Abstract
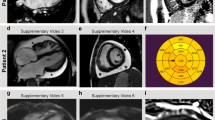
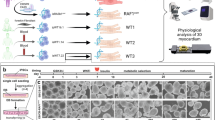
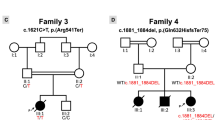
Noonan and LEOPARD syndromes are developmental disorders with overlapping features, including cardiac abnormalities, short stature and facial dysmorphia. Increased RAS signaling owing to PTPN11, SOS1 and KRAS mutations causes ∼60% of Noonan syndrome cases1,2,3,4,5,6, and PTPN11 mutations cause 90% of LEOPARD syndrome cases7. Here, we report that 18 of 231 individuals with Noonan syndrome without known mutations (corresponding to 3% of all affected individuals) and two of six individuals with LEOPARD syndrome without PTPN11 mutations have missense mutations in RAF1, which encodes a serine-threonine kinase that activates MEK1 and MEK2. Most mutations altered a motif flanking Ser259, a residue critical for autoinhibition of RAF1 through 14-3-3 binding. Of 19 subjects with a RAF1 mutation in two hotspots, 18 (or 95%) showed hypertrophic cardiomyopathy (HCM), compared with the 18% prevalence of HCM among individuals with Noonan syndrome in general. Ectopically expressed RAF1 mutants from the two HCM hotspots had increased kinase activity and enhanced ERK activation, whereas non–HCM-associated mutants were kinase impaired. Our findings further implicate increased RAS signaling in pathological cardiomyocyte hypertrophy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Carta, C. et al. Germline missense mutations affecting KRAS isoform B are associated with a severe Noonan syndrome phenotype. Am. J. Hum. Genet. 79, 129–135 (2006).
Kontaridis, M.I., Swanson, K.D., David, F.S., Barford, D. & Neel, B.G. PTPN11 (Shp2) mutations in LEOPARD syndrome have dominant negative, not activating, effects. J. Biol. Chem. 281, 6785–6792 (2006).
Schubbert, S. et al. Germline KRAS mutations cause Noonan syndrome. Nat. Genet. 38, 331–336 (2006).
Tartaglia, M. et al. Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome. Nat. Genet. 29, 465–468 (2001).
Roberts, A.E. et al. Germline gain-of-function mutations in SOS1 cause Noonan syndrome. Nat. Genet. 39, 70–74 (2007).
Tartaglia, M. et al. Gain-of-function SOS1 mutations cause a distinctive form of Noonan syndrome. Nat. Genet. 39, 75–79 (2007).
Digilio, M.C. et al. Grouping of multiple-lentigines/LEOPARD and Noonan syndromes on the PTPN11 Gene. Am. J. Hum. Genet. 71, 389–394 (2002).
Aoki, Y. et al. Germline mutations in HRAS proto-oncogene cause Costello syndrome. Nat. Genet. 37, 1038–1040 (2005).
Niihori, T. et al. Germline KRAS and BRAF mutations in cardio-facio-cutaneous syndrome. Nat. Genet. 38, 294–296 (2006).
Rodriguez-Viciana, P. et al. Germline mutations in genes within the MAPK pathway cause cardio-facio-cutaneous syndrome. Science 311, 1287–1290 (2006).
Tartaglia, M. et al. Diversity and functional consequences of germline and somatic PTPN11 mutations in human disease. Am. J. Hum. Genet. 78, 279–290 (2006).
Tartaglia, M. et al. PTPN11 mutations in Noonan syndrome: molecular spectrum, genotype-phenotype correlation, and phenotypic heterogeneity. Am. J. Hum. Genet. 70, 1555–1563 (2002).
Burch, M. et al. Cardiologic abnormalities in Noonan syndrome: phenotypic diagnosis and echocardiographic assessment of 118 patients. J. Am. Coll. Cardiol. 22, 1189–1192 (1993).
Van Driest, S.L. et al. Myosin binding protein C mutations and compound heterozygosity in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 44, 1903–1910 (2004).
Wellbrock, C., Karasarides, M. & Marais, R. The RAF proteins take centre stage. Nat. Rev. Mol. Cell Biol. 5, 875–885 (2004).
Emuss, V., Garnett, M., Mason, C. & Marais, R. Mutations of C-RAF are rare in human cancer because C-RAF has a low basal kinase activity compared with B-RAF. Cancer Res. 65, 9719–9726 (2005).
Zebisch, A. et al. Two transforming C-RAF germ-line mutations identified in patients with therapy-related acute myeloid leukemia. Cancer Res. 66, 3401–3408 (2006).
Greenman, C. et al. Patterns of somatic mutation in human cancer genomes. Nature 446, 153–158 (2007).
Muslin, A.J., Tanner, J.W., Allen, P.M. & Shaw, A.S. Interaction of 14–3-3 with signaling proteins is mediated by the recognition of phosphoserine. Cell 84, 889–897 (1996).
