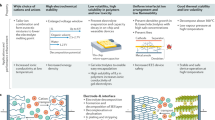
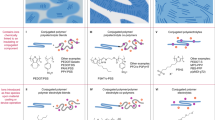
Abstract
Ionic liquids are room-temperature molten salts, composed mostly of organic ions that may undergo almost unlimited structural variations. This review covers the newest aspects of ionic liquids in applications where their ion conductivity is exploited; as electrochemical solvents for metal/semiconductor electrodeposition, and as batteries and fuel cells where conventional media, organic solvents (in batteries) or water (in polymer-electrolyte-membrane fuel cells), fail. Biology and biomimetic processes in ionic liquids are also discussed. In these decidedly different materials, some enzymes show activity that is not exhibited in more traditional systems, creating huge potential for bioinspired catalysis and biofuel cells. Our goal in this review is to survey the recent key developments and issues within ionic-liquid research in these areas. As well as informing materials scientists, we hope to generate interest in the wider community and encourage others to make use of ionic liquids in tackling scientific challenges.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Earle, M. J. et al. The distillation and volatility of ionic liquids. Nature 439, 831–834 (2006).
Wasserscheid, P. & Welton, T. (eds). Ionic Liquids in Synthesis (Wiley-VCH, 2007).
Endres, F., Abbott, A. & MacFarlane, D. R. (eds). Electrodeposition from Ionic Liquids (Wiley-VCH, 2008).
Abbott, A. P. & McKenzie, K. J. Application of ionic liquids to the electrodeposition of metals. Phys. Chem. Chem. Phys. 8, 4265–4279 (2006).
Ziegler, K. & Lehmkuhl, H. Die elektrolytische Abscheidung von Aluminium aus organischen Komplexverbindungen. Z. Anorg. Allg. Chem. 283, 414–424 (1956).
Kautek, W. & Birkle, S. Aluminum-electrocrystallization from metal organic electrolytes. Electrochim. Acta 34, 1213–1218 (1989).
Caporali, S. et al. Aluminium electroplated from ionic liquids as protective coating against steel corrosion. Corros. Sci. 50, 534–539 (2008).
Zein El Abedin, S. Coating of mild steel by aluminium in the ionic liquid [EMIm]Tf2N and its corrosion performance. Z. Phys. Chem. 220, 1293–1308 (2006).
Liu, Q. X., Zein El Abedin, S. & Endres, F. Electroplating of mild steel by aluminium in a first generation ionic liquid: a green alternative to commercial Al-plating in organic solvents. Surf. Coat. Tech. 201, 1352–1356 (2006).
Zein El Abedin, S., Moustafa, E. M., Hempelmann, R., Natter, H. & Endres, F. Electrodeposition of nano- and microcrystalline aluminium in three different air and water stable ionic liquids. ChemPhysChem 7, 1535–1543 (2006).
Deng, M. J. et al. Dicyanamide anion based ionic liquids for electrodeposition of metals. Electrochem. Commun. 10, 213–216 (2008).
Zein El Abedin, S., Welz-Biermann, S. U. & Endres, F. A study on the electrodeposition of tantalum on NiTi alloy in an ionic liquid and corrosion behaviour of the coated alloy. Electrochem. Commun. 7, 941–946 (2005).
Arnould, C. J., Delhalle, J. & Mekhalif, Z. Multifunctional hybrid coating on titanium towards hydroxyapatite growth: electrodeposition of tantalum and its molecular functionalization with organophosphonic acids films. Electrochim. Acta 53, 5632–5638 (2008).
Endres, F. et al. On the electrodeposition of titanium in ionic liquids. Phys. Chem. Chem. Phys. 10, 2189–2199 (2008).
Redel, E., Thomann, R. & Janiak, C. Use of ionic liquids (ILs) for the IL-anion size-dependent formation of Cr, Mo and W nanoparticles from metal carbonyl M(CO)6 precursors. Chem. Commun. 15, 1789–1791 (2008).
