Abstract
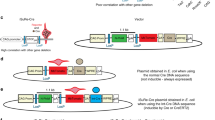
Chromosome loss in early human embryos is thought to cause a large proportion of spontaneous abortions1; when it occurs in specific cell lineages in older embryos or adults, it can result in neoplasia2. Although early embryonic chromosome loss can be modelled by breeding mice carrying robertsonian translocation chromosomes3, there is currently no method for producing mice with tissue-specific monosomies. Here we demonstrate that DNA recombination mediated by the site-specific recombinase Cre4 causes loss of a chromosome carrying loxP sites (Cre recognition sites) in an inverted orientation. Thus, when male mice carrying a Y-linked transgene containing inverted loxP sites are mated with females carrying a cre gene that is ubiquitously expressed in the early embryo, almost all their XY progeny lose the Y chromosome early in embryogenesis and develop as XO females. Because inverted loxP sites can be targetted to any mouse chromosome and mice can be produced that express cre in specific cell lineages, these data suggest a method for engineering tissue-specific loss of particular chromosomes to provide mouse models for human diseases caused by or associated with specific monosomies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Epstein, C.J., Consequences of Chromosome Imbalance (Cambridge University Press, Cambridge, UK, (1986).
Heim, S. & Mitelman, F., Cancer Cytogenetics (Wiley-Liss, New York, