Abstract
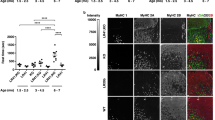
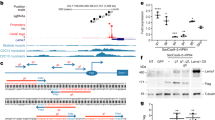
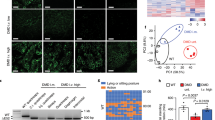
Myotonic dystrophy (DM1), the most common muscular dystrophy in adults, is caused by an expanded (CTG)n tract in the 3′ UTR of the gene encoding myotonic dystrophy protein kinase (DMPK)1, which results in nuclear entrapment of the 'toxic' mutant RNA and interacting RNA-binding proteins (such as MBNL1) in ribonuclear inclusions2. It is unclear if therapy aimed at eliminating the toxin would be beneficial. To address this, we generated transgenic mice expressing the DMPK 3′ UTR as part of an inducible RNA transcript encoding green fluorescent protein (GFP). We were surprised to find that mice overexpressing a normal DMPK 3′ UTR mRNA reproduced cardinal features of myotonic dystrophy, including myotonia, cardiac conduction abnormalities, histopathology and RNA splicing defects in the absence of detectable nuclear inclusions. However, we observed increased levels of CUG-binding protein (CUG-BP1) in skeletal muscle, as seen in individuals with DM1. Notably, these effects were reversible in both mature skeletal and cardiac muscles by silencing transgene expression. These results represent the first in vivo proof of principle for a therapeutic strategy for treatment of myotonic dystrophy by ablating or silencing expression of the toxic RNA molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
18 August 2006
In the version of this article initially published online, the sentence at the bottom of page 2 misrepresented one of the authors' results. It should read, “Furthermore, we performed RT-PCR for Clcn-1 and Tnnt3 in our mice and uncovered splicing abnormalities (Fig. 2c) similar to those in transgenic mice overexpressing CUG repeats15, in the Mbnl1ΔE3 knockout mouse16 and in individuals with myotonic dystrophy14–16.” The error has been corrected for all versions of the article.
References
Mahadevan, M. et al. Myotonic dystrophy mutation: an unstable CTG repeat in the 3′ untranslated region of the gene. Science 255, 1253–1255 (1992).
Day, J.W. & Ranum, L.P. RNA pathogenesis of the myotonic dystrophies. Neuromuscul. Disord. 15, 5–16 (2005).
Taneja, K.L., McCurrach, M., Schalling, M., Housman, D. & Singer, R.H. Foci of trinucleotide repeat transcripts in nuclei of myotonic dystrophy cells and tissues. J. Cell Biol. 128, 995–1002 (1995).
Davis, B.M., McCurrach, M.E., Taneja, K.L., Singer, R.H. & Housman, D.E. Expansion of a CUG trinucleotide repeat in the 3′ untranslated region of myotonic dystrophy protein kinase transcripts results in nuclear retention of transcripts. Proc. Natl. Acad. Sci. USA 94, 7388–7393 (1997).
Amack, J.D., Paguio, A.P. & Mahadevan, M.S. Cis and trans effects of the myotonic dystrophy (DM) mutation in a cell culture model. Hum. Mol. Genet. 8, 1975–1984 (1999).
Furling, D., Lemieux, D., Taneja, K. & Puymirat, J. Decreased levels of myotonic dystrophy protein kinase (DMPK) and delayed differentiation in human myotonic dystrophy myoblasts. Neuromuscul. Disord. 11, 728–735 (2001).
Mankodi, A. et al. Myotonic dystrophy in transgenic mice expressing an expanded CUG repeat. Science 289, 1769–1773 (2000).
Berul, C.I., Maguire, C.T., Gehrmann, J. & Reddy, S. Progressive atrioventricular conduction block in a mouse myotonic dystrophy model. J. Interv. Card. Electrophysiol. 4, 351–358 (2000).
O'Cochlain, D.F. et al. Transgenic overexpression of human DMPK accumulates into hypertrophic cardiomyopathy, myotonic myopathy and hypotension traits of myotonic dystrophy. Hum. Mol. Genet. 13, 2505–2518 (2004).
Storbeck, C.J., Sabourin, L.A., Waring, J.D. & Korneluk, R.G. Definition of regulatory sequence elements in the promoter region and the first intron of the myotonic dystrophy protein kinase gene. J. Biol. Chem. 273, 9139–9147 (1998).
Jansen, G. et al. Abnormal myotonic dystrophy protein kinase levels produce only mild myopathy in mice. Nat. Genet. 13, 316–324 (1996).
Mankodi, A. et al. Muscleblind localizes to nuclear foci of aberrant RNA in myotonic dystrophy types 1 and 2. Hum. Mol. Genet. 10, 2165–2170 (2001).
Melacini, P. et al. Correlation between cardiac involvement and CTG trinucleotide repeat length in myotonic dystrophy. J. Am. Coll. Cardiol. 25, 239–245 (1995).
Charlet, B.N. et al. Loss of the muscle-specific chloride channel in type 1 myotonic dystrophy due to misregulated alternative splicing. Mol. Cell 10, 45–53 (2002).
Mankodi, A. et al. Expanded CUG repeats trigger aberrant splicing of ClC-1 chloride channel pre-mRNA and hyperexcitability of skeletal muscle in myotonic dystrophy. Mol. Cell 10, 35–44 (2002).
Kanadia, R.N. et al. A muscleblind knockout model for myotonic dystrophy. Science 302, 1978–1980 (2003).
