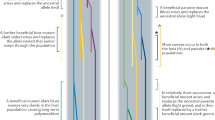
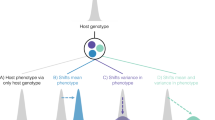
Abstract
Co-evolution between host and pathogen is, in principle, a powerful determinant of the biology and genetics of infection and disease. Yet co-evolution has proven difficult to demonstrate rigorously in practice, and co-evolutionary thinking is only just beginning to inform medical or veterinary research in any meaningful way, even though it can have a major influence on how genetic variation in biomedically important traits is interpreted. Improving our understanding of the biomedical significance of co-evolution will require changing the way in which we look for it, complementing the phenomenological approach traditionally favored by evolutionary biologists with the exploitation of the extensive data becoming available on the molecular biology and molecular genetics of host–pathogen interactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stearns, S.C. Evolution in Health & Disease (Oxford University Press, Oxford, 1999).
Rainey, P.B., Buckling, A., Kassen, R. & Travisano, M. The emergence and maintenance of diversity: insights from bacterial populations. Trends Microbiol. 15, 243–247 (2000).
Thompson, J.N. & Burdon, J.J. Gene-for-gene coevolution between plants and parasites. Nature 360, 121–125 (1992).
Thompson, J.N. The Coevolutionary Process (University of Chicago Press, Chicago, 1994).
Flor, H.H. The complementary genic systems in flax and flax rust. Adv. Genet. 8, 29–54 (1956).
Chao, L., Levin, B.R. & Stewart, F.M. A complex community in a simple habitat: an experimental study with bacteria and phage. Ecology 58, 369–378 (1977).
Lenski, R.E. & Levin, B.R. Constraints on the coevolution of bacteria and virulent phage: a model, some experiments, and predictions for natural communities. Am. Nat. 125, 585–602 (1985).
Kniskern, J. & Rausher, M.D. Two modes of host–enemy evolution. Popul. Ecol. 43, 3–14 (2001).
Little, T.J. The evolutionary significance of parasitism: do parasite-driven dynamics occur ex silico? J. Evol. Biol. 15, 1–9 (2002).
Satta, Y., Ohuigin, C., Takahata, N. & Klein, J. Intensity of natural selection at the major histocompatibility complex loci. Proc. Natl Acad. Sci. USA 91, 7184–7188 (1994).
Ebert, D. Virulence and local adaptation of a new horizontally transmitted parasite. Science 265, 1084–1086 (1994).
Frank, S.A. Models of parasite virulence. Q. Rev. Biol. 71, 37–78 (1996).
Levin, B.R. The evolution and maintenance of virulence in microparasites. Emerg. Infect. Dis. 2, 93–102 (1996).
Mode, C.J. A mathematical model for the coevolution of obligate parasites and their hosts. Evolution 12, 138–165 (1958).
Hamilton, W.D., Axelrod, R. & Tanese, R. Sexual reproduction as an adaptation to resist parasites (a review). Proc. Natl Acad. Sci. USA 87, 3566–3573 (1990).
Morand, S., Manning, S.D. & Woolhouse, M.E.J. Parasite–host coevolution and geographic patterns of parasite infectivity and host susceptibility. Proc. R. Soc. Lond. B Biol. Sci. 263, 119–128 (1996).
Lively, C.M. & Apanius, V. Genetic diversity in host-parasite interactions. in Ecology of Infectious Diseases in Natural Populations (eds Grenfell, B.T. & Dobson, A.P.) 421–449 (Cambridge University Press, Cambridge, 1995).
Anderson, R.M. & May, R.M. Infectious Diseases of Humans: Dynamics and Control (Oxford University Press, Oxford, 1991).
Sasaki, A. & Godfray, H.C.J. A model for the coevolution of resistance and virulence in coupled host–parasitoid interactions. Proc. R. Soc. Lond. B Biol. Sci. 266, 455–463 (1999).
Gandon, S. & Michalakis, Y. Evolution of parasite virulence against qualitative or quantitative resistance. Proc. R. Soc. Lond. B Biol. Sci. 267, 985–990 (2000).
