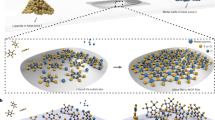
Abstract
Integrating metal–organic frameworks (MOFs) in microelectronics has disruptive potential because of the unique properties of these microporous crystalline materials. Suitable film deposition methods are crucial to leverage MOFs in this field. Conventional solvent-based procedures, typically adapted from powder preparation routes, are incompatible with nanofabrication because of corrosion and contamination risks. We demonstrate a chemical vapour deposition process (MOF-CVD) that enables high-quality films of ZIF-8, a prototypical MOF material, with a uniform and controlled thickness, even on high-aspect-ratio features. Furthermore, we demonstrate how MOF-CVD enables previously inaccessible routes such as lift-off patterning and depositing MOF films on fragile features. The compatibility of MOF-CVD with existing infrastructure, both in research and production facilities, will greatly facilitate MOF integration in microelectronics. MOF-CVD is the first vapour-phase deposition method for any type of microporous crystalline network solid and marks a milestone in processing such materials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Furukawa, H., Cordova, K. E., O’Keeffe, M. & Yaghi, O. M. The chemistry and applications of metal–organic frameworks. Science 341, 1230444 (2013).
Liu, J. et al. Applications of metal–organic frameworks in heterogeneous supramolecular catalysis. Chem. Soc. Rev. 43, 6011–6061 (2014).
Wilmer, C. E. et al. Large-scale screening of hypothetical metal–organic frameworks. Nature Chem. 4, 83–89 (2011).
Li, J.-R., Sculley, J. & Zhou, H.-C. Metal–organic frameworks for separations. Chem. Rev. 112, 869–932 (2011).
Peplow, M. Materials science: The hole story. Nature 520, 148–150 (2015).
Bétard, A. & Fischer, R. A. Metal–organic framework thin films: From fundamentals to applications. Chem. Rev. 112, 1055–1083 (2012).
Falcaro, P. et al. MOF positioning technology and device fabrication. Chem. Soc. Rev. 43, 5513–5560 (2014).
Stavila, V., Talin, A. A. & Allendorf, M. D. MOF-based electronic and opto-electronic devices. Chem. Soc. Rev. 43, 5994–6010 (2014).
Kreno, L. E. et al. Metal–organic framework materials as chemical sensors. Chem. Rev. 112, 1105–1125 (2012).
Baklanov, M. R., Adelmann, C., Zhao, L. & De Gendt, S. Advanced interconnects: Materials, processing, and reliability. ECS J. Solid State Sci. Technol. 4, Y1–Y4 (2014).
Usman, M., Mendiratta, S. & Lu, K.-L. Metal–organic frameworks: New interlayer dielectric materials. ChemElectroChem 2, 786–788 (2015).
Talin, A. A. et al. Tunable electrical conductivity in metal–organic framework thin-film devices. Science 343, 66–69 (2014).
Allendorf, M. D., Schwartzberg, A., Stavila, V. & Talin, A. A. A roadmap to implementing metal–organic frameworks in electronic devices: Challenges and critical directions. Chemistry 17, 11372–11388 (2011).
Semiconductor Industry Association, The International Technology Roadmap for Semiconductors (2013); http://www.itrs.net
Pierson, H. O. Handbook of Chemical Vapor Deposition (William Andrew, Elsevier, 1997).
Martin, P. M. Handbook of Technologies for Films and Coatings (William Andrew, Elsevier, 2005).
Hirsch, A. The era of carbon allotropes. Nature Mater. 9, 868–871 (2010).
Coclite, A. M. et al. 25th anniversary article: CVD polymers: A new paradigm for surface modification and device fabrication. Adv. Mater. 25, 5392–5423 (2013).
Lee, B. H., Yoon, B., Abdulagatov, A. I., Hall, R. a. & George, S. M. Growth and properties of hybrid organic–inorganic metalcone films using molecular layer deposition techniques. Adv. Funct. Mater. 23, 532–546 (2013).
Salmi, L. D. et al. Studies on atomic layer deposition of MOF-5 thin films. Microporous Mesoporous Mater. 182, 147–154 (2013).
Kojima, T., Choi, W. & Kawano, M. Single-crystal growth of coordination networks via the gas phase and dependence of iodine encapsulation on the crystal size. Chem. Commun. 50, 13793–13796 (2014).
Welte, L. et al. Organization of coordination polymers on surfaces by direct sublimation. Adv. Mater. 21, 2025–2028 (2009).
Fischer, D., Meyer, L. V., Jansen, M. & Müller-Buschbaum, K. Highly luminescent thin films of the dense framework ∞3[EuIm2] with switchable transparency formed by scanning femtosecond-pulse laser deposition. Angew. Chem. Int. Ed. 53, 706–710 (2014).
