Abstract
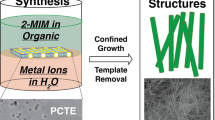
Shaped zeolite nanocrystals and larger zeolite particles with three-dimensionally ordered mesoporous (3DOm) features hold exciting technological implications for manufacturing thin, oriented molecular sieve films and realizing new selective, molecularly accessible and robust catalysts. A recognized means for controlled synthesis of such nanoparticulate and imprinted materials revolves around templating approaches, yet identification of an appropriately versatile template has remained elusive. Because of their highly interconnected pore space, ordered mesoporous carbon replicas serve as conceptually attractive materials for carrying out confined synthesis of zeolite crystals. Here, we demonstrate how a wide range of crystal morphologies can be realized through such confined growth within 3DOm carbon, synthesized by replication of colloidal crystals composed of size-tunable (about 10–40 nm) silica nanoparticles. Confined crystal growth within these templates leads to size-tunable, uniformly shaped silicalite-1 nanocrystals as well as 3DOm-imprinted single-crystal zeolite particles. In addition, novel crystal morphologies, consisting of faceted crystal outgrowths from primary crystalline particles have been discovered, providing new insight into constricted crystal growth mechanisms underlying confined synthesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lai, Z. P. et al. Microstructural optimization of a zeolite membrane for organic vapor separation. Science 300, 456–460 (2003).
Snyder, M. A. & Tsapatsis, M. Hierarchical nano-manufacturing: From shaped zeolite nanoparticles to high performance separation membranes. Angew. Chem. Int. Ed. 46, 7560–7573 (2007).
Yoon, K. B. Organization of zeolite microcrystals for production of functional materials. Acc. Chem. Res. 40, 29–40 (2007).
Choi, M. et al. Amphiphilic organosilane-directed synthesis of crystalline zeolite with tunable mesoporosity. Nature Mater. 5, 718–723 (2006).
Corma, A. & Davis, M. E. Issues in the synthesis of crystalline molecular sieves: Towards the crystallization of low framework-density structures. ChemPhysChem 5, 304–313 (2004).
Ogura, M. Towards realization of a micro- and mesoporous composite silicate catalyst. Catal. Surv. Asia 12, 16–27 (2008).
Srivastava, R., Choi, M. & Ryoo, R. Mesoporous materials with zeolite framework: Remarkable effect of the hierarchical structure for retardation of catalyst deactivation. Chem. Commun. 43, 4489–4491 (2006).
Sun, Y. & Prins, R. Friedel-Crafts alkylations over hierarchical zeolite catalysts. Appl. Catal. A 336, 11–16 (2008).
Tao, Y. S., Kanoh, H., Abrams, L. & Kaneko, K. Mesopore-modified zeolites: Preparation, characterization, and applications. Chem. Rev. 106, 896–910 (2006).
Kim, S.-S., Shah, J. & Pinnavaia, T. J. Colloid-imprinted carbons as templates for the nanocasting synthesis of mesoporous ZSM-5 zeolite. Chem. Mater. 15, 1664–1668 (2003).
Schmidt, I., Madsen, C. & Jacobsen, C. J. H. Confined space synthesis. A novel route to nanosized zeolites. Inorg. Chem. 39, 2279–2283 (2000).
Jacobsen, C. J. H., Madsen, C., Houzvicka, J., Schmidt, I. & Carlsson, A. Mesoporous zeolite single crystals. J. Am. Chem. Soc. 122, 7116–7117 (2000).
Yokoi, T. et al. Periodic arrangement of silica nanospheres assisted by amino acids. J. Am. Chem. Soc. 128, 13664–13665 (2006).
Fang, Y. & Hu, H. An ordered mesoporous aluminosilicate with completely crystalline zeolite wall structure. J. Am. Chem. Soc. 128, 10636–10637 (2006).
Ogura, M., Zhang, Y., Elangovan, S. P. & Okubo, T. Formation of ZMM-n: The composite materials having both natures of zeolites and mesoporous silica materials. Microporous Mesoporous Mater. 101, 224–230 (2007).
Schmidt, I. et al. Carbon nanotube templated growth of mesoporous zeolite single crystals. Chem. Mater. 13, 4416–4418 (2001).
Tao, Y., Kanoh, H. & Kaneko, K. ZSM-5 monolith of uniform mesoporous channels. J. Am. Chem. Soc. 125, 6044–6045 (2003).
Wang, J., Groen, J. C., Yue, W., Zhou, W. & Coppens, M.-O. Facile synthesis of ZSM-5 composites with hierarchical porosity. J. Mater. Chem. 18, 468–474 (2008).
Jacobsen, C. J. H., Madsen, C., Janssen, A. H., Jakobsen, H. J. & Skibsted, J. Zeolites by confined space synthesis - characterization of the acid sites in nanosized ZSM-5 by ammonia desorption and 27Al/29Si-MAS NMR spectroscopy. Microporous Mesoporous Mater. 39, 393–401 (2000).
Janssen, A. H., Schmidt, I., Jacobsen, C. J. H., Koster, A. J. & de Jong, K. P. Exploratory study of mesopore templating with carbon during zeolite synthesis. Microporous Mesoporous Mater. 65, 59–75 (2003).
Madsen, C. & Jacobsen, C. J. H. Nanosized zeolite crystals—convenient control of crystal size distribution by confined space synthesis. Chem. Commun. 673–674 (1999).
Dessau, R. M., Valyocsik, E. W. & Goeke, N. H. Aluminum zoning in ZSM-5 as revealed by selective silica removal. Zeolites 12, 776–779 (1992).
Groen, J. C., Jansen, J. C., Moulijn, J. A. & Perez-Ramirez, J. Optimal aluminium-assisted mesoporosity development in MFI zeolites by desilication. J. Phys. Chem. B 108, 13062–13065 (2004).
Ogura, M. et al. Formation of uniform mesopores in ZSM-5 zeolite through treatment in alkaline solution. Chem. Lett. 882–883 (2000).
Ogura, M. et al. Alkali-treatment technique—New method for modification of structural and acid-catalytic properties of ZSM-5 zeolites. Appl. Catal. A 219, 33–43 (2001).
Lew, C. M., Li, Z., Zones, S. I., Sun, M. & Yan, Y. Control of size and yield of pure-silica-zeolite MFI nanocrystals by addition of methylene blue to the synthesis solution. Microporous Mesoporous Mater. 105, 10–14 (2007).
Lee, S., Carr, C. S. & Shantz, D. F. Anionic microemulsion-mediated low temperature synthesis of anisotropic silicalite-1 nanocrystals. Langmuir 21, 12031–12036 (2005).
Davis, T. M. et al. Mechanistic principles of nanoparticle evolution to zeolite crystals. Nature Mater. 5, 400–408 (2006).
Fedeyko, J. M., Rimer, J. D., Lobo, R. F. & Vlachos, D. G. Spontaneous formation of silica nanoparticles in basic solutions of small tetraalkylammonium cations. J. Phys. Chem. B 108, 12271–12275 (2004).
Kragten, D. D. et al. Structure of the silica phase extracted from silica/(TPA)OH solutions containing nanoparticles. J. Phys. Chem. B 107, 10006–10016 (2003).
Schoeman, B. J. Analysis of the nucleation and growth of TPA-silicalite-1 at elevated temperatures with the emphasis on colloidal stability. Microporous Mesoporous Mater. 22, 9–22 (1998).
Schoeman, B. J. & Regev, O. A study of the initial stage in the crystallization of TPA-silicalite-1. Zeolites 17, 447–456 (1996).
Yang, S. Y., Navrotsky, A., Wesolowski, D. J. & Pople, J. A. Study on synthesis of TPA-silicalite-1 from initially clear solutions of various base concentrations by in situ calorimetry, potentiometry, and SAXS. Chem. Mater. 16, 210–219 (2004).
Stober, W. & Fink, A. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 26, 62–69 (1968).