Sasaki, A. Host–parasite coevolution in a multilocus gene-for-gene system. Proc. R. Soc. Lond. B Biol. Sci. 267, 2183–2188 (2000).
Gandon, S. Local adaptation and the geometry of host–parasite coevolution. Ecol. Lett. 5, 246–256 (2002).
Kreitman, M. Methods to detect selection in populations with applications to humans. Ann. Rev. Genomics Hum. Genet. 1, 539–559 (2000).
Haldane, J.B.S. Disease and evolution. Ric. Sci. 19, 68–76 (1949).
Fenner, F. & Fantini, B. Biological Control of Vertebrate Pests (CABI Publishing, Wallingford, 1999).
Lively, C.M. & Dybdahl, M.F. Parasite adaptation to locally common host genotypes. Nature 405, 679–681 (2000).
Dybdahl, M.F. & Lively, C.M. Host–parasite coevolution: evidence for rare advantage and time-lagged selection in a natural population. Evolution 52, 1057–1066 (1998).
de la Torre, J.C. et al. Coevolution of cells and viruses in a persistent infection of foot-and-mouth disease virus in cell culture. J. Virol. 62, 2050–2058 (1988).
Braun-Breton, C. & Hofnung, M. In vivo and in vitro functional alterations of the bacteriophage lambda receptor in lamB missense mutants of Escherichia coli K-12. J. Bacteriol. 148, 845–852 (1981).
Hofnung, M., Jezierska, A. & Braun-Breton, C. LamB mutations in E. coli K12: growth of lambda host range mutants and effect of nonsense suppressors. Mol. Gen. Genet. 145, 207–213 (1976).
Woolhouse, M.E.J. et al. A centuries-long epidemic of scrapie in British sheep? Trends Microbiol. 6, 67–70 (2001).
Levin, B.R., Bull, J.J. & Stewart, F.M. The intrinsic rate of increase of HIV/AIDS: epidemiological and evolutionary implications. Math. Biosci. 132, 69–96 (1996).
Lively, C.M. Migration, virulence and the geographic mosaic of adaptation by parasites. Am. Nat. 153, S34–S47 (1999).
Carius, H.J., Little, T.J. & Ebert, D. Genetic variation in a host–parasite association: potential for coevolution and frequency-dependent selection. Evolution 55, 1136–1145 (2001).
Thompson, J.N. Specific hypotheses on the geographical mosaic of coevolution. Am. Nat. 153, S1–S14 (1999).
Nuismer, S.L., Thompson, J.N. & Gomulkiewicz, R. Gene flow and geographically structured coevolution. Proc. R. Soc. Lond. B Biol. Sci. 266, 605–609 (1998).
Kraaijeveld, A.R. & Godfray, H.C.J. Geographic patterns in the evolution of resistance and virulence in Drosophila and its parasitoids. Am. Nat. 153, S61–S74 (1999).
Woolhouse, M.E.J. & Webster, J.P. In search of the Red Queen. Parasitol. Today, 16, 506–508 (2000).
Covacci, A., Telford, J.L., Del Giudice, G., Parsonnet, J. & Rappuoli, R. Helicobacter pylori virulence and genetic geography. Science 284, 1328–1333 (1999).
Ong, C.K. et al. Evolution of human papillomavirus type 18: an ancient phylogenetic root in Africa and intratype diversity reflect coevolution with human ethnic groups. J. Virol. 67, 6424–6423 (1993).
Agostini, H.T., Yanagihara, R., Davis, V., Ryschkewitsch, C.F. & Stoner, G.L. Asian genotypes of JC virus in native Americans and in a Pacific island population: markers of viral evolution and human migration. Proc. Natl Acad. Sci. USA 94, 14542–14546 (1997).
Harvey, P.H. & Pagel, M.D. The Comparative Method in Evolutionary Biology (Oxford University Press, Oxford, 1991).
Huelsenbeck, J.P., Rannala, B. & Masly, J.P. Accommodating phylogenetic uncertainty in evolutionary studies. Science 288, 2349–2350 (2000).
