Abstract
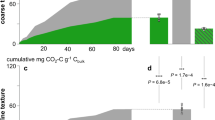
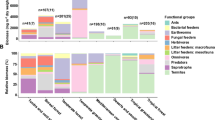
Soil organic matter is the largest terrestrial carbon pool1. The pool size depends on the balance between formation of soil organic matter from decomposition of plant litter and its mineralization to inorganic carbon. Knowledge of soil organic matter formation remains limited2 and current C numerical models assume that stable soil organic matter is formed primarily from recalcitrant plant litter3. However, labile components of plant litter could also form mineral-stabilized soil organic matter4. Here we followed the decomposition of isotopically labelled above-ground litter and its incorporation into soil organic matter over three years in a grassland in Kansas, USA, and used laboratory incubations to determine the decay rates and pool structure of litter-derived organic matter. Early in decomposition, soil organic matter formed when non-structural compounds were lost from litter. Soil organic matter also formed at the end of decomposition, when both non-structural and structural compounds were lost at similar rates. We conclude that two pathways yield soil organic matter efficiently. A dissolved organic matter–microbial path occurs early in decomposition when litter loses mostly non-structural compounds, which are incorporated into microbial biomass at high rates, resulting in efficient soil organic matter formation. An equally efficient physical-transfer path occurs when litter fragments move into soil.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Schlesinger, W. H. & Bernhardt, E. S. Biogeochemistry: An Analysis of Global Change (Academic Press, 2013).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Smith, P., Smith, J. U., Powlson, D. S., McGill, W. B. & Arah, J. R. M. A comparison of the performance of nine soil organic matter models using datasets from seven long-term experiments. Geoderma 81, 153–225 (1997).
Cotrufo, M. F., Wallenstein, M., Boot, M. C., Denef, K. & Paul, E. A. The Microbial Efficiency-Matrix Stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: Do labile plant inputs form stable soil organic matter? Glob. Change Biol. 19, 988–995 (2013).
Prescott, C. Litter decomposition: What controls it and how can we alter it to sequester more carbon in forest soils? Biogeochemistry 101, 133–149 (2010).
Adair, E. C. et al. Simple three-pool model accurately describes patterns of long-term litter decomposition in diverse climates. Glob. Change Biol. 14, 2636–2660 (2008).
Melillo, J. M., Aber, J. D. & Muratore, J. F. Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63, 621–626 (1982).
Marschner, B. et al. How relevant is recalcitrance for the stabilization of organic matter in soils? J. Plant Nutr. Soil Sci. 171, 91–110 (2008).
Grandy, A. S. & Neff, J. C. Molecular C dynamics downstream: The biochemical decomposition sequence and its impact on soil organic matter structure and function. Sci. Total Environ. 404, 297–307 (2008).
Miltner, A., Bombarch, P., Schmid, B. & Kastner, M. SOM genesis: Microbial biomass as a significant source. Biogeochemistry 111, 41–55 (2011).
Berg, B. & McClaugherty, C. Plant Litter: Decomposition, Humus Formation, Carbon Sequestration (Springer, 2008).
Voroney, R. P., Paul, E. A. & Anderson, D. W. Decomposition of wheat straw and stabilization of microbial products. Can. J. Soil Sci. 69, 63–77 (1989).
Martin, J. P., Haider, K., Farmer, W. J. & Fustecma, E. Decomposition and distribution of residual activity of some 14C-labeled microbial polysaccharides and cells, glucose, cellulose and wheat straw in soil. Soil Biol. Biochem. 6, 221–230 (1974).
Rubino, M. et al. Carbon input belowground is the major C flux contributing to leaf litter mass loss: Evidences from a 13C labelled-leaf litter experiment. Soil Biol. Biochem. 42, 1009–1016 (2010).
Soong, J. & Cotrufo, M. F. Annual burning of a tallgrass prairie inhibits C and N cycling in soil, increasing recalcitrant pyrogenic organic matter storage while reducing N availability. Glob. Change Biol. 21, 2321–2333 (2015).
Kaiser, K. & Kalbitz, K. Cycling downwards—dissolved organic matter in soils. Soil Biol. Biochem. 52, 29–32 (2012).
Mambelli, S., Bird, J. A., Gleixner, G., Dawson, T. E. & Torn, M. S. Relative contribution of foliar and fine root pine litter to the molecular composition of soil organic matter after in situ degradation. Org. Geochem. 42, 1099–1108 (2011).
Preston, C. M., Nault, J. R. & Trofymow, J. A. Chemical changes during 6 years of decomposition of 11 litters in some Canadian forest sites. Part 2. 13C abundance, solid-state 13C NMR spectroscopy and the meaning of “lignin”. Ecosystems 12, 1078–1102 (2009).
Klotzbucher, T., Kaiser, K., Guggenberger, G., Gatzek, C. & Kalbitz, K. A new conceptual model for the fate of lignin in decomposing plant litter. Ecology 92, 1052–1062 (2011).
Talbot, J. M. & Treseder, K. K. Interactions among lignin, cellulose, and nitrogen drive litter chemistry-decay relationships. Ecology 93, 345–354 (2012).
Hatton, P. J., Castanha, C., Torn, M. S. & Bird, J. A. Litter type control on soil C and N stabilization dynamics in a temperate forest. Glob. Change Biol. 21, 1358–1367 (2015).
