Abstract
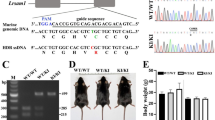
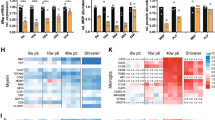
Mutations in the human gene for the myelin recognition molecule protein zero (P0) give rise to severe and progressive forms of dominantly inherited peripheral neuropathies. We have previously reported that mice homozygous for a null mutation in P0 have severely hypomyelinated nerves ten weeks after birth. Here we show hypomyelination already exists at day four with subsequent demyelination and impaired nerve conduction. Furthermore, heterozygous mutants show normal myelination, but develop progressive demyelination after four months of age. Thus, the pathology of homo– and heterozygous P0 mutants resembles that of the severely affected Déjérine–Sottas and the more mildly affected Charcot–Marie–Tooth type 1B patients, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Giese, K.P., Martini, R., Lemke, G., Soriano, P. & Schachner, M. Mouse P0 gene disruption leads to hypomyelination, abnormal expression of recognition molecules, and degeneration of myelin and axons. Cell 71, 565–676 (1992).
Hayasaka, K. et al. Charcot-Marie-Tooth neuropathy type 1B is associated with mutations of the myelin P0 gene. Nature Genet 5, 31–34 (1993).
Hayasaka, K. et al. De novo mutation of the myelin P0 gene in Dejerine-Sottas disease (hereditary motor and sensory neuropathy type III). Nature Genet. 5, 266–268 (1993).
Hayasaka, K., Ohnishi, A., Takada, G., Fukushima, Y. & Murai, Y. Mutation of the myelin P0 gene in Charcot-Marie-Tooth neuropathy type 1. Biochem. biophys. Res. Comm. 194, 1317–1322 (1993).
Hayasaka, K., Takada, G. & lonasescu, V.V. Mutation of the myelin P0 gene in Charcot-Marie-Tooth neuropathy type 1B. Hum. molec. Genet. 2, 1369–1372 (1993).
Himoro, M. et al. New mutation of the myelin P0 gene in a pedigree of Charcot-Marie-Tooth neuropathy type 1 Biochem. molec. Biol. Int. 31, 169–173 (1993).
Kulkens, T. et al. Deletion of the serine 34 codon from the major peripheral myelin protein P0 gene in Charcot-Marie-Tooth disease type 1B. Nature Genet. 5, 35–39 (1993).
Su, Y. et al. Myelin protein zero gene mutated in Charcot-Marie-Tooth type 1B patients. Proc. natl. Acad. Sci. U.S.A. 90, 10856–10860 (1993).
Nelis, E. et al. Linkage and mutation analysis in an extended family with Charcot-Marie-Tooth disease 1B. J. mod. Genet. 31, 811–815 (1994).
Rautenstrauß, B., Nelis, E., Grehl, H., Pfeiffer, R.A. & Van Broeckhoven, C. Identification of a de novo insertions mutation in P0 in a patient with a Déjérine-Sottas syndrome (DSS) phenotype. Hum. molec. Genet. 3, 1701–1702 (1994).
Martini, R., Mohajeri, M.H., Kasper, S., Giese, K.P. & Schachner, M. Mice doubly deficient in the genes for P0 and myelin basic protein show that both proteins contribute to the formation of the major dense line in peripheral nerve myelin. J. Neurosci. 15, 4488–4495.
Dyck, P.J., Chance, P., Lebo, R. & Carney, J.I. Hereditary motor and sensory neuropathies. in Peripheral Neuropathy 3rd edn. (eds Dyck, R.J. et al.) 1094–1136 (Saunders, Philadelphia, 1993).
Landon, D.N. & Hall, S. The myelinated nerve fibre, in The Peripheral Nerve (ed. Landon, D.N.) 1–90 (Chapman & Hall, London, 1976).
Reynolds, R.J., Little, G.J., Lin, M. & Heath, J.W. Imaging myelinated nerve fibres by confocal fluorescence microscopy: individual fibres in whole nerve trunks traced through multiple consecutive internodes. J. Neurocytol. 23, 555–564 (1994).
Martini, R., Schachner, M. & Faissner, A. Enhanced expression of the extraceltuiar matrix moiecule J1/tenascin in the regenerating adult mouse sciatic nerve. J. Neurocytol. 19, 601–616 (1990).