Tsuda, T., Arimoto, S., Kuwabata, S. & Hussey, C. L. Electrodeposition of Al–Mo–Ti ternary alloys in the Lewis acidic aluminum chloride-1-ethyl-3-methylimidazolium chloride room-temperature ionic liquid. J. Electrochem. Soc. 155, D256–D262 (2008).
Kazeminezhad, I. et al. Templated electrodeposition of silver nanowires in a nanoporous polycarbonate membrane from a nonaqueous ionic liquid electrolyte. Appl. Phys. A 86, 373–375 (2007).
Yang, P. X., An, M. Z., Su, C. N. & Wang, F. P. Electrodeposition of cobalt nanowires array from an ionic liquid. Chinese J. Inorg. Chem. 23, 1501–1504 (2007).
Al-Salman, R. et al. Template assisted electrodeposition of germanium and silicon nanowires in an ionic liquid. Phys. Chem. Chem. Phys. 10, 6233–6237 (2008).
Al-Salman, R., Zein El Abedin, S. & Endres, F. Electrodeposition of Ge, Si and SixGe1−x from an air- and water-stable ionic liquid. Phys. Chem. Chem. Phys. 10, 4650–4657 (2008).
Armand, M. & Tarascon, J.-M. Building better batteries. Nature 451, 652–657 (2008).
Matsumoto, H., Sakaebe, H. & Tatsumi, K. Preparation of room temperature ionic liquids based on aliphatic onium cations and asymmetric amide anions and their electrochemical properties as a lithium battery electrolyte. J. Power Sources 160, 1308–1313 (2006).
Seki, S. et al. Lithium secondary batteries using modified-imidazolium room-temperature ionic liquid. J. Phys. Chem. B 110, 10228–10230 (2006).
Shin, J.-H., Henderson, W. A. & Passerini, S. PEO-based polymer electrolytes with ionic liquids and their use in lithium metal-polymer electrolyte batteries. J. Electrochem. Soc. 152, A978–A983 (2005).
Garcia, B., Lavallée, S., Perron, G., Michot, C. & Armand, M. Room temperature molten salts as lithium battery electrolyte. Electrochim. Acta 49, 4583–4588 (2004).
Howlett, P. C., MacFarlane, D. R. & Hollenkamp, A. F. High lithium metal cycling efficiency in a room-temperature ionic liquid. Electrochem. Solid-State Lett. 7, A97–A101 (2004).
Fernicola, A. et al. LiTFSI-BEPyTFSI as an improved ionic liquid electrolyte for rechargeable lithium batteries. J. Power Sources 174, 342–348 (2007).
Seki, S. et al. Reversibility of lithium secondary batteries using a room-temperature ionic liquid mixture and lithium metal. Electrochem. Solid-State Lett. 8, A577–A578 (2005).
Ogasawara, T. et al. Rechargeable Li2O2 electrode for lithium batteries. J. Am. Chem. Soc. 128, 1390–1393 (2006).
Kordeesch, K. & Simader, G. (eds). Fuel Cells and Their Applications (VCH, 1996).
Kreur, K. D. Handbook of Fuel Cells: Fundamental, Technology & Applications Vol. 3 (eds Vielstich, W., Lamm, A. & Gasteiger, H. A.) (Wiley, 2003).
Fuller, J., Breda, A. C. & Carlin, R. T. Ionic liquid-polymer gel electrolytes. J. Electrochem. Soc. 144, L67–L70 (1997).
Navarra, M. A., Panero, S. & Scrosati, B. Novel, ionic-liquid-based, gel-type proton membranes. Electrochem. Solid-State Lett. 8, A324–A327 (2005).
Susan, M. A. B. H., Kaneko, T., Noda, A. & Watanabe, M. Ion gels prepared by in situ radical polymerization of vinyl monomers in an ionic liquid and their characterization as polymer electrolytes. J. Am. Chem. Soc. 127, 4976–4983 (2005).
Winther-Jensen, B., Winther-Jensen, O., Forsyth, M. & MacFarlane, D. R. High rates of oxygen reduction over a vapor phase-polymerized PEDOT electrode. Science 321, 671–674 (2008).
Simon, P. & Gogotsi, Y. Materials for electrochemical capacitors. Nature Mater. 7, 845–854 (2008).