Timchenko, N.A. et al. Overexpression of CUG triplet repeat-binding protein, CUGBP1, in mice inhibits myogenesis. J. Biol. Chem. 279, 13129–13139 (2004).
Savkur, R.S., Philips, A.V. & Cooper, T.A. Aberrant regulation of insulin receptor alternative splicing is associated with insulin resistance in myotonic dystrophy. Nat. Genet. 29, 40–47 (2001).
Mankodi, A., Lin, X., Blaxall, B.C., Swanson, M.S. & Thornton, C.A. Nuclear RNA foci in the heart in myotonic dystrophy. Circ. Res. 97, 1152–1155 (2005).
Storbeck, C.J. et al. Inhibition of myogenesis in transgenic mice expressing the human DMPK 3′-UTR. Hum. Mol. Genet. 13, 589–600 (2004).
Seznec, H. et al. Mice transgenic for the human myotonic dystrophy region with expanded CTG repeats display muscular and brain abnormalities. Hum. Mol. Genet. 10, 2717–2726 (2001).
Lin, X. et al. Failure of MBNL1-dependent postnatal splicing transitions in myotonic dystrophy. Hum. Mol. Genet. 15, 2087–2097 (2006).
Margolis, J.M., Schoser, B.G., Moseley, M.L., Day, J.W. & Ranum, L.P. DM2 intronic expansions: evidence for CCUG accumulation without flanking sequence or effects on ZNF9 mRNA processing or protein expression. Hum. Mol. Genet. 15, 1808–1815 (2006).
Amack, J.D. & Mahadevan, M.S. The myotonic dystrophy expanded CUG repeat tract is necessary but not sufficient to disrupt C2C12 myoblast differentiation. Hum. Mol. Genet. 10, 1879–1887 (2001).
Houseley, J.M. et al. Myotonic dystrophy associated expanded CUG repeat muscleblind positive ribonuclear foci are not toxic to Drosophila. Hum. Mol. Genet. 14, 873–883 (2005).
Ho, T.H. et al. Colocalization of muscleblind with RNA foci is separable from mis-regulation of alternative splicing in myotonic dystrophy. J. Cell Sci. 118, 2923–2933 (2005).
Ho, T.H. et al. Muscleblind proteins regulate alternative splicing. EMBO J. 23, 3103–3112 (2004).
Ho, T.H., Bundman, D., Armstrong, D.L. & Cooper, T.A. Transgenic mice expressing CUG-BP1 reproduce splicing mis-regulation observed in myotonic dystrophy. Hum. Mol. Genet. 14, 1539–1547 (2005).
Mahadevan, M.S. et al. Structure and genomic sequence of the myotonic dystrophy (DM kinase) gene. Hum. Mol. Genet. 2, 299–304 (1993).
Langlois, M.A., Lee, N.S., Rossi, J.J. & Puymirat, J. Hammerhead ribozyme-mediated destruction of nuclear foci in myotonic dystrophy myoblasts. Mol. Ther. 7, 670–680 (2003).
Acknowledgements
We wish to thank P. Mahadevan and A. Tucker for their insights and continued support. MBNL antibodies were provided by M. Swanson and C. Thornton. Human tissues were provided by J. Puymirat and C. Thornton and purchased from the University of Miami Brain and Tissue Bank. Mouse tissues from other myotonic dystrophy models were provided by J. Puymirat, B. Wieringa and G. Gourdon. Transgenic mice were generated by the University of Wisconsin-Madison Transgenic Core Facility. All studies were done under the auspices of the University of Virginia Animal Care and Use Committee and Institutional Review Board. This work was supported by the Muscular Dystrophy Association and the US National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Author information
Authors and Affiliations
Contributions
M.S.M., R.S.Y., Q.Y., C.D.F.-M., T.D.B. and L.H.P. performed experimental work and data analysis. S.B. generated the transgene constructs. M.S.M. was responsible for conceptual design and execution.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Transgene expression. (PDF 477 kb)
Supplementary Fig. 2
RNA foci in all three muscle lineages in (CTG)200 mice. (PDF 202 kb)
Supplementary Fig. 3
CUG-BP1 levels elevated in DM1 muscle. (PDF 208 kb)
Supplementary Fig. 4
CUG-BP1 levels increased by toxic RNA. (PDF 288 kb)
Supplementary Fig. 5
Comparison of transgene expression in different mouse models of myotonic dystrophy. (PDF 496 kb)
Supplementary Fig. 6
Model for the splicing balance created by the mutual antagonism between MBNL1 and CUG-BP1 for splicing events. (PDF 429 kb)
Rights and permissions
About this article
Cite this article
Mahadevan, M., Yadava, R., Yu, Q. et al. Reversible model of RNA toxicity and cardiac conduction defects in myotonic dystrophy. Nat Genet 38, 1066–1070 (2006). https://doi.org/10.1038/ng1857
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1857
This article is cited by
-
Myotonic dystrophy RNA toxicity alters morphology, adhesion and migration of mouse and human astrocytes
Nature Communications (2022)
-
Antisense oligonucleotides: the next frontier for treatment of neurological disorders
Nature Reviews Neurology (2018)
-
Non-invasive monitoring of alternative splicing outcomes to identify candidate therapies for myotonic dystrophy type 1
Nature Communications (2018)
-
Contracting CAG/CTG repeats using the CRISPR-Cas9 nickase
Nature Communications (2016)
-
Dynamic changes of nuclear RNA foci in proliferating DM1 cells
Histochemistry and Cell Biology (2015)