Abstract
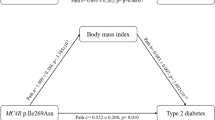
We identified a locus on chromosome 6q16.3–q24.2 (ref. 1) associated with childhood obesity that includes 2.4 Mb common to eight genome scans for type 2 diabetes (T2D) or obesity1,2,3,4,5,6,7,8. Analysis of the gene ENPP1 (also called PC-1), a candidate for insulin resistance9,10, in 6,147 subjects showed association between a three-allele risk haplotype (K121Q, IVS20delT–11 and A→G+1044TGA; QdelTG) and childhood obesity (odds ratio (OR) = 1.69, P = 0.0006), morbid or moderate obesity in adults (OR = 1.50, P = 0.006 or OR = 1.37, P = 0.02, respectively) and T2D (OR = 1.56, P = 0.00002). The Genotype IBD Sharing Test suggested that this obesity-associated ENPP1 risk haplotype contributes to the observed chromosome 6q linkage with childhood obesity. The haplotype confers a higher risk of glucose intolerance and T2D to obese children and their parents and associates with increased serum levels of soluble ENPP1 protein in children. Expression of a long ENPP1 mRNA isoform, which includes the obesity-associated A→G+1044TGA SNP, was specific for pancreatic islet beta cells, adipocytes and liver. These findings suggest that several variants of ENPP1 have a primary role in mediating insulin resistance and in the development of both obesity and T2D, suggesting that an underlying molecular mechanism is common to both conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Meyre, D. et al. A genome-wide scan for childhood obesity-associated traits in French families shows significant linkage on chromosome 6q22.31-q23.2. Diabetes 53, 803–811 (2004).
Atwood, L.D. et al. Genomewide linkage analysis of body mass index across 28 years of the Framingham heart study. Am. J. Hum. Genet. 71, 1044–1050 (2002).
Duggirala, R. et al. A major locus for fasting insulin concentrations and insulin resistance on chromosome 6q with strong pleiotropic effects on obesity-related phenotypes in nondiabetic Mexican Americans. Am. J. Hum. Genet. 68, 1149–1164 (2001).
Abney, M., Ober, C. & McPeek, M.S. Quantitative-trait homozygosity and association mapping and empirical genomewide significance in large, complex pedigrees: fasting serum-insulin level in the Hutterites. Am. J. Hum. Genet. 70, 920–934 (2002).
Ehm, M.G. et al. Genomewide search for type 2 diabetes susceptibility genes in four American populations. Am. J. Hum. Genet. 66, 1871–1881 (2000).
Ghosh, S. et al. The Finland-United States investigation of non-insulin-dependent diabetes mellitus genetics (FUSION) study. I. An autosomal genome scan for genes that predispose to type 2 diabetes. Am. J. Hum. Genet. 67, 1174–1185 (2000).
Demenais, F. et al. A meta-analysis of four European genome screens (GIFT Consortium) shows evidence for a novel region on chromosome 17p11.2-q22 linked to type 2 diabetes. Hum. Mol. Genet. 12, 1865–1873 (2003).
Xiang, K. et al. Genome-wide search for type 2 diabetes/impaired glucose homeostasis susceptibility genes in the Chinese: significant linkage to chromosome 6q21-q23 and chromosome 1q21-q24. Diabetes 53, 228–234 (2004).
Maddux, B.A. et al. Membrane glycoprotein PC-1 and insulin resistance in non-insulin-dependent diabetes mellitus. Nature 373, 448–451 (1995).
Dong, H. et al. Increased hepatic levels of the insulin receptor inhibitor, PC-1/NPP1, induce insulin resistance and glucose intolerance. Diabetes 54, 367–372 (2005).
Maddux, B.A. & Goldfine, I.D. Membrane glycoprotein PC-1 inhibition of insulin receptor function occurs via direct interaction with the receptor alpha-subunit. Diabetes 49, 13–19 (2000).
Hager, J. et al. A genome-wide scan for human obesity genes reveals a major susceptibility locus on chromosome 10. Nat. Genet. 20, 304–308 (1998).
Eaves, I.A. et al. Transmission ratio distortion at the INS-IGF2 VNTR. Nat. Genet. 22, 324–325 (1999).
Li, C., Scott, L.J. & Boehnke, M. Assessing whether an allele can account in part for a linkage signal: the Genotype-IBD Sharing Test (GIST). Am. J. Hum. Genet. 74, 418–431 (2004).
Costanzo, B.V. et al. The Q allele variant (GLN121) of membrane glycoprotein PC-1 interacts with the insulin receptor and inhibits insulin signaling more effectively than the common K allele variant (LYS121). Diabetes 50, 831–836 (2001).
Rutsch, F. et al. PC-1 nucleoside triphosphate pyrophosphohydrolase deficiency in idiopathic infantile arterial calcification. Am. J. Pathol. 158, 543–554 (2001).
Weill, J., Vanderbecken, S. & Froguel, P. Understanding the rising incidence of type 2 diabetes in adolescence. Arch. Dis. Child. 89, 502–504 (2004).
Stefanovic, V. & Antic, S. Plasma cell membrane glycoprotein 1 (PC-1): a marker of insulin resistance in obesity, uremia and diabetes mellitus. Clin. Lab. 50, 271–278 (2004).
Bruning, J.C. et al. Role of brain insulin receptor in control of body weight and reproduction. Science 289, 2122–2125 (2000).
Kim, J.K. et al. Redistribution of substrates to adipose tissue promotes obesity in mice with selective insulin resistance in muscle. J. Clin. Invest. 105, 1791–1797 (2000).
Barroso, I. et al. Candidate gene association study in type 2 diabetes indicates a role for genes involved in beta-cell function as well as insulin action. PLoS Biol. 1, E20 (2003).
Johnson, M.S., Figueroa-Colon, R., Huang, T.T., Dwyer, J.H. & Goran, M.I. Longitudinal changes in body fat in African American and Caucasian children: influence of fasting insulin and insulin sensitivity. J. Clin. Endocrinol. Metab. 86, 3182–3187 (2001).
Hercberg, S. et al. A primary prevention trial using nutritional doses of antioxidant vitamins and minerals in cardiovascular diseases and cancers in a general population: the SU.VI.MAX study–design, methods, and participant characteristics. SUpplementation en VItamines et Mineraux AntioXydants. Control. Clin. Trials 19, 336–351 (1998).
Oberkofler, H. et al. Complex haplotypes of the PGC-1alpha gene are associated with carbohydrate metabolism and type 2 diabetes. Diabetes 53, 1385–1393 (2004).
Lafay, L. et al. Determinants and nature of dietary underreporting in a free-living population: the Fleurbaix Laventie Ville Sante (FLVS) Study. Int. J. Obes. Relat. Metab. Disord. 21, 567–573 (1997).
Seltzer, H.S., Allen, E.W., Herron, A.L., Jr. & Brennan, M.T. Insulin secretion in response to glycemic stimulus: relation of delayed initial release to carbohydrate intolerance in mild diabetes mellitus. J. Clin. Invest. 46, 323–335 (1967).
Tregouet, D.A., Escolano, S., Tiret, L., Mallet, A. & Golmard, J.L. A new algorithm for haplotype-based association analysis: the Stochastic-EM algorithm. Ann. Hum. Genet. 68, 165–177 (2004).
Acknowledgements
We thank the affected individuals and their families who participated in this study; T. Woodage and Applied Biosystems for technical support; S. Gaget, S. Lobbens, E. Durand, S. Lahmidi, S. Poulain, M. Deweirder, F. Allegaert, V. Delannoy-Vasseur and C. Letierce for technical assistance; and M. Tauber for giving access to the Toulouse obese children DNA Bank. This work was supported in part by Conseil Regional Nord-Pas de Calais/FEDER ARCir and “Association Française des Diabétiques” funding and by AFERO/Roche and ALFEDIAM/Roche Diagnostics research prizes. I.D.G. and B.A.M. were supported by a grant from the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Correlation of the ENPP1 protein serum level with Z score of BMI in 279 children. (PDF 217 kb)
Supplementary Fig. 2
mRNA expression of PC-1/ENPP1 in human tissues. (PDF 217 kb)
Supplementary Table 1
Phenotypic characteristics of “6q-evidence” families. (PDF 62 kb)
Supplementary Table 2
Polymorphisms in 5′ and 3′ proximal regions, exons and boundaries of ENPP1 gene. (PDF 47 kb)
Supplementary Table 3
Association (p<0.05) of genotypes and alleles of ENPP1 gene SNPs with obesity. (PDF 68 kb)
Supplementary Table 4
Description of the French and Austrian samples used in the study. (PDF 48 kb)
Rights and permissions
About this article
Cite this article
Meyre, D., Bouatia-Naji, N., Tounian, A. et al. Variants of ENPP1 are associated with childhood and adult obesity and increase the risk of glucose intolerance and type 2 diabetes. Nat Genet 37, 863–867 (2005). https://doi.org/10.1038/ng1604
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1604
This article is cited by
-
Genetics and Epigenetics in Obesity: What Do We Know so Far?
Current Obesity Reports (2023)
-
A new perspective on the function of Tissue Non-Specific Alkaline Phosphatase: from bone mineralization to intra-cellular lipid accumulation
Molecular and Cellular Biochemistry (2022)
-
A review on interplay between obesity, lipoprotein profile and nutrigenetics with selected candidate marker genes of type 2 diabetes mellitus
Molecular Biology Reports (2022)
-
The genetic susceptibility profile of type 2 diabetes and reflection of its possible role related to reproductive dysfunctions in the southern Indian population of Hyderabad
BMC Medical Genomics (2021)
-
Targets and regulation of microRNA-652-3p in homoeostasis and disease
Journal of Molecular Medicine (2021)