Sato, T., Masuda, G. & Takagi, K. Electrochemical properties of novel ionic liquids for electric double layer capacitor applications. Electrochim. Acta 49, 3603–3611 (2004).
Arbizzani, C. et al. Safe high-energy supercapacitors based on solvent-free ionic liquid electrolytes ionic liquid electrolytes. J. Power Sources 185, 1575–1579 (2008).
Fukaya, Y., Sugimoto, A. & Ohno, H. Superior solubility of polysaccharides in low viscosity, polar, and halogen-free 1,3-dialkylimidazolium formates. Biomacromolecules 7, 3295–3297 (2006).
Hermanutz, F. et al. New developments in dissolving and processing of cellulose in ionic liquids. Macromol. Symp. 262, 23–27 (2008).
Swatloski, R. P., Spear, S. K., Holbrey, J. D. & Rogers, R. D. Dissolution of cellulose with ionic liquids. J. Am. Chem. Soc. 124, 4974–4975 (2002).
Wu, J. et al. Homogeneous acetylation of cellulose in a new ionic liquid. Biomacromolecules 5, 266–268 (2004).
Anderson, J. L., Ding, J., Welton, T. & Armstrong, D. W. Characterizing ionic liquids on the basis of multiple solvation interactions. J. Am. Chem. Soc. 124, 14247–14254 (2002).
Fukaya, Y., Hayashi, K., Wada, M. & Ohno, H. Cellulose dissolution with polar ionic liquids under mild conditions: required factors for anions. Green Chem. 10, 44–46 (2008).
Ohno, H., Suzuki, C., Fukumoto, K., Yoshizawa, M. & Fujita, K. Electron transfer process of poly(ethylene oxide) modified cytochrome c in imidazolium type ionic liquid. Chem. Lett. 32, 450–451 (2003).
Nakashima, K., Maruyama, T., Kamiya, N. & Goto, M. Comb-shaped poly(ethylene glycol)-modified subtilisin Carlsberg is soluble and highly active in ionic liquids. Chem. Commun. 4297–4299 (2005).
Shimojo, K., Nakashima, K., Kamiya, N. & Goto, M. Crown ether-mediated extraction and functional conversion of cytochrome c in ionic liquids. Biomacromolecules 7, 2–5 (2006).
Fujita, K., MacFarlane, D. R. & Forsyth, M. Protein solubilising and stabilising ionic liquids. Chem. Commun. 4804–4806 (2005).
Fujita, K. et al. Unexpected improvement in stability and utility of cytochrome c by solution in biocompatible ionic liquids. Biotechnol. Bioeng. 94, 1209–1213 (2006).
Fujita, K. et al. Solubility and stability of cytochrome c in hydrated ionic liquids: effect of oxo acid residues and kosmotropicity. Biomacromolecules 8, 2080–2086 (2007).
Tamura, K. & Ohno, H. Solubility of cytochrome c in ionic liquids and redox response at high temperature. 2nd Internat. Congr. Ionic Liquids, abstr. 2P09-090, 306 (2007).
DiCarlo, C. M., Compton, D. L., Evans, K. O. & Laszlo, J. A. Bioelectrocatalysis in ionic liquids. Examining specific cation and anion effects on electrode-immobilized cytochrome c. Bioelectrochemistry 68, 134–143 (2006).
Turner, M. B. et al. Ionic liquid salt-induced inactivation and unfolding of cellulase from Trichoderma reesei. Green Chem. 5, 443–447 (2003).
Motoyama, Y., Nakamura, N. & Ohno, H. An ethanol/dioxygen biofuel cell using hydrophobic ionic liquid as electrolyte. 2nd Internat. Congr. Ionic Liquids, abstr. 1P09-091, 213 (2007).
Lu, W. et al. Use of ionic liquids for pi-conjugated polymer electrochemical devices. Science 297, 983–987 (2002).
Ding, J. et al. Use of ionic liquids as electrolytes in electromechanical actuator systems based on inherently conducting polymers. Chem. Mater. 15, 2392–2398 (2003).