Davis, T. M., Snyder, M. A., Krohn, J. E. & Tsapatsis, M. Nanoparticles in lysine–silica sols. Chem. Mater. 18, 5814–5816 (2006).
Snyder, M. A., Lee, J. A., Davis, T. M., Scriven, L. E. & Tsapatsis, M. Silica nanoparticle crystals and ordered coatings using Lys-Sil and a novel coating device. Langmuir 23, 9924–9928 (2007).
Glatter, O. New method for evaluation of small-angle scattering data. J. Appl. Cryst. 10, 415–421 (1977).
Glatter, O. Interpretation of real-space information from small-angle scattering experiments. J. Appl. Cryst. 12, 166–175 (1979).
Iler, R. K. The Chemistry of Silica (Wiley, 1979).
Chang, S. M., Lee, M. & Kim, W.-S. Preparation of large monodispersed spherical silica particles using seed particle growth. J. Colloid Interface Sci. 286, 536–542 (2005).
Giesche, H. Synthesis of monodispersed silica powders II. Controlled growth reaction and continuous production process. J. Eur. Ceram. Soc. 14, 205–214 (1994).
Dokter, W. H., Vangarderen, H. F., Beelen, T. P. M., Vansanten, R. A. & Bras, W. Homogeneous versus heterogeneous zeolite nucleation. Angew. Chem. Int. Ed. 34, 73–75 (1995).
Houssin, C. J. Y. et al. Combined in situ Si-29 NMR and small-angle X-ray scattering study of precursors in MFI zeolite formation from silicic acid in TPAOH solutions. Phys. Chem. Chem. Phys. 5, 3518–3524 (2003).
Mintova, S., Olson, N. H., Senker, J. & Bein, T. Mechanism of the transformation of silica precursor solutions into Si-MFI zeolite. Angew. Chem. Int. Ed. 41, 2558 (2002).
Mintova, S., Olson, N. H., Valtchev, V. & Bein, T. Mechanism of zeolite A nanocrystal growth from colloids at room temperature. Science 283, 958–960 (1999).
Nikolakis, V., Kokkoli, E., Tirrell, M., Tsapatsis, M. & Vlachos, D. G. Zeolite growth by addition of subcolloidal particles: Modeling and experimental validation. Chem. Mater. 12, 845–853 (2000).
Xu, W. Y., Dong, J. S., Li, J. P., Li, J. P. & Wu, F. A novel method for the preparation of zeolite ZSM-5. J. Chem. Soc., Chem. Commun. 755–756 (1990).
Talmon, Y. Modern Characterization Methods of Surfactant systems (Marcel Dekker, 1999).
Holland, B. T., Abrams, L. & Stein, A. Dual templating of macroporous silicates with zeolitic microporous frameworks. J. Am. Chem. Soc. 121, 4308–4309 (1999).
Acknowledgements
The authors gratefully acknowledge support for this work from the National Science Foundation (CMMI-0707610). The authors also acknowledge T. M. Davis for cryo-TEM images taken of the nanoparticle sols. Characterization was carried out at the Minnesota Characterization Facility, which receives support from the NSF through the National Nanotechnology Infrastructure Network.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
Supplementary Information (PDF 22903 kb)
Rights and permissions
About this article
Cite this article
Fan, W., Snyder, M., Kumar, S. et al. Hierarchical nanofabrication of microporous crystals with ordered mesoporosity. Nature Mater 7, 984–991 (2008). https://doi.org/10.1038/nmat2302
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat2302
This article is cited by
-
Elemental zoning enhances mass transport in zeolite catalysts for methanol to hydrocarbons
Nature Catalysis (2023)
-
Production of Trioxane from Formaldehyde via Hierarchical Beta Zeolite Synthesized Using a Cationic Polymer
Catalysis Letters (2023)
-
Nanoreactors for particle synthesis
Nature Reviews Materials (2022)
-
Crystal growth in confinement
Nature Communications (2022)
-
Dominance of heat transfer limitations in conventional sol-gel synthesis of LTA revealed by microcrystallization
Journal of Flow Chemistry (2022)