Webster, J.P. & Davies, C.M. Coevolution and compatibility in the snail-schistosome system. Parasitology 123, S42–S56 (2001).
Wakelin, D. & Blackwell, J.M. (eds). Genetics of Resistance to Bacterial and Parasitic Infection (Taylor & Francis, London, 1988).
Hill, A.V.S., Jepson, A., Plebanski, M. & Gilbert, S.C. Genetic analysis of host–parasite coevolution in human malaria. Philos. Trans. R. Soc. Lond. B Biol. Sci. 352, 1317–1325 (1997).
Agrawal, A.A. Phenotypic plasticity in the interactions and evolution of species. Science 294, 321–326 (2001).
Yan, G., Severson, D.W. & Christensen, B.M. Costs and benefits of mosquito refractoriness to malaria parasites: implications for genetic variability of mosquitoes and genetic control of malaria. Evolution 51, 441–450 (1997).
Webster, J.P. & Woolhouse, M.E.J. Cost of resistance: relationship between reduced fertility and increased resistance in a Schistosoma host-parasite system. Proc. R. Soc. Lond. B Biol. Sci. 266, 391–396 (1999).
Kraaijeveld, A.R. & Godfray, H.C.J. Trade-off between parasitoid resistance and larval competitor ability in Drosophila melanogaster. Nature 389, 278–280 (1997).
Searle, S. & Blackwell, J.M. Evidence for a functional-repeat polymorphism in the promoter of human NRAMP1 gene that correlates with autoimmune versus infectious disease susceptibility. J. Med. Gen. 36, 295–299 (1999).
Falconer, D.S. & Mackay, T.F.C. An Introduction to Quantitative Genetics, 4th Edition (Longman, London, 1996).
Agrawal, A. & Lively, C.M. Infection genetics: gene-for-gene versus matching allele models, and all points in between. Evol. Ecol. Res. 4, 1–12 (2002).
Taylor, L.H., Latham, S.M. & Woolhouse, M.E.J. Risk factors for human disease emergence. Philos. Trans. R. Soc. Lond. B Biol. Sci. 356, 983–990 (2001).
Webster, J.P. & Macdonald, D.W. Parasites of wild brown-rats (Rattus norvegicus) on UK farms. Parasitology 111, 247–255 (1995).
Read, A.F. & Taylor, L.R. The ecology of genetically diverse infections. Science 292, 1099–1102 (2001).
Ochman, H. & Moran, N.A. Genes lost and genes found: evolution of bacterial pathogenesis and symbiosis. Science 292, 1096–1098 (2001).
Fraser, C.M., Eisen, J.A. & Salzberg, S.L. Microbial gene sequencing. Nature 406, 799–803 (2000).
Lord, C.C. et al. Aggregation and distribution of strains in microparasites. Philos. Trans. R. Soc. Lond. B Biol. Sci. 354, 799–807 (1999).
Basanez, M.G. et al. Density-dependent processes in the transmission of human onchocerciasis—relationship between the numbers of microfilariae ingested and successful larval development in the simuliid vector. Parasitology 110, 409–427 (1995).
Woolhouse, M.E.J., Taylor, L.H. & Haydon, D.T. Population biology of multi-host pathogens. Science 292, 1109–1112 (2001).
Bohannan, B.J.M., Travisano, M. & Lenski, R.E. Epistatic interactions can lower the cost of resistance to multiple consumers. Evolution 53, 292–295 (1999).
Ebert, D. Experimental evolution of parasites. Science 282, 1432–1435 (1998).
Weaver, S.C., Brault, A.C., Kang, W.L. & Holland J.J. Genetic and fitness changes accompanying adaptation of an arbovirus to vertebrate and invertebrate cells. J. Virol. 73, 4316–4326 (1999).
Davies, N.B. & de L. Brooke, M. An experimental study of co-evolution between the cuckoo, Cuculus canorus, and its hosts. II Host egg markings, chick discrimination and general discussion. J. Anim. Ecol. 58, 225–236 (1989).
Schneider-Schaulies, J. Cellular receptors for viruses: links to tropism and pathogenesis. J. Gen. Virol. 81, 1413–1429 (2000).