Light, Y., Paterson, H. & Marais, R. 14–3-3 antagonizes Ras-mediated Raf-1 recruitment to the plasma membrane to maintain signaling fidelity. Mol. Cell. Biol. 22, 4984–4996 (2002).
Wan, P.T. et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell 116, 855–867 (2004).
Muslin, A.J. Role of raf proteins in cardiac hypertrophy and cardiomyocyte survival. Trends Cardiovasc. Med. 15, 225–229 (2005).
Bueno, O.F. et al. The MEK1–ERK1/2 signaling pathway promotes compensated cardiac hypertrophy in transgenic mice. EMBO J. 19, 6341–6350 (2000).
Hunter, J.J., Tanaka, N., Rockman, H.A., Ross, J. Jr. & Chien, K.R. Ventricular expression of a MLC-2v-ras fusion gene induces cardiac hypertrophy and selective diastolic dysfunction in transgenic mice. J. Biol. Chem. 270, 23173–23178 (1995).
Gripp, K.W. et al. HRAS mutation analysis in Costello syndrome: genotype and phenotype correlation. Am. J. Med. Genet. A. 140, 1–7 (2006).
Yamaguchi, O. et al. Cardiac-specific disruption of the c-raf-1 gene induces cardiac dysfunction and apoptosis. J. Clin. Invest. 114, 937–943 (2004).
Patel, R. et al. Simvastatin induces regression of cardiac hypertrophy and fibrosis and improves cardiac function in a transgenic rabbit model of human hypertrophic cardiomyopathy. Circulation 104, 317–324 (2001).
Sarkozy, A. et al. Clinical and molecular analysis of 30 patients with multiple lentigines LEOPARD syndrome. J. Med. Genet. 41, e68 (2004).
Ucar, C., Calyskan, U., Martini, S. & Heinritz, W. Acute myelomonocytic leukemia in a boy with LEOPARD syndrome (PTPN11 gene mutation positive). J. Pediatr. Hematol. Oncol. 28, 123–125 (2006).
Merks, J.H., Caron, H.N. & Hennekam, R.C. High incidence of malformation syndromes in a series of 1,073 children with cancer. Am. J. Med. Genet. A. 134, 132–143 (2005).
Acknowledgements
We are indebted to the affected individuals and families who participated in the study, the physicians who referred the subjects and the Joint Genome Institute's production sequencing group. We thank D.K. Morrison (National Cancer Institute, US National Institutes of Health) for critical reagents and suggestions. We also thank G. Crisponi (Clinica Sant'Anna, Cagliari, Italy), F. Faravelli (Ospedale Galliera, Genova, Italy), L. Memo (ULLS9, Treviso, Italy) and F. Stanzial (Regional Hospital, Bolzano, Italy) for their valuable clinical assistance. This work was supported by Telethon-Italy grant GGP07115 (M.T.) and Programma di Collaborazione Italia-USA/malattie rare 2007 grants (M.T. and A.S.); US National Institutes of Health Grants HL71207, HD01294, HL074728 (B.D.G.), HD042569 (M.J.A.) and DK57683 (P.M.); the American Heart Association (M.J.A.); the Dr. Scholl Foundation (M.J.A.); the CJ Foundation for SIDS and the Mayo Foundation (M.J.A.) and Italian Ministry of Health Grant RC 2006-2007, Italian Ministry of University and Research C26A06KTTN and RBIP06PMF2_005 (B.D.). Research conducted at the E.O. Lawrence Berkeley National Laboratory and the Joint Genome Institute was performed under the Berkeley Program for Genomic Applications, funded by the US National Heart, Lung and Blood Institute (HL066681) and Department of Energy Contract DE-AC02-05CH11231 (University of California). We regret our inability to cite certain references describing work similar to references that were cited due to the limits of this journal.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1, Supplementary Table 2, Supplementary Figure 1 (PDF 517 kb)
Rights and permissions
About this article
Cite this article
Pandit, B., Sarkozy, A., Pennacchio, L. et al. Gain-of-function RAF1 mutations cause Noonan and LEOPARD syndromes with hypertrophic cardiomyopathy. Nat Genet 39, 1007–1012 (2007). https://doi.org/10.1038/ng2073
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng2073
This article is cited by
-
The role of CRAF in cancer progression: from molecular mechanisms to precision therapies
Nature Reviews Cancer (2024)
-
Temporospatial inhibition of Erk signaling is required for lymphatic valve formation
Signal Transduction and Targeted Therapy (2023)
-
RASopathies and sigmoid-shaped ventricular septum morphology: evidence of a previously unappreciated cardiac phenotype
Pediatric Research (2023)
-
An Assessment of the Therapeutic Landscape for the Treatment of Heart Disease in the RASopathies
Cardiovascular Drugs and Therapy (2023)
-
Molecular and cellular evidence for the impact of a hypertrophic cardiomyopathy-associated RAF1 variant on the structure and function of contractile machinery in bioartificial cardiac tissues
Communications Biology (2023)