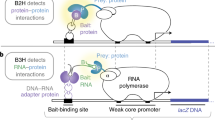
Abstract
The interpretation of genome sequences requires reliable and standardized methods to assess protein function at high throughput. Here we describe a fast and reliable pipeline to study protein function in mammalian cells based on protein tagging in bacterial artificial chromosomes (BACs). The large size of the BAC transgenes ensures the presence of most, if not all, regulatory elements and results in expression that closely matches that of the endogenous gene. We show that BAC transgenes can be rapidly and reliably generated using 96-well-format recombineering. After stable transfection of these transgenes into human tissue culture cells or mouse embryonic stem cells, the localization, protein-protein and/or protein-DNA interactions of the tagged protein are studied using generic, tag-based assays. The same high-throughput approach will be generally applicable to other model systems.
NOTE: In the version of this article initially published online, the name of one individual was misspelled in the Acknowledgments. The second sentence of the Acknowledgments paragraph should read, “We thank I. Cheesman for helpful discussions.” The error has been corrected for all versions of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Change history
29 April 2008
In the version of this article initially published online, the name of one individual was misspelled in the Acknowledgments. The second sentence of the Acknowledgments paragraph should read, "We thank I. Cheesman for helpful discussions." The error has been corrected for all versions of the article.
References
Kittler, R. et al. Genome-scale RNAi profiling of cell division in human tissue culture cells. Nat. Cell Biol. 9, 1401–1412 (2007).
Paddison, P.J. et al. A resource for large-scale RNA-interference-based screens in mammals. Nature 428, 427–431 (2004).
Kittler, R. et al. An endoribonuclease-prepared siRNA screen in human cells identifies genes essential for cell division. Nature 432, 1036–1040 (2004).
Gavin, A.C. et al. Proteome survey reveals modularity of the yeast cell machinery. Nature 440, 631–636 (2006).
Gavin, A.C. et al. Functional organization of the yeast proteome by systematic analysis of protein complexes. Nature 415, 141–147 (2002).
Ghaemmaghami, S. et al. Global analysis of protein expression in yeast. Nature 425, 737–741 (2003).
Huh, W.K. et al. Global analysis of protein localization in budding yeast. Nature 425, 686–691 (2003).
Krogan, N.J. et al. Global landscape of protein complexes in the yeast Saccharomyces cerevisiae. Nature 440, 637–643 (2006).
Muyrers, J.P., Zhang, Y., Testa, G. & Stewart, A.F. Rapid modification of bacterial artificial chromosomes by ET-recombination. Nucleic Acids Res. 27, 1555–1557 (1999).
Zhang, Y., Buchholz, F., Muyrers, J. & Stewart, A. A new logic for DNA engineering using recombination in Escherichia coli. Nat. Genet. 20, 123–128 (1998).
Kittler, R. et al. RNA interference rescue by bacterial artificial chromosome transgenesis in mammalian tissue culture cells. Proc. Natl. Acad. Sci. USA 102, 2396–2401 (2005).
Lipp, J.J., Hirota, T., Poser, I. & Peters, J.M. Aurora B controls the association of condensin I but not condensin II with mitotic chromosomes. J. Cell Sci. 120, 1245–1255 (2007).
Sarov, M. et al. A recombineering pipeline for functional genomics applied to Caenorhabditis elegans. Nat. Methods 3, 839–844 (2006).
Venken, K.J., He, Y., Hoskins, R.A. & Bellen, H.J. P[acman]: a BAC transgenic platform for targeted insertion of large DNA fragments in D. melanogaster. Science 314, 1747–1751 (2006).
Yang, Z. et al. Modified bacterial artificial chromosomes for zebrafish transgenesis. Methods 39, 183–188 (2006).
Testa, G. et al. BAC engineering for the generation of ES cell-targeting constructs and mouse transgenes. Methods Mol. Biol. 256, 123–139 (2004).
Yang, L., Guan, T. & Gerace, L. Integral membrane proteins of the nuclear envelope are dispersed throughout the endoplasmic reticulum during mitosis. J. Cell Biol. 137, 1199–1210 (1997).
Copeland, N.G., Jenkins, N.A. & Court, D.L. Recombineering: a powerful new tool for mouse functional genomics. Nat. Rev. Genet. 2, 769–779 (2001).
Cheeseman, I.M. & Desai, A. A combined approach for the localization and tandem affinity purification of protein complexes from metazoans. Sci. STKE 266, pl1 (2005).
Angrand, P.O., Daigle, N., van der Hoeven, F., Scholer, H.R. & Stewart, A.F. Simplified generation of targeting constructs using ET recombination. Nucleic Acids Res. 27, e16 (1999).
Fischle, W. et al. Regulation of HP1-chromatin binding by histone H3 methylation and phosphorylation. Nature 438, 1116–1122 (2005).
Hayakawa, T., Haraguchi, T., Masumoto, H. & Hiraoka, Y. Cell cycle behavior of human HP1 subtypes: distinct molecular domains of HP1 are required for their centromeric localization during interphase and metaphase. J. Cell Sci. 116, 3327–3338 (2003).
Carmena, M. & Earnshaw, W.C. The cellular geography of aurora kinases. Nat. Rev. Mol. Cell Biol. 4, 842–854 (2003).
Barbieri, M.A. et al. Epidermal growth factor and membrane trafficking. EGF receptor activation of endocytosis requires Rab5a. J. Cell Biol. 151, 539–550 (2000).
