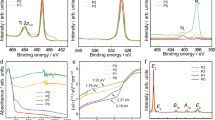
Abstract
The most widely used oxide for photocatalytic applications owing to its low cost and high activity is TiO2. The discovery of the photolysis of water on the surface of TiO2 in 19721 launched four decades of intensive research into the underlying chemical and physical processes involved2,3,4,5. Despite much collected evidence, a thoroughly convincing explanation of why mixed-phase samples of anatase and rutile outperform the individual polymorphs has remained elusive6. One long-standing controversy is the energetic alignment of the band edges of the rutile and anatase polymorphs of TiO2 (ref. 7). We demonstrate, through a combination of state-of-the-art materials simulation techniques and X-ray photoemission experiments, that a type-II, staggered, band alignment of ~ 0.4 eV exists between anatase and rutile with anatase possessing the higher electron affinity, or work function. Our results help to explain the robust separation of photoexcited charge carriers between the two phases and highlight a route to improved photocatalysts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Fujishima, A. & Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972).
Dunnill, C. W. et al. Nanoparticulate silver coated-titania thin films-Photo-oxidative destruction of stearic acid under different light sources and antimicrobial effects under hospital lighting conditions. J. Photochem. Photobiol. A 220, 113–123 (2011).
Gratzel, M. Photoelectrochemical cells. Nature 414, 338–344 (2001).
Khan, S. U. M., Al-Shahry, M. & Ingler, W. B. Efficient photochemical water splitting by a chemically modified n-TiO2 . Science 297, 2243–2245 (2002).
Yang, H. G. et al. Anatase TiO2 single crystals with a large percentage of reactive facets. Nature 453, 638–641 (2008).
Li, G. H. & Gray, K. A. The solid-solid interface: Explaining the high and unique photocatalytic reactivity of TiO2-based nanocomposite materials. Chem. Phys. 339, 173–187 (2007).
Deak, P., Aradi, B. & Frauenheim, T. Band lineup and charge carrier separation in mixed rutile-anatase systems. J. Phys. Chem. C 115, 3443–3446 (2011).
Kavan, L., Gratzel, M., Gilbert, S. E., Klemenz, C. & Scheel, H. J. Electrochemical and photoelectrochemical investigation of single-crystal anatase. J. Am. Chem. Soc. 118, 6716–6723 (1996).
Kawahara, T. et al. A patterned TiO2(anatase)/TiO2(rutile) bilayer-type photocatalyst: Effect of the anatase/rutile junction on the photocatalytic activity. Angew. Chem. Int. Ed. 41, 2811–2813 (2002).
Miyagi, T., Kamei, M., Mitsuhashi, T., Ishigaki, T. & Yamazaki, A. Charge separation at the rutile/anatase interface: A dominant factor of photocatalytic activity. Chem. Phys. Lett. 390, 399–402 (2004).
Nakajima, H., Mori, T., Shen, Q. & Toyoda, T. Photoluminescence study of mixtures of anatase and rutile TiO2 nanoparticles: Influence of charge transfer between the nanoparticles on their photo luminescence excitation bands. Chem. Phys. Lett. 409, 81–84 (2005).
Xiong, G. et al. Photoemission electron microscopy of TiO2 anatase films embedded with rutile nanocrystals. Adv. Funct. Mater 17, 2133–2138 (2007).
Hurum, D. C. et al. Probing reaction mechanisms in mixed phase TiO2 by EPR. J. Electron Spectrosc. 150, 155–163 (2006).
Hurum, D. C., Agrios, A. G., Gray, K. A., Rajh, T. & Thurnauer, M. C. Explaining the enhanced photocatalytic activity of Degussa P25 mixed-phase TiO2 using EPR. J. Phys. Chem. B 107, 4545–4549 (2003).
Hurum, D. C., Gray, K. A., Rajh, T. & Thurnauer, M. C. Recombination pathways in the Degussa P25 formulation of TiO2: Surface versus lattice mechanisms. J. Phys. Chem. B 109, 977–980 (2005).
Leytner, S. & Hupp, J. T. Evaluation of the energetics of electron trap states at the nanocrystalline titanium dioxide/aqueous solution interface via time-resolved photoacoustic spectroscopy. Chem. Phys. Lett. 330, 231–236 (2000).
Kresse, G. & Hafner, J. Ab-initio molecular-dynamics simulation of the liquid-metal amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Krukau, A. V., Vydrov, O. A., Izmaylov, A. F. & Scuseria, G. E. Influence of the exchange screening parameter on the performance of screened hybrid functionals. J. Chem. Phys. 125, 224106 (2006).
Madelung, E. The electric field in systems of regularly arranged point charges. Phys. Z 19, 524–533 (1918).
Dick, B. G. & Overhauser, A. W. Theory of the dielectric constants of alkali halide crystals. Phys. Rev. 112, 90–103 (1958).
