer, the Fonds spéciaux de la Recherche (University of Liège), the Centre Anticancéreux près l'Université de Liège, the Fonds Léon Fredericq (University of Liège), the Direction Generale des Technologies et de la Recherche en Region Wallonne (DGTRE), the Interuniversity Attraction Poles Program—Belgian Science Policy (Brussels, Belgium). F.B. is recipient of a Televie-FNRS grant. This work was also supported by a grant from the Deutsche Forschungsgemeinschaft to J.P.S. under the auspices of Schwerpunktprogramm (SPP 1190 “The tumor-vessel interface”).
Author information
Authors and Affiliations
Contributions
F.B. wrote the draft of the manuscript and performed most of the experiments with L.M.-L.; S.B. did all quantification work based on image analyses; G.R. contributed in setting up the model; and M.T. did the electron microscopy analysis. L.M., F.F., P.C., K.A., C.L., J.P.S. contributed to the work by either scientific advices or by providing essential materials or reagents. A.N. conceived the study and participated in its design and coordination. J.-M.F. contributed to work supervision. All authors read and approved the final manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Methods (PDF 2424 kb)
Supplementary Video 1
Lymphatic thoracic duct dissection: step 1. Thoracic ducts are obtained by microsurgery using very thin microdissecting forceps and Vannas scissors. The periaortic fibroadipose tissue is carefully dissected. (MOV 18796 kb)
Supplementary Video 2
Lymphatic thoracic duct dissection: step 2. Fat tissue surrounding the lymphatic ring was removed and care was taken not to damage the lymphatic wall. (MOV 24793 kb)
Supplementary Video 3
Lymphatic thoracic duct dissection: removal of fat tissue. (MOV 22940 kb)
Supplementary Video 4
Lymphatic thoracic duct dissection: step 3. Both edges of the thoracic duct are cut and the duct is immediately transferred to a culture dish. (MOV 15518 kb)
Rights and permissions
About this article
Cite this article
Bruyère, F., Melen-Lamalle, L., Blacher, S. et al. Modeling lymphangiogenesis in a three-dimensional culture system. Nat Methods 5, 431–437 (2008). https://doi.org/10.1038/nmeth.1205
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1205
This article is cited by
-
A 3D Human Lymphatic Vessel-on-Chip Reveals the Roles of Interstitial Flow and VEGF-A/C for Lymphatic Sprouting and Discontinuous Junction Formation
Cellular and Molecular Bioengineering (2023)
-
Cardiac macrophage subsets differentially regulate lymphatic network remodeling during pressure overload
Scientific Reports (2021)
-
Fluorescent reporter transgenic mice for in vivo live imaging of angiogenesis and lymphangiogenesis
Angiogenesis (2018)
-
TGFβ counteracts LYVE-1-mediated induction of lymphangiogenesis by small hyaluronan oligosaccharides
Journal of Molecular Medicine (2018)
-
Cooperative Effects of Vascular Angiogenesis and Lymphangiogenesis
Regenerative Engineering and Translational Medicine (2018)