Simpson, J.C., Wellenreuther, R., Poustka, A., Pepperkok, R. & Wiemann, S. Systematic subcellular localization of novel proteins identified by large-scale cDNA sequencing. EMBO Rep. 1, 287–292 (2000).
Ren, B. et al. Genome-wide location and function of DNA binding proteins. Science 290, 2306–2309 (2000).
Iyer, V.R. et al. Genomic binding sites of the yeast cell-cycle transcription factors SBF and MBF. Nature 409, 533–538 (2001).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Johnson, D.S., Mortazavi, A., Myers, R.M. & Wold, B. Genome-wide mapping of in vivo protein-DNA interactions. Science 316, 1497–1502 (2007).
Mikkelsen, T.S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Robertson, G. et al. Genome-wide profiles of STAT1 DNA association using chromatin immunoprecipitation and massively parallel sequencing. Nat. Methods 4, 651–657 (2007).
Seoane, S. & Perez-Fernandez, R. The vitamin D receptor represses transcription of the pituitary transcription factor Pit-1 gene without involvement of the retinoid X receptor. Mol. Endocrinol. 20, 735–748 (2006).
Carroll, J.S. et al. Chromosome-wide mapping of estrogen receptor binding reveals long-range regulation requiring the forkhead protein FoxA1. Cell 122, 33–43 (2005).
Shen, Y. & Hendershot, L.M. Identification of ERdj3 and OBF-1/BOB-1/OCA-B as direct targets of XBP-1 during plasma cell differentiation. J. Immunol. 179, 2969–2978 (2007).
Choi, D. et al. In vitro differentiation of mouse embryonic stem cells: enrichment of endodermal cells in the embryoid body. Stem Cells 23, 817–827 (2005).
Haase, I. et al. In vitro differentiation of murine embryonic stem cells into keratinocyte-like cells. Eur. J. Cell Biol. 86, 801–805 (2007).
Soundararajan, P., Lindsey, B.W., Leopold, C. & Rafuse, V.F. Easy and rapid differentiation of embryonic stem cells into functional motoneurons using sonic hedgehog-producing cells. Stem Cells 25, 1697–1706 (2007).
Poueymirou, W.T. et al. F0 generation mice fully derived from gene-targeted embryonic stem cells allowing immediate phenotypic analyses. Nat. Biotechnol. 25, 91–99 (2007).
Neumann, B. et al. High-throughput RNAi screening by time-lapse imaging of live human cells. Nat. Methods 3, 385–390 (2006).
Pepperkok, R. & Ellenberg, J. High-throughput fluorescence microscopy for systems biology. Nat. Rev. Mol. Cell Biol. 7, 690–696 (2006).
Acknowledgements
We are grateful to J. Ellenberg and Z. Maliga for stimulating discussions, to K. Neugebauer for the help in establishing the ChIP protocol, and to O. Hudecz, C. Stingl and G. Mitulovic (Institute of Molecular Pathology) and A. Ssykor, M. Biesold, D. Richter, K. Kozak and D. Drechsel (Max Planck Institute for Molecular Cell Biology and Genetics) for excellent assistance. We thank I. Cheesman for helpful discussions. This work has been supported by the 6th Framework Program of the European Union, Integrated Project 'MitoCheck' (LSHG-CT-2004-503464), and by NGFN2 grant SMP-RNAi (01GR0402). Work in the laboratories of J.-M.P. and K.M. is supported by Boehringer Ingelheim, the GenAu Program, the Austrian Research Promotion Agency (FFG), the European Science Foundation and the Austrian Science Fund (FWF) via the EuroDynaProgram. A.F.S. received funding from the 6th Framework Program of the European Union, Integrated Project 'Heroic' (LSHG-CT-2005-018883). K.P.W. is supported by grant 1R01HG004428-01 from the National Human Genome Research Institute of the US National Institutes of Health. R.K. is supported by a long-term fellowship of the Human Frontier Science Program Organization. Y.T. was supported by the Uehara Memorial Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Y.Z. and A.F.S. are shareholders, and Y.Z. is an employee, of GeneBridges GmbH, which holds the exclusive commercial rights to the Red/ET recombineering methodologies.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Tables 1 and 2, Supplementary Methods (PDF 17838 kb)
Supplementary Table 3
Binding Sites for XBP1-S in MCF-7 Cells Identified by Chromatin Immunoprecipitation (for Chromosome 4, 15, 18, and 20). (XLS 315 kb)
Rights and permissions
About this article
Cite this article
Poser, I., Sarov, M., Hutchins, J. et al. BAC TransgeneOmics: a high-throughput method for exploration of protein function in mammals. Nat Methods 5, 409–415 (2008). https://doi.org/10.1038/nmeth.1199
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1199
This article is cited by
-
A nanobody-based strategy for rapid and scalable purification of human protein complexes
Nature Protocols (2024)
-
Temporal-iCLIP captures co-transcriptional RNA-protein interactions
Nature Communications (2023)
-
Loss of PML nuclear bodies in familial amyotrophic lateral sclerosis-frontotemporal dementia
Cell Death Discovery (2023)
-
High-content high-throughput imaging reveals distinct connections between mitochondrial morphology and functionality for OXPHOS complex I, III, and V inhibitors
Cell Biology and Toxicology (2023)
-
Deep learning for de-convolution of Smad2 versus Smad3 binding sites
BMC Genomics (2022)