Zhou, D. Z. et al. Solid state actuators based on polypyrrole and polymer-in-ionic liquid electrolytes. Electrochim. Acta 48, 2355–2359 (2003).
Xi, B. B. et al. Poly(3-methylthiophene) electrochemical actuators showing increased strain and work per cycle at higher operating stresses. Polymer (Guildf.) 47, 7720–7725 (2006).
Lu, W., Norris, I. D. & Mattes, B. R. Electrochemical actuator devices based on polyaniline yarns and ionic liquid electrolytes. Aust. J. Chem. 58, 263–269 (2005).
MacFarlane, D. R. et al. Lewis base ionic liquids. Chem. Commun. 1905–1917 (2006).
Pringle, J. et al. The influence of the monomer and the ionic liquid on the electrochemical preparation of polythiophene. Polymer (Guildf.) 47, 2047–2058 (2005).
Lim, H. T., Lef, J. W. & Yoo, Y. T. Actuation behavior of a carbon nanotube/nafion™ IPMC actuator containing an ionic liquid. J. Korean Phys. Soc. 49, 1101–1106 (2006).
Barisci, J. N., Wallace, G. G., MacFarlane, D. R. & Baughman, R. H. Investigation of ionic liquids as electrolytes for carbon nanotube electrodes. Electrochem. Commun. 6, 22–27 (2004).
Vidal, F., Plesse, C., Teyssie, D. & Chevrot, C. Long-life air working conducting semi-IPN/ionic liquid based actuator. Synth. Met. 142, 287–291 (2004).
Vidal, F., Juger, J., Chevrot, C. & Teyssie, D. Interpenetrating polymer networks from polymeric imidazolium-type ionic liquid and polybutadiene. Polym. Bull. 57, 473–480 (2006).
Vidal, F. et al. Long-life air working semi-IPN/ionic liquid: new precursor of artificial muscles. Mol. Cryst. Liq. Cryst. 448, 95–102 (2006).
Cho, M. S. et al. A solid state actuator based on the PEDOT/NBR system. Sens. Actuators B 119, 621–624 (2006).
Cho, M. S. et al. A solid state actuator based on the PEDOT/NBR system: effect of anion size of imidazolium ionic liquid. Mol. Cryst. Liq. Cryst. 464, 633–638 (2007).
MacFarlane, D. R. & Forsyth, M. Plastic crystal electrolyte materials: new perspectives on solid state ionics. Adv. Mater. 13, 957–966 (2001).
Long, S., MacFarlane, D. R. & Forsyth, M. Fast ion conduction in molecular plastic crystals. Solid State Ionics 161, 105–112 (2003).
Bennett, M. D. & Leo, D. J. Ionic liquids as stable solvents for ionic polymer transducers. Sens. Actuators A 115, 79–90 (2004).
Wang, J., Xu, C. Y., Taya, M. & Kuga, Y. A. Flemion-based actuator with ionic liquid as solvent. Smart Mater. Struct. 16, S214–S219 (2007).
Ohno, H. (ed). Electrochemical Aspects of Ionic Liquids (Wiley, 2005).
Acknowledgements
We thank M. Mastragostino for discussions and information concerning supercapacitors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Armand, M., Endres, F., MacFarlane, D. et al. Ionic-liquid materials for the electrochemical challenges of the future. Nature Mater 8, 621–629 (2009). https://doi.org/10.1038/nmat2448
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat2448
This article is cited by
-
Solid electrolytes based on cellulose nanocrystals with protic ionic liquid for next-generation fuel cells
Cellulose (2024)
-
Ionic liquids in green energy storage devices: lithium-ion batteries, supercapacitors, and solar cells
Monatshefte für Chemie - Chemical Monthly (2024)
-
Effect of bulky anions on the liquid-liquid phase transition in phosphonium ionic liquids: Ambient and high-pressure dielectric studies
Scientific Reports (2023)
-
Long-chain anionic surfactants enabling stable perovskite/silicon tandems with greatly suppressed stress corrosion
Nature Communications (2023)
-
Machine learning-guided discovery of ionic polymer electrolytes for lithium metal batteries
Nature Communications (2023)