Meyer, L. V. et al. Organic melt, electride, and CVD induced in situ deposition of luminescent lanthanide imidazolate MOFs on nanostructured alumina. Inorg. Chem. Front. 2, 237–245 (2015).
Park, K. S. et al. From the cover: Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proc. Natl Acad. Sci. USA 103, 10186–10191 (2006).
Reboul, J. et al. Mesoscopic architectures of porous coordination polymers fabricated by pseudomorphic replication. Nature Mater. 11, 717–723 (2012).
Stassen, I. et al. Solvent-free synthesis of supported ZIF-8 films and patterns through transformation of deposited zinc oxide precursors. CrystEngComm 15, 9308–9311 (2013).
Khaletskaya, K. et al. Self-directed localization of ZIF-8 thin film formation by conversion of ZnO nanolayers. Adv. Funct. Mater. 24, 4804–4811 (2014).
Hou, C., Xu, Q., Peng, J., Ji, Z. & Hu, X. (110)-oriented ZIF-8 thin films on ITO with controllable thickness. ChemPhysChem 14, 140–144 (2013).
Eslava, S. et al. Metal–organic framework ZIF-8 films as low-κ dielectrics in microelectronics. Chem. Mater. 25, 27–33 (2013).
Shekhah, O. & Eddaoudi, M. The liquid phase epitaxy method for the construction of oriented ZIF-8 thin films with controlled growth on functionalized surfaces. Chem. Commun. 49, 10079–10081 (2013).
Bux, H. et al. Zeolitic imidazolate framework membrane with molecular sieving properties by microwave-assisted solvothermal synthesis. J. Am. Chem. Soc. 131, 16000–16001 (2009).
Bux, H., Chmelik, C., Van Baten, J. M., Krishna, R. & Caro, J. Novel MOF-membrane for molecular sieving predicted by IR-diffusion studies and molecular modeling. Adv. Mater. 22, 4741–4743 (2010).
Johnson, R. W., Hultqvist, A. & Bent, S. F. A brief review of atomic layer deposition: From fundamentals to applications. Mater. Today 17, 236–246 (2014).
Thommes, M. & Cychosz, K. a. Physical adsorption characterization of nanoporous materials: Progress and challenges. Adsorption 20, 233–250 (2014).
Rouquerol, F., Rouquerol, J. & Sing, K. Adsorption by Powders and Porous Solids (Academic, 1999).
Nezbeda, I. & Aim, K. Perturbed hard-sphere equations of state of real fluids. II. Effective hard-sphere diameters and residual properties. Fluid Phase Equilib. 17, 1–18 (1984).
Jiménez, P., Roux, M. V. & Turrión, C. Thermochemical properties of N-heterocyclic compounds IV. Enthalpies of combustion, vapour pressures and enthalpies of sublimation, and enthalpies of formation of 2-methylimidazole and 2-ethylimidazole. J. Chem. Thermodyn. 24, 1145–1149 (1992).
Hughes, J. T., Bennett, T. D., Cheetham, A. K. & Navrotsky, A. Thermochemistry of zeolitic imidazolate frameworks of varying porosity. J. Am. Chem. Soc. 135, 598–601 (2013).
Shi, Q., Chen, Z., Song, Z., Li, J. & Dong, J. Synthesis of ZIF-8 and ZIF-67 by steam-assisted conversion and an investigation of their tribological behaviors. Angew. Chem. Int. Ed. 50, 672–675 (2011).
Meng, X. & Xiao, F. S. Green routes for synthesis of zeolites. Chem. Rev. 114, 1521–1543 (2014).
Mottillo, C. et al. Mineral neogenesis as an inspiration for mild, solvent-free synthesis of bulk microporous metal–organic frameworks from metal (Zn, Co) oxides. Green Chem. 15, 2121–2131 (2013).
Cliffe, M. J., Mottillo, C., Stein, R. S., Bučar, D.-K. & Friščić, T. Accelerated aging: A low energy, solvent-free alternative to solvothermal and mechanochemical synthesis of metal–organic materials. Chem. Sci. 3, 2495–2500 (2012).
Qi, F., Stein, R. S. & Friščić, T. Mimicking mineral neogenesis for the clean synthesis of metal–organic materials from mineral feedstocks: Coordination polymers, MOFs and metal oxide separation. Green Chem. 16, 121–132 (2013).
Lu, G., Farha, O. K., Zhang, W., Huo, F. & Hupp, J. T. Engineering ZIF-8 thin films for hybrid MOF-based devices. Adv. Mater. 24, 3970–3974 (2012).
Doherty, C. M. et al. Combining UV lithography and an imprinting technique for patterning metal–organic frameworks. Adv. Mater. 25, 4701–4705 (2013).
Keitz, B. K., Yu, C. J., Long, J. R. & Ameloot, R. Lithographic deposition of patterned metal–organic framework coatings using a photobase generator. Angew. Chem. Int. Ed. 53, 5561–5565 (2014).
Migliorini, E. et al. Acceleration of neuronal precursors differentiation induced by substrate nanotopography. Biotechnol. Bioeng. 108, 2736–2746 (2011).
Paek, J. & Kim, J. Microsphere-assisted fabrication of high aspect-ratio elastomeric micropillars and waveguides. Nature Commun. 5, 3324 (2014).
Lee, H., Lee, B. P. & Messersmith, P. B. A reversible wet/dry adhesive inspired by mussels and geckos. Nature 448, 338–341 (2007).
Lu, G. & Hupp, J. Metal–organic frameworks as sensors: A ZIF-8 based Fabry–Pérot device as a selective sensor for chemical vapors and gases. J. Am. Chem. Soc. 132, 7832–7833 (2010).
Klug, H. P. & Alexander, L. E. X-ray Diffraction Procedures: For Polycrystalline and Amorphous Materials 2nd edn (Wiley-Interscience, 1974).
Schindelin, J. et al. Fiji: An open-source platform for biological-image analysis. Nature Methods 9, 676–682 (2012).
Nečas, D. & Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Cent. Eur. J. Phys. 10, 181–188 (2012).
Campagnol, N., Stassen, I., Binnemans, K., de Vos, D. E. & Fransaer, J. Metal–organic framework deposition on dealloyed substrates. J. Mater. Chem. A 3, 19747–19753 (2015).
Willems, T. F., Rycroft, C. H., Kazi, M., Meza, J. C. & Haranczyk, M. Algorithms and tools for high-throughput geometry-based analysis of crystalline porous materials. Microporous Mesoporous Mater. 149, 134–141 (2012).
Norby, P. In-situ time resolved synchrotron powder diffraction studies of syntheses and chemical reactions. Mater. Sci. Forum 228–231, 147–152 (1996).
Rietveld, H. M. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 2, 65–71 (1969).
Young, R. A. The Rietveld Method (International Union of Crystallography Monographs on Crystallography, 1993).
Hachuła, B., Nowak, M. & Kusz, J. Crystal and molecular structure analysis of 2-methylimidazole. J. Chem. Crystallogr. 40, 201–206 (2010).
O’Connor, B. H. & Raven, M. D. Application of the Rietveld refinement procedure in assaying powdered mixtures. Powder Diffr. 3, 2–6 (2013).
Acknowledgements
The authors are grateful to the Agency of Innovation by Science and Technology (IWT) for support in SBO project MOFShape. I.S. thanks Research Foundation—Flanders (FWO) for a PhD fellowship. R.A. is grateful to KU Leuven for a starting grant. The authors thank imec for support. In particular, the ESTORE team is acknowledged for their assistance in providing the substrates and the imec MCA department for TEM and TOF-SIMS measurements. D.D.V. is grateful to the Belgian Science Policy Office (BELSPO) for support in IAP project 7/05 and to KU Leuven for CASAS Methusalem funding. S.D.F. is grateful to the European Research Council (ERC) for funding (Grant Agreement No. 340324).
Author information
Authors and Affiliations
Contributions
I.S. and R.A. conceived and designed the experiments. I.S. carried out and analysed all film deposition and characterization experiments. M.S., P.F. and R.A. designed and conducted the X-ray diffraction measurements. M.S. and I.S. analysed the X-ray diffraction data. H.V.G. and W.V. conducted the atomic force microscopy experiments. G.G. and P.F. designed and manufactured the photolithography and soft lithography patterned substrates. The manuscript was primarily written by I.S. and R.A., with the input of all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 5195 kb)
Rights and permissions
About this article
Cite this article
Stassen, I., Styles, M., Grenci, G. et al. Chemical vapour deposition of zeolitic imidazolate framework thin films. Nature Mater 15, 304–310 (2016). https://doi.org/10.1038/nmat4509
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4509
This article is cited by
-
On-liquid-gallium surface synthesis of ultrasmooth thin films of conductive metal–organic frameworks
Nature Synthesis (2024)
-
Spectroscopic visualization of reversible hydrogen spillover between palladium and metal–organic frameworks toward catalytic semihydrogenation
Nature Communications (2024)
-
Metal-organic frameworks: Synthetic methods for industrial production
Nano Research (2023)
-
The Recent Advances of Metal–Organic Frameworks in Electric Vehicle Batteries
Journal of Inorganic and Organometallic Polymers and Materials (2023)
-
Solvent-free bottom-up patterning of zeolitic imidazolate frameworks
Nature Communications (2022)