Abstract
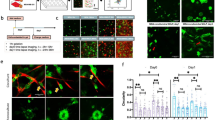
The identification of stem-cell-like cancer cells through conventional methods that depend on stem cell markers is often unreliable. We developed a mechanical method for selecting tumorigenic cells by culturing single cancer cells in fibrin matrices of ~100 Pa in stiffness. When cultured within these gels, primary human cancer cells or single cancer cells from mouse or human cancer cell lines grew within a few days into individual round colonies that resembled embryonic stem cell colonies. Subcutaneous or intravenous injection of 10 or 100 fibrin-cultured cells in syngeneic or severe combined immunodeficiency mice led to the formation of solid tumours at the site of injection or at the distant lung organ much more efficiently than control cancer cells selected using conventional surface marker methods or cultured on conventional rigid dishes or on soft gels. Remarkably, as few as ten such cells were able to survive and form tumours in the lungs of wild-type non-syngeneic mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
18 May 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41563-021-01032-0
References
Bonnet, D. & Dick, J. E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nature Med. 3, 730–737 (1997).
Schmidt, P. et al. Eradication of melanomas by targeted elimination of a minor subset of tumour cells. Proc. Natl Acad. Sci. USA 108, 2474–2479 (2011).
Civenni, G. et al. Human CD271-positive melanoma stem cells associated with metastasis establish tumour heterogeneity and long-term growth. Cancer Res. 71, 3098–3109 (2011).
Gupta, P. B., Chaffer, C. L. & Weinberg, R. A. Cancer stem cells: mirage or reality? Nature Med. 15, 1010–1012 (2009).
Shipitsin, M. & Polyak, K. The cancer stem cell hypothesis: In search of definitions, markers, and relevance. Lab. Invest. 88, 459–463 (2008).
Quintana, E. et al. Efficient tumour formation by single human melanoma cells. Nature 456, 593–598 (2008).
Dieter, S. M. et al. Distinct types of tumour-initiating cells form human colon cancer tumours and metastases. Cell Stem Cell 9, 357–365 (2011).
Gupta, P. B. et al. Stochastic state transitions give rise to phenotypic equilibrium in populations of cancer cells. Cell 146, 633–644 (2011).
Chaffer, C. L. et al. Normal and neoplastic nonstem cells can spontaneously convert to a stem-like state. Proc. Natl Acad. Sci. USA 108, 7950–7955 (2011).
Chowdhury, F. et al. Material properties of the cell dictate stress-induced spreading and differentiation in embryonic stem cells. Nature Mater. 9, 82–88 (2010).
Chowdhury, F. et al. Soft substrates promote homogeneous self-renewal of embryonic stem cells via downregulating cell-matrix tractions. PLoS ONE 5, e15655 (2010).
Paszek, M. J. et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 8, 241–254 (2005).
Levental, K. R. et al. Matrix crosslinking forces tumour progression by enhancing integrin signaling. Cell 139, 891–906 (2009).
Samuel, M. S. et al. Actomyosin-mediated cellular tension drives increased tissue stiffness and β-catenin activation to induce epidermal hyperplasia and tumour growth. Cancer Cell 19, 776–791 (2011).
Uibo, R. et al. Soft materials to treat central nervous system injuries: Evaluation of the suitability of non-mammalian fibrin gels. Biochim. Biophys. Acta 1793, 924–930 (2009).
Ju, Y. E., Janmey, P. A., McCormick, M. E., Sawyer, E. S. & Flanagan, L. A. Enhanced neurite growth from mammalian neurons in three-dimensional salmon fibrin gels. Biomaterials 28, 2097–2108 (2007).
Winer, J. P., Oake, S. & Janmey, P. A. Non-linear elasticity of extracellular matrices enables contractile cells to communicate local position and orientation. PLoS ONE 4, e6382 (2009).
Maeda, M., Suga, T., Takasuka, N., Hoshi, A. & Sasaki, T. Effect of bis(bilato)-1,2-cyclohexanediammineplatinum(II) complexes on lung metastasis of B16-F10 melanoma cells in mice. Cancer Lett. 55, 143–147 (1990).
Hiyama, E. & Hiyama, K. Telomere and telomerase in stem cells. Br. J. Cancer 96, 1020–1024 (2007).
Engler, A. J., Sen, S., Sweeney, H. L. & Discher, D. E. Matrix elasticity directs stem cell lineage specification. Cell 126, 677–689 (2006).
Gilbert, P. M. et al. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science 329, 1078–1081 (2010).
Poh, Y. C., Chowdhury, F., Tanaka, T. S. & Wang, N. Embryonic stem cells do not stiffen on rigid substrates. Biophys. J. 99, L01–L03 (2010).
Weisel, J. W. Fibrinogen and fibrin. Adv. Protein Chem. 70, 247–299 (2005).
Hong, H. & Stegemann, J. P. 2D and 3D collagen and fibrin biopolymers promote specific ECM and integrin gene expression by vascular smooth muscle cells. J. Biomater. Sci. Polym. Ed. 19, 1279–1293 (2008).
Ramgolam, K. et al. Melanoma spheroids grown under neural crest cell conditions are highly plastic migratory/invasive tumour cells endowed with immunomodulator function. PLoS ONE 6, e18784 (2011).
Scaffidi, P. & Misteli, T. In vitro generation of human cells with cancer stem cell properties. Nature Cell Biol. 13, 1051–1061 (2011).
Sanz-Moreno, V. et al. ROCK and JAK1 signaling cooperate to control actomyosin contractility in tumour cells and stroma. Cancer Cell 20, 229–245 (2011).
Mierke, C. T., Frey, B., Fellner, M., Herrmann, M. & Fabry, B. Integrin α5β1 facilitates cancer cell invasion through enhanced contractile forces. J. Cell Sci. 124, 369–383 (2011).
Pathak, A. & Kumar, S. Biophysical regulation of tumour cell invasion: Moving beyond matrix stiffness. Integ. Biol. 3, 267–278 (2011).
Costantini, V. & Zacharski, L. R. The role of fibrin in tumour metastasis. Cancer Metastasis Rev. 11, 283–290 (1992).
Palumbo, J. S. & Degen, J. L. Fibrinogen and tumour cell metastasis. Haemostasis 31 (Suppl 1), 11–15 (2001).
Palumbo, J. S. et al. Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell-mediated elimination of tumour cells. Blood 105, 178–185 (2005).
Malik, G. et al. Plasma fibronectin promotes lung metastasis by contributions to fibrin clots and tumour cell invasion. Cancer Res. 70, 4327–4334 (2010).
Van Sluis, G. L. et al. Endogenous activated protein C is essential for immune-mediated cancer cell elimination from the circulation. Cancer Lett. 306, 106–110 (2011).
Liang, Y. et al. A cell-instructive hydrogel to regulate malignancy of 3D tumour spheroids with matrix rigidity. Biomaterials 32, 9308–9315 (2011).
Bissell, M. J. & Hines, W. C. Why don’t we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nature Med. 17, 320–329 (2011).
Tilghman, R. W. et al. Matrix rigidity regulates cancer cell growth and cellular phenotype. PLoS. ONE 5, e12905 (2010).
Cordenonsi, M. et al. The Hippo transducer TAZ confers cancer stem cell-related traits on breast cancer cells. Cell 147, 759–772 (2011).
Dupont, S. et al. Role of YAP/TAZ in mechanotransduction. Nature 474, 179–183 (2011).
Valastyan, S. & Weinberg, R. A. Tumour metastasis: molecular insights and evolving paradigms. Cell 147, 275–292 (2011).
Wirtz, D., Konstantopoulos, K. & Searson, P. C. The physics of cancer: The role of physical interactions and mechanical forces in metastasis. Nature Rev. Cancer 11, 512–522 (2011).
Chaffer, C. L. & Weinberg, R. A. A perspective on cancer cell metastasis. Science 331, 1559–1564 (2011).
Acknowledgements
We would like to thank J. Winer for help with fibrin-gel protocols. This work was supported by funds from Huazhong University of Science and Technology (HUST), the National Basic Research Program of China (2012CB932500) (B.H.), the China Natural National Science Foundation (30911120482) (B.H.), the Fundamental Research Funds for the Central Universities (HUST-2011TS027) (B.H.), and USA NIH grant GM072744 (N.W.).
Author information
Authors and Affiliations
Contributions
N.W. and B.H. conceived the project; B.H., N.W., J.L. and Y.T. designed the experiments. B.H., J.L., Y.T., H.Z., Y.Z., P.X., J.C., Y-C.P. and K.T. carried out the experiments and analysed the data. N.W., B.H., J.L. and Y.T. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2798 kb)
Rights and permissions
About this article
Cite this article
Liu, J., Tan, Y., Zhang, H. et al. Soft fibrin gels promote selection and growth of tumorigenic cells. Nature Mater 11, 734–741 (2012). https://doi.org/10.1038/nmat3361
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3361
This article is cited by
-
Application of tumor microparticles in tumor prevention and treatment
Cancer Nanotechnology (2023)
-
Extracellular matrix-derived mechanical force governs breast cancer cell stemness and quiescence transition through integrin-DDR signaling
Signal Transduction and Targeted Therapy (2023)
-
Biophysics in tumor growth and progression: from single mechano-sensitive molecules to mechanomedicine
Oncogene (2023)
-
Cooperative phagocytosis of solid tumours by macrophages triggers durable anti-tumour responses
Nature Biomedical Engineering (2023)
-
A biomechanical view of epigenetic tumor regulation
Journal of Biological Physics (2023)