Woolhouse, M.E.J. Population biology of emerging and re-emerging pathogens. Trends Microbiol. 10, S3–S7 (2002).
Baranowski, E., Ruiz-Jarabo, C.M. & Domingo, E. Evolution of cell recognition by viruses. Science 292, 1102–1105 (2001).
Stebbins, C.E. & Galan, J.E. Structural mimicry in bacterial virulence. Nature 412, 701–705 (2001).
Gilbert, S.C. et al. Association of malaria parasite population structure, HLA, and immunological antagonism. Science 279, 1173–1177 (1998).
Slatkin, M. & Rannala, B. Estimating allele age. Ann. Rev. Genomics Hum. Genet. 1, 225–249 (2000).
Altschuler, E.L. Plague as HIV vaccine adjuvant. Med. Hypotheses 54, 1003–1004 (2000).
Schliekelman, P., Garner, C. & Slatkin, M. Natural selection and resistance to HIV. Nature 411, 545–546 (2001).
Berger, E.A., Murphy, P.M. & Farber, J.M. Chemokine receptors as HIV-1 co-receptors: roles in viral entry, tropism and disease. Ann. Rev. Immunol. 17, 657–700 (1999).
Moore, C.B. et al. Evidence of HIV-1 adaptation to HLA-restricted immune responses at a population level. Science 296, 1439–1443 (2002).
Day, C.L. et al. Relative dominance of epitope-specific cytotoxic T-lymphocyte responses in human immunodeficiency virus type 1-infected persons with shared HLA alleles. J. Virol. 75, 6279–6291 (2001).
Goulder, P.J.R. et al. Patterns of immunodominance in HIV-1-specific cytotoxic T lymphocyte responses in two human histocompatibility leukocyte antigens (HLA)-identical siblings with HLA-A*0201 are influenced by epitope mutation. J. Exp. Med. 185, 1423–1433 (1997).
Altfeld, M. et al. Cellular immune responses and viral diversity in individuals treated during acute and early HIV-1 infection. J. Exp. Med. 193, 169–180 (2001).
Bergelson, J., Kreitman, M., Stahl, E.A. & Tian, D.C. Evolutionary dynamics of plant R-genes. Science 292, 2281–2285 (2001).
Klein, J., Satta, Y., Ohuigin, C. & Takahata, N. The molecular descent of the major histocompatibility complex. Ann. Rev. Immunol. 11, 269–295 (1993).
Vogel, T.U. et al. Major histocompatibility complex class I genes in primates: coevolution with pathogens. Immunol. Rev. 167, 327–337 (1999).
Apanius, V., Penn, D., Slev, P.R., Ruff, L.R. & Potts, W.K. The nature of selection on the major histocompatibility complex. Crit. Rev. Immunol. 17, 179–224 (1997).
Yang, Z. & Bielawski, J.P. Statistical methods for detecting molecular adaptation. Trends Ecol. Evol. 15, 496–503 (2000).
Vartanian, J.P., Henry, M. & Wain-Hobson, S. Simulating pseudogene evolution in vitro: determining the true number of mutations in a lineage Proc. Natl Acad. Sci. USA 98, 13172–13176 (2001).
Domingo, E., Webster, R.G. & Holland, J. Origin and Evolution of Viruses (Academic Press, San Diego, 1999).
Bush, R.M. et al. Predicting the evolution of human influenza A. Science 286, 1921–1925 (1999).
International SNP Map Working Group. A map of human genome sequence variation containing 1.42 million single-nucleotide polymorphisms. Nature 409, 928–933 (2001).
Domingo, E., Biebricher, C., Eigen, M. & Holland, J.J. Quasispecies and RNA Virus Evolution: Principles and Consequences (Landes Bioscience, Landes, 2001).
Charlesworth, D., Charlesworth, B. & McVean, G. Genome sequences and molecular evolution, a two-way interaction. Trends Ecol. Evol. 16, 235–242 (2001).
Gandon, S., Mackinnon, M.J., Nee, S. & Read, A.F. Imperfect vaccines and the evolution of pathogen virulence. Nature 414, 751–756 (2001).
Woolhouse, M.E.J. & Dye, C. (eds) Population biology of emerging and re-emerging pathogens. Philos. Trans. R. Soc. Lond. B Biol. Sci. 356, 979–1106 (2001).
Crow, J.F. & Kimura, M. An Introduction to Population Genetics Theory (Harper and Row, New York, 1970).
Webster, J.P. & Woolhouse, M.E.J. Heritability and strain specificity in compatibility between snail intermediate hosts and their parasitic schistosomes. Evolution 52, 1627–1634 (1998).
Oppliger, A., Vernet, R. & Baez, M. Parasite local maladaptation in the Canarian lizard Gallotia galloti (Reptilia: Lacertidae) parasitized by haemogregarian blood parasite. J. Evol. Biol. 12, 951–955 (1999).
Cao, W. et al. Identification of α-dystroglycan as a receptor for lymphocytic choriomeningitis virus and Lassa fever virus. Science 282, 2079–1081 (1998).
Overbaugh, J. & Bangham, C.R.M. Selection forces and constraints on retroviral sequence variation. Science 292, 1106–1109 (2001).
Sorci, G., Moller, A.P. & Boulinier, T. Genetics of host–parasite interactions. Trends Ecol. Evol. 12, 196–200 (1997).
Helmby, H., Kullberg, M. & Troye-Blomberg, M. Altered immune responses in mice with concomitant Schistosoma mansoni and Plasmodium chabaudi infections. Infect. Immun. 66, 5167–5174 (1998).
Allen, J.E. & Maizels, R.M. Th1-Th2: reliable paradigm or dangerous dogma? Immunol. Today 18, 387–392 (1997).
Sullivan, J.T. et al. Schistosoma mansoni, NIH-SM-PR-2 strain, in non-susceptible Biomphalaria glabrata—protection by Echinostoma paraensei. Int. J. Parasitol. 11, 481–484 (1981).
Stosor, V. & Wolinsky, S. GB virus C and mortality from HIV infection. N. Engl. J. Med. 345, 761–762 (2001).
Donnenberg, M.S. Pathogenic strategies of enteric bacteria. Nature 406, 768–774 (2000).
Pellizzari, A., Pang, H. & Lingwood, C.A. Binding of verocytotoxin 1 to its receptor is influenced by differences in receptor fatty acid content. Biochemistry 31, 1363–1370 (1992).
McFadden, G. Getting to know you—viruses meet CD40 ligand. Nature Med. 1, 408–409 (1995).
Faure, S. et al. Rapid progression to AIDS in HIV+ individuals with a structural variant of the chemokine receptor CX(3)CR1. Science 287, 2274–2277 (2000).
Acknowledgements
We thank A. Read, L. Taylor, D. Haydon, M. Charleston, R. Phillips, C. Davies, P. Goulder, C. Lively and E. Baranowski for comments and discussion and gratefully acknowledge support from the Wellcome Trust, the Leverhulme Trust, the Royal Society, Ministerio de Ciencia y Tecnologia and Fundacion R Areces.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Woolhouse, M., Webster, J., Domingo, E. et al. Biological and biomedical implications of the co-evolution of pathogens and their hosts. Nat Genet 32, 569–577 (2002). https://doi.org/10.1038/ng1202-569
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1202-569
This article is cited by
-
Metatranscriptome analysis of symptomatic bitter apple plants revealed mixed viral infections with a putative novel polerovirus
BMC Genomics (2024)
-
Monogeneans in intergeneric hybrids of leuciscid fish: Is parasite infection driven by hybrid heterosis, genetic incompatibilities, or host-parasite coevolutionary interactions?
Frontiers in Zoology (2023)
-
Balancing selection on the complement system of a wild rodent
BMC Ecology and Evolution (2023)
-
Coevolution between marine Aeromonas and phages reveals temporal trade-off patterns of phage resistance and host population fitness
The ISME Journal (2023)
-
Coevolutionary Dynamics of Host Immune and Parasite Virulence Based on an Age-Structured Epidemic Model
Bulletin of Mathematical Biology (2023)