Fruttiger, M., Schachner, M. & Martini, R. Tenascin-C expression during Wallerian degeneration in C57BL/Wlds mice: P0ssible implications for axonal regeneration. J. Neurocytol. 24, 1–14 (1995).
Fruttiger, M., Montag, D., Schachner, M. & Martini, R. Crucial role for the myelin-associated glycoprotein for the maintenance of axon-myelin integrity. Eur. J. Neurosci. 7, 511–615 (1995).
Tachi, N., Ishikawa, Y. & Minami, R. Two cases of congenital hyP0myelination neuropathy. Brain Devi. 6, 560–565 (1984).
Ouvrier, R.A., McLeod, J.G. & Conchin, T.E. The hypertrophic forms of hereditary motor and sensory neuropathy. Brain 110, 121–148 (1987).
Hoogendijk, J.E. et al. Ultrastrucutral features of defective myelination in hereditary motor and sensory neuropathy type 1B (abstract). J. Neurol. 242 [Suppl. 2], 149 (1995).
Uchida, Y., Tomonaga, M. & Nomura, K. Age-related changes of myelin proteins in the rat peripheral nervous system. J. Neurochem. 46, 1376–1381 (1986).
Mitchell, L.S. et al. Expression of myelin protein gene transcripts by Schwann cells of regenerating nerve. J. neurosci. Res. 27, 125–135 (1990).
Shine, H.D., Readhead, C., Popko, B., Hood, L. & Sidman, R.L. Morphometric analysis of normal mutant and transgenic CNS: correlation of myelin basic protein expression to myelinogenesis. J. Neurochem. 58, 342–349 (1992).
Nave, K.-A. Neurological mouse mutants and the genes of myelin. J. neurosci. Res. 38, 607–612 (1994).
Suter, U., Welcher, A.A. & Snipes, G.J. Progress in the molecular understanding of hereditary peripheral neuropathies reveals new insights into the biology of the peripheral nervous system. Trends Neurosci. 16, 50–56 (1993).
Adlkofer, K. et al. Hypermyelination and demyelinating peripheral neuropathy in Pmp22-deficient mice. Nature Genet. 11, 274–280 (1995).
Gow, A., Friedrich, V.L. & Lazzarini, R.A. Many naturally occurring mutations of myelin proteolipid protein impair its intracellular transP0rt. J. neurosci. Res. 37, 574–583 (1994).
Kirschner, D.A. & Saavedra, R.A. Mutations in demyelinating peripheral neuropathies supP0rt molecular model of myelin P0–glycoprotein extracellular domain. J. neurosci. Res. 39, 63–69 (1994).
Husmann, K., Faissner, A. & Schachner, M. Tenascin promotes cerebellar granule cell migration and neurite outgrowth by different domains in the fibronectin type III repeats. J. Cell Biol. 116, 1475–1486 (1992).
Montag, D. et al. Mice deficient for the myelin-associated glycoprotein show subtle abnormalities in myelin. Neuron 13, 229–246 (1994).
Hartung, H.R., SchSfer, B., Heininger, K., Stoll, G. & Toyka, K.V. The role of macrophages and eicosanoids in the pathogenesis of experimental allergic neuritis. Serial clinical, electrophysiological, biochemical, and morphological observations. Brain 111, 1039–1059 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martini, R., Zielasek, J., Toyka, K. et al. Protein zero (P0)–deficient mice show myelin degeneration in peripheral nerves characteristic of inherited human neuropathies. Nat Genet 11, 281–286 (1995). https://doi.org/10.1038/ng1195-281
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1195-281
This article is cited by
-
NRG1 type I dependent autoparacrine stimulation of Schwann cells in onion bulbs of peripheral neuropathies
Nature Communications (2019)
-
Delaying histone deacetylase response to injury accelerates conversion into repair Schwann cells and nerve regeneration
Nature Communications (2017)
-
Endogenous antibodies contribute to macrophage-mediated demyelination in a mouse model for CMT1B
Journal of Neuroinflammation (2015)
-
GJB1-associated X-linked Charcot–Marie–Tooth disease, a disorder affecting the central and peripheral nervous systems
Cell and Tissue Research (2015)
-
Functional Recovery of Regenerating Motor Axons is Delayed in Mice Heterozygously Deficient for the Myelin Protein P0 Gene
Neurochemical Research (2013)