Abstract
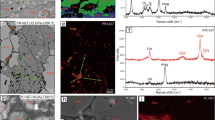
A significant fraction of Earth’s carbon resides in the mantle1, but the mode of carbon storage presents a long-standing problem. The mantle contains fluids rich in carbon dioxide and methane, carbonate-bearing melts, carbonate minerals, graphite, diamond and carbides, as well as dissolved carbon atoms in metals. However, it is uncertain whether these can sufficiently account for the total amount of carbon thought to be stored in the mantle and the volume of carbon degassed from the mantle at volcanoes2. Moreover, such carbon hosts should significantly affect the physical and chemical behaviour of the mantle, including its melting temperature, electrical conductivity and oxidation state3,4. Here we use in situ transmission electron microscopy to measure the storage of carbon within common mantle mineral analogues—nickel-doped lanthanum chromate perovskite and titanium dioxide—in laboratory experiments at high pressure and temperature. We detect elevated carbon concentrations at defect sites in the nanocrystals, maintained at high pressures within annealed carbon nanocages. Specifically, our experiments show that small stacking faults within the mantle analogue materials are effective carbon sinks at mantle conditions, potentially providing an efficient mechanism for carbon storage in the mantle. Furthermore, this carbon can be readily released under lower pressure conditions, and may therefore help to explain carbon release in volcanic eruptions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Jambon, A. Earth degassing and large-scale geochemical cycling of volatile elements. Rev. Mineral. 30, 479–518 (1994).
Dasgupta, R. & Hirschmann, M. M. The deep carbon cycle and melting in Earth’s interior. Earth Planet. Sci. Lett. 298, 1–13 (2010).
Gaillard, F., Malki, M., Iacono-Marziano, G., Pichavant, M. & Scaillet, B. Carbonatite melts and electrical conductivity in the asthenosphere. Science 322, 1363–1365 (2008).
Rohrbach, A. & Schmidt, M. W. Redox freezing and melting in the Earth’s deep mantle resulting from carbon–iron redox coupling. Nature 472, 209–212 (2011).
Keppler, H., Wiedenbeck, M. & Shcheka, S. S. Carbon solubility in olivine and the mode of carbon storage in the Earth’s mantle. Nature 424, 414–416 (2003).
Hayden, L. A. & Watson, E. B. Grain boundary mobility of carbon in Earth’s mantle: A possible carbon flux from the core. Proc. Natl Acad. Sci. USA 105, 8537–8541 (2008).
Kramar, N., Cosca, M. A. & Hunziker, J. C. Heterogeneous 40Ar distributions in naturally deformed muscovite: In situ UV-laser ablation evidence for microstructurally controlled intragrain diffusion. Earth Planet. Sci. Lett. 192, 377–388 (2001).
Blavette, D., Cadel, E., Fraczkiewicz, A. & Menand, A. Three-dimensional atomic-scale imaging of impurity segregation to line defects. Science 286, 2317–2319 (1999).
Rothová, V. & Buršı´k, J. Anomalous diffusion in presence of twin boundaries. J. Phys. Chem. Solids 68, 785–790 (2007).
Banhart, F. & Ajayan, P. M. Carbon onions as nanoscopic pressure cells for diamond formation. Nature 382, 433–435 (1996).
Sun, L. et al. Carbon nanotubes as high-pressure cylinders and nanoextruders. Science 312, 1199–1202 (2006).
Murakami, M., Hirose, K., Ono, S. & Ohishi, Y. Stability of CaCl2-type and α-PbO2-type SiO2 at high pressure and temperature determined by in-situ X-ray measurements. Geophys. Res. Lett. 30, 1207 (2003).
Swamy, V. et al. Compression behavior of nanocrystalline anatase TiO2 . Solid State Commun. 125, 111–115 (2003).
Swamy, V. et al. Size-dependent pressure-induced amorphization in nanoscale TiO2 . Phys. Rev. Lett. 96, 135702 (2006).