Catlow, C. R. A. et al. Advances in computational studies of energy materials. Philos. Trans. R. Soc. A-Math. Phys. Eng. Sci. 368, 3379–3456 (2010).
Wang, J. et al. Measurement of wurtzite ZnO/rutile TiO2 heterojunction band offsets by X-ray photoelectron spectroscopy. Appl. Phys. A 103, 1099–1103 (2011).
Sokol, A. A., Bromley, S. T., French, S. A., Catlow, C. R. A. & Sherwood, P. Hybrid QM/MM embedding approach for the treatment of localized surface states in ionic materials. Int. J. Quantum Chem. 99, 695–712 (2004).
Sherwood, P. et al. QUASI: A general purpose implementation of the QM/MM approach and its application to problems in catalysis. J. Mol. Struct.-Theochem. 632, 1–28 (2003).
Cheng, J. & Sprik, M. Aligning electronic energy levels at the TiO2/H2O interface. Phys. Rev. B 82, 081406 (2010).
Swank, R. K. Surface properties of 2–6 compounds. Phys. Rev. 153, 844–849 (1967).
Sokol, A. A. et al. Point defects in ZnO. Faraday Discuss. 134, 267–282 (2007).
Grant, R. W., Kraut, E. A., Kowalczyk, S. P. & Waldrop, J. R. Measurement of potential at semiconductor interfaces by electron-spectroscopy. J. Vac. Sci. Technol. B 1, 320–327 (1983).
Kraut, E. A., Grant, R. W., Waldrop, J. R. & Kowalczyk, S. P. Semiconductor core-level to valence-band maximum binding-energy differences—precise determination by X-ray photoelectron-spectroscopy. Phys. Rev. B 28, 1965–1977 (1983).
Waldrop, J. R., Kowalczyk, S. P., Grant, R. W., Kraut, E. A. & Miller, D. L. XPS measurement of GaAs-AlAs heterojunction band discontinuities—growth sequence dependence. J. Vac. Sci. Technol. 19, 573–575 (1981).
Acknowledgements
The work presented here made use of the UCL Legion HPC Facility, the IRIDIS cluster provided by the EPSRC-funded Centre for Innovation (EP/K000144/1 and EP/K000136/1), and the HECToR supercomputer through our membership of the UK’s HPC Materials Chemistry Consortium, which is funded by EPSRC grant EP/F067496. The work in Dublin was supported by SFI through the PI programme (PI grant numbers 06/IN.1/I92 and 06/IN.1/I92/EC07), and made use of the Kelvin supercomputer as maintained by TCHPC. A.W. acknowledges support from the Royal Society for a University Research Fellowship and EU-FP7 under grant agreement 316494. D.O.S. and C.W.D. are grateful to the Ramsay Memorial Trust and University College London for the provision of their Ramsay Fellowships. D.O.S., R.G.P. and A.W. acknowledge membership of the Materials Design Network.
Author information
Authors and Affiliations
Contributions
D.O.S. wrote the manuscript with input from A.W. and A.A.S. D.O.S. and A.A.S. designed the computational experiments. C.W.D., M.J.P., R.G.P. and I.P.P. designed and performed the sample growth and XPS experiments. A.A.S., S.M.W. and C.R.A.C. calculated and analysed the band offsets using the method of interatomic potentials, D.O.S., S.A.S. and G.W.W. performed and analysed the periodic DFT calculations, and J.B., A.J.L., A.A.S., T.W.K. and P.S. developed, performed and analysed the QM/MM alignments. All authors contributed to the scientific discussion and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 844 kb)
Rights and permissions
About this article
Cite this article
Scanlon, D., Dunnill, C., Buckeridge, J. et al. Band alignment of rutile and anatase TiO2. Nature Mater 12, 798–801 (2013). https://doi.org/10.1038/nmat3697
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3697
This article is cited by
-
Electrochemistry and band structure of semiconductors (TiO2, SnO2, ZnO): Avoiding pitfalls and textbook errors
Journal of Solid State Electrochemistry (2024)
-
Critical Review on Titania-Based Nanoparticles: Synthesis, Characterization, and Application as a Photocatalyst
Chemistry Africa (2024)
-
Lattice Oxygen and Feed Oxygen Exchange for the High Rate of H2 + O2 Recombination on Ti1-xPdxO2-x Catalyst at Room Temperature
Catalysis Letters (2024)
-
A doped heterojunction photoanode: a strategy-driven approach to improve the charge carrier dynamics and photoconversion performance of the TiO2-based dye-sensitized solar cell
Journal of Materials Science: Materials in Electronics (2024)
-
Function-reversible facets enabling SrTiO3 nanocrystals for improved photocatalytic hydrogen evolution
Frontiers in Energy (2024)