Bird, J. A., Kleber, M. & Torn, M. S. C-13 and N-15 stabilization dynamics in soil organic matter fractions during needle and fine root decomposition. Org. Geochem. 39, 465–477 (2008).
Parton, W. et al. Global-scale similarities in nitrogen release patterns during long-term decomposition. Science 315, 362–364 (2007).
Castellano, M., Mueller, K., Olk, D., Sawyer, J. & Six, J. Integrating plant litter quality, soil organic matter stabilization and the carbon saturation concept. Glob. Change Biol. http://dx.doi.org/10.1111/gcb.12982 (2015).
Soong, J. L., Parton, W. J., Calderon, F. J., Campbell, N. & Cotrufo, M. F. A new conceptual model on the fate and controls of fresh and pyrolized plant litter decomposition. Biogeochemistry http://dx.doi.org/10.0007/s10533-015-0079-2 (2015).
Moorhead, D. L., Lashermes, G., Sinsabaugh, R. L. & Weintraub, M. N. Calculating co-metabolic costs of lignin decay and their impacts on carbon use efficiency. Soil Biol. Biochem. 66, 17–19 (2013).
Sinsabaugh, R. L., Manzoni, S., Moorhead, D. L. & Richter, A. Carbon use efficiency of microbial communities: Stoichiometry, methodology and modelling. Ecol. Lett. 16, 930–939 (2013).
Kleber, M., Sollins, P. & Sutton, R. A conceptual model of organo-mineral interactions in soils: Self-assembly of organic molecular fragments into zonal structures on mineral surfaces. Biogeochemistry 85, 9–24 (2007).
von Lützow, M. et al. Stabilization mechanisms of organic matter in four temperate soils: Development and application of a conceptual model. J. Plant Nutr. Soil Sci. 171, 111–124 (2008).
Knapp, A. K., Briggs, J. M., Hartnett, D. C. & Collins, S. L. Grassland Dynamics; Long-Term Ecological Research in Tallgrass Prairie (Oxford Univ. Press, 1998).
Knapp, A. K. et al. Rainfall variability, carbon cycling, and plant species diversity in a mesic grassland. Science 298, 2202–2205 (2002).
Soong, J. et al. Design and operation of a continuous 13C and 15N labeling chamber for uniform or differential, metabolic and structural, plant tissue isotope labeling. J. Vis. Exp. 83, 1–9 (2014).
Van Soest, P. J., Robertson, J. B. & Lewis, B. A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 74, 3583–3597 (1991).
Christensen, B. T. Physical fractionation of soil and structural and functional complexity in organic matter turnover. Eur. J. Soil Sci. 52, 345–353 (2001).
Gomez, J. D., Denef, K., Stewart, C. E., Zheng, J. & Cotrufo, M. F. Biochar addition rate influences soil microbial abundance and activity in temperate soils. Eur. J. Soil Sci. 65, 28–39 (2014).
Denef, K. et al. Community shifts and carbon translocation within metabolically-active rhizosphere microorganisms in grasslands under elevated CO2 . Biogeosciences 4, 769–779 (2007).
Stewart, C. E., Zheng, J., Botte, J. & Cotrufo, M. F. Co-generated fast pyrolysis biochar mitigates greenhouse gas emissions and increases carbon sequestration in temperate soils. Glob. Change Biol. Bioenergy 5, 153–164 (2013).
Efron, B. & Tibshirani, R. J. An Introduction to the Bootstrap (CRC Press, 1994).
Davison, A. C. & Hinkley, D. V. Bootstrap Methods and Their Applications (Cambridge Univ. Press, 1997).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2014).
Haddix, M. L. et al. The role of soil characteristics on temperature sensitivity of soil organic matter. Soil Sci. Soc. Am. J. 75, 56–68 (2011).
Acknowledgements
This work was supported by the National Science Foundation-Division of Environmental Biology grant no. 0918482 and the National Science Foundation Graduate Research Fellowship Program. Additional support during data analyses and writing came from the Cotrufo–Hoppess fund for soil ecological research and the OECD’s Cooperative Research Programme.
Author information
Authors and Affiliations
Contributions
M.F.C. developed the concepts, designed and led the research, contributed to data analyses and wrote the paper. J.L.S. and A.J.H. conducted all the research work and analyses. E.E.C. and M.L.H. contributed to data and statistical analyses. D.H.W. and W.J.P. were co-PI on the project and contributed to the development of research concepts. All authors contributed to writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 706 kb)
Rights and permissions
About this article
Cite this article
Cotrufo, M., Soong, J., Horton, A. et al. Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nature Geosci 8, 776–779 (2015). https://doi.org/10.1038/ngeo2520
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ngeo2520
This article is cited by
-
Carbon Stocks and Transfers in Coniferous Boreal Forests Along a Latitudinal Gradient
Ecosystems (2024)
-
Litter quality controls the contribution of microbial carbon to main microbial groups and soil organic carbon during its decomposition
Biology and Fertility of Soils (2024)
-
Contrasting effects of maize straw and its biochar on aggregation and soil organic matter stabilization
Plant and Soil (2024)
-
Biophysical Controls on Soil Carbon Cycling in a Northern Hardwood Forest
Ecosystems (2024)
-
Positive effects of vegetation restoration on the soil properties of post-mining land
Plant and Soil (2024)