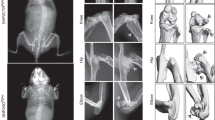
Abstract
Enchondromas are common benign cartilage tumors of bone. They can occur as solitary lesions or as multiple lesions in enchondromatosis (Ollier and Maffucci diseases). Clinical problems caused by enchondromas include skeletal deformity and the potential for malignant change to chondrosarcoma1,2,3. The extent of skeletal involvement is variable in enchondromatosis and may include dysplasia that is not directly attributable to enchondromas4. Enchondromatosis is rare, obvious inheritance of the condition is unusual and no candidate loci have been identified. Enchondromas are usually in close proximity to, or in continuity with, growth-plate cartilage. Consequently, they may result from abnormal regulation of proliferation and terminal differentiation of chondrocytes in the adjoining growth plate. In normal growth plates, differentiation of proliferative chondrocytes to post-mitotic hypertrophic chondrocytes is regulated in part by a tightly coupled signaling relay involving parathyroid hormone related protein (PTHrP) and Indian hedgehog (IHH)5,6,7,8,9. PTHrP delays the hypertrophic differentiation of proliferating chondrocytes, whereas IHH promotes chondrocyte proliferation. We identified a mutant PTH/PTHrP type I receptor (PTHR1) in human enchondromatosis that signals abnormally in vitro and causes enchondroma-like lesions in transgenic mice. The mutant receptor constitutively activates Hedgehog signaling, and excessive Hedgehog signaling is sufficient to cause formation of enchondroma-like lesions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Levesque, J. et al. Enchondroma. in A Clinical Guide to Primary Bone Tumors 98–107 (Williams and Wilkins, Philadelphia, 1998).
Huvos, A.G. Solitary and multiple osteochondromas and enchondromas: jusxtacortical chondroma; maffuci's disease. in Bone Tumors, Diagnosis Treatment and Prognosis. 264–294 (WB Saunders, Philadelphia, 1991).
Schwartz, H.S. et al. The malignant potential of enchondromatosis. J. Bone Joint Surg. Am. 69, 269–274 (1987).
Zack, P. & Beighton, P. Spondyloenchondromatosis: syndromic identity and evolution of the phenotype. Am. J. Med. Genet. 55, 478–482 (1995).
Vortkamp, A. et al. Regulation of rate of cartilage differentiation by Indian hedgehog and PTH-related protein. Science 273, 613–622 (1996).
Lanske, B. et al. PTH/PTHrP receptor in early development and Indian hedgehog-regulated bone growth. Science 273, 663–666 (1996).
Karp, S.J. et al. Indian hedgehog coordinates endochondral bone growth and morphogenesis via parathyroid hormone related-protein-dependent and -independent pathways. Development 127, 543–548 (2000).
St. Jacques, B., Hammerschmidt, M. & McMahon, A.P. Indian hedgehog signaling regulates proliferation and differentiation of chondrocytes and is essential for bone formation. Genes Dev. 13, 2072–2086 (1999).
Chung, U.I., Lanske, B., Lee, K., Li, E. & Kronenberg, H. The parathyroid hormone/parathyroid hormone-related peptide receptor coordinates endochondral bone development by directly controlling chondrocyte differentiation. Proc. Natl Acad. Sci. USA 95, 13030–13035 (1998).
Halal, F. & Azouz, E.M. Generalized enchondromatosis in a boy with only platyspondyly in the father. Am. J. Med. Genet. 38, 588–592 (1991).
Juppner, H. et al. A G protein-linked receptor for parathyroid hormone and parathyroid hormone-related peptide. Science 254, 1024–1026 (1991).
Schipani, E. et al. Constitutively activated receptors for parathyroid hormone and parathyroid hormone-related peptide in Jansen's metaphyseal chondrodysplasia. N. Engl. J. Med. 335, 708–714 (1996).
Schipani, E., Kruse, K. & Juppner, H. A constitutively active mutant PTH-PTHrP receptor in Jansen-type metaphyseal chondrodysplasia. Science 268, 98–100 (1995).
Jobert, A.S. et al. Absence of functional receptors for parathyroid hormone and parathyroid hormone-related peptide in Blomstrand chondrodysplasia. J. Clin. Invest. 102, 34–40 (1998).
Karaplis, A.C. et al. Inactivating mutation in the human parathyroid hormone receptor type 1 gene in Blomstrand chondrodysplasia. Endocrinology 139, 5255–5258 (1998).
Guo, J., Chung, U., Bringhurst, R.F. & Kronenberg, H.M. Failure of phospholipase C activation via the PTH/PTHrP receptor leads to abnormalities in embryonic bone development. J. Bone Miner. Res. 15, S152 (2000).
Jaffe, H.L. Certain other anomalies of skeletal development. in Metabolic, Degenerative, and Inflammatory Diseases of Bones and Joints 222–226 (Lea and Feibiger, Philadelphia, 1972).
Bruggeman, L.A., Xie, H.X., Brown, K.S. & Yamada, Y. Developmental regulation for collagen II gene expression in transgenic mice. Teratology 44, 203–208 (1991).
Yamada, Y. et al. Regulation of the collagen II gene in vitro and in transgenic mice. Ann. NY Acad. Sci. 580, 81–87 (1990).
O'Keefe, R.J., Puzas, J.E., Loveys, L., Hicks, D.G. & Rosier, R.N. Analysis of type II and type X collagen synthesis in cultured growth plate chondrocytes by in situ hybridization: rapid induction of type X collagen in culture. J. Bone Miner. Res. 9, 1713–1722 (1994).
Vu, T.H. et al. MMP-9/gelatinase B is a key regulator of growth plate angiogenesis and apoptosis of hypertrophic chondrocytes. Cell 93, 411–422 (1998).
Mo, R. et al. Specific and redundant functions of Gli2 and Gli3 zinc finger genes in skeletal patterning and development. Development 124, 113–123 (1997).
Sasaki, H., Nishizaki, Y., Hui, C., Nakafuku, M. & Kondoh, H. Regulation of Gli2 and Gli3 activities by an amino-terminal repression domain: implication of Gli2 and Gli3 as primary mediators of Shh signaling. Development 126, 3915–3924 (1999).
Grachtchouk, M. et al. Basal cell carcinomas in mice overexpressing Gli2 in skin. Nature Genet. 24, 216–217 (2000).
Hopyan, S. et al. Expression of osteocalcin and its transcriptional regulators core-binding factor α 1 and MSX2 in osteoid-forming tumours. J. Orthop. Res. 17, 633–638 (1999).
Xie, L.Y. & Abou-Samra, A.B. Epitope tag mapping of the extracellular and cytoplasmic domains of the rat parathyroid hormone (PTH)/PTH-related peptide receptor. Endocrinology 139, 4563–4567 (1998).
Li, C., Bapat, B. & Alman, B. A. Adenomatous polyposis coli gene mutation alters proliferation through its beta-catenin-regulatory function in aggressive fibromatosis (desmoid tumor). Am. J. Pathol. 153, 709–714 (1998).
Carter, P.H., Jüppner, H. & Gardella, T.J. Studies of the N-terminal region of a parathyroid hormone-related peptide (1-36) analog: receptor subtype-selective agonists, antagonists, and photochemical cross-linking agents. Endocrinology 140, 4972–4981 (1999).
Schipani E, et al. Pseudohypoparathyroidism type Ib is not caused by mutations in the coding exons of the human parathyroid hormone (PTH)/PTH-related peptide receptor gene. J. Clin. Endocrinol. Metab. 80, 1611–1621 (1995).
Acknowledgements
We thank affected individuals and their families for their cooperation, C.-C. Hui for Gli2 and Gli2-luciferase cDNA, U.-I. Chung for PTHR1−/− embryonic stem cells, Y. Yamada for Col2A1 promoter/enhancer cDNA, T. Ingolia (Ontogeny) for Shh-N protein, A. Karaplis for c-Myc–tagged PTHR1 cDNA (Hkrk), F. de Sauvage (Genentech) for Smo cDNA, S. Tondat and L. Schwartz for pronuclear microinjections, L. Morikawa for histology, M. Ho for anti-c-Myc immunohistochemistry, M. To for assistance with cAMP assays, S. Cheon for assistance with SSCP analysis, A. Davis for help with statistical analysis, J. Ahier for patient contact, members of the Andrulis and Alman labs for support, A. Gross, C. Hutchison and J. Wright for tissue specimens; and J. Aubin, U. Chung, J. Dennis, S. Egan, J. Haigh, C.-C. Hui, H. Kronenberg and J. Rossant for critical advice. This work was supported by grants from the National Cancer Institute of Canada with funds from the Terry Fox Run (to B.A.A., I.L.A. and R.S.B.), the Canadian Institutes of Health Research (to J.S.W., B.A.A. and W.G.C.) and The Hospital for Sick Children Research Institute Trainees' Start-Up Fund (to S.H.). S.H. is supported by a Clinician-Scientist Fellowship from the Research Training Centre of The Hospital for Sick Children Research Institute, and B.A.A. is supported by the Canadian Research Chair Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hopyan, S., Gokgoz, N., Poon, R. et al. A mutant PTH/PTHrP type I receptor in enchondromatosis. Nat Genet 30, 306–310 (2002). https://doi.org/10.1038/ng844
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng844
This article is cited by
-
New bone formation accelerates during lower limb lengthening and deformity correction in children with Ollier’s disease
Journal of Orthopaedics and Traumatology (2023)
-
Enchondromatosis and Growth Plate Development
Current Osteoporosis Reports (2021)
-
The role of GPCRs in bone diseases and dysfunctions
Bone Research (2019)
-
Widespread and debilitating hemangiomas in a patient with enchondromatosis and D-2-hydroxyglutaric aciduria
Skeletal Radiology (2018)
-
Hereditäre Knochentumoren
Der Pathologe (2017)