Vogt, T., Hriljac, J. A., Hyatt, N. C. & Woodward, P. Pressure-induced intermediate-to-low spin state transition in LaCoO3 . Phys. Rev. B 67, 140401(R) (2003).
Grogger, W., Schaffer, B., Krishnan, K. M. & Hofer, F. Energy-filtering TEM at high magnification: Spatial resolution and detection limits. Ultramicroscopy 96, 481–489 (2003).
Grogger, W. et al. Energy-filtering transmission electron microscopy on the nanometer length scale. J. Electron Spectrosc. Relat. Phenom. 143, 139–147 (2005).
Faulkner, R. G. Non-equilibrium grain-boundary segregation in austenitic alloys. J. Mater. Sci. 16, 373–383 (1981).
Hirth, J. P. & Lothe, J. Theory of Dislocations 763–764 (McGraw-Hill, 1968).
Zhang, X. & Lu, G. Calculation of fast pipe diffusion along a dislocation stacking fault ribbon. Phys. Rev. B 82, 012101 (2010).
Köhler, C., Hajnal, Z., Deák, P., Frauenheim, T. & Suhai, S. Theoretical investigation of carbon defects and diffusion in α-quartz. Phys. Rev. B 64, 085333 (2001).
Di Valentin, C., Pacchioni, G. & Selloni, A. Theory of carbon doping of titanium dioxide. Chem. Mater. 17, 6656–6665 (2005).
Dupas-Bruzek, C., Sharp, T. G., Rubie, D. C. & Durham, W. B. Mechanisms of transformation and deformation in Mg1.8Fe0.2SiO4 olivine and wadsleyite under non-hydrostatic stress. Phys. Earth Planet. Inter. 108, 33–48 (1998).
Wang, Y., Guyot, F. & Liebermann, R. C. Electron microscopy of (Mg,Fe)SiO3 perovskite: Evidence for structural phase transitions and implications for the lower mantle. J. Geophys. Res. 97, 12327–12347 (1992).
Oganov, A. R., Martoňák, R., Laio, A., Raiteri, P. & Parrinello, M. Anisotropy of Earth’s D” layer and stacking faults in the MgSiO3 post-perovskite phase. Nature 438, 1142–1144 (2005).
McNamara, A. K., van Keken, P. E. & Karato, S. I. Development of anisotropic structure in the Earth’s lower mantle by solid-state convection. Nature 416, 310–314 (2002).
Hÿtch, M. J. Geometric phase analysis of high resolution electron microscope images. Scanning Microsc. 11, 53–66 (1997).
Shilo, D., Ravichandran, G. & Bhattacharya, K. Investigation of twin-wall structure at the nanometre scale using atomic force microscopy. Nature Mater. 3, 453–457 (2004).
Solomatov, V. S., El-Khozondar, R. & Tikare, V. Grain size in the lower mantle: Constraints from numerical modeling of grain growth in two-phase systems. Phys. Earth Planet. Inter. 129, 265–282 (2002).
Kawahara, K., Tsurekawa, S. & Nakashima, H. in Towards Innovation in Superplasticity II (eds Sakuma, T., Aizawa, T. & Higashi, K.) 519–524 (Trans Tech Publications, 1999).
Acknowledgements
This work was financially supported by NSF grant EAR-0948535. We acknowledge the use of facilities within the LeRoy Eyring Center for Solid State Science at Arizona State University. D. Bell and B. Watson are thanked for helpful comments and K. Weiss and D. Wright for technical assistance.
Author information
Authors and Affiliations
Contributions
This was a joint project. J.W. initiated the idea and did all the experimental work with P.R.B. providing advice throughout the project. J.W. and P.R.B. jointly wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Wu, J., Buseck, P. Carbon storage at defect sites in mantle mineral analogues. Nature Geosci 6, 875–878 (2013). https://doi.org/10.1038/ngeo1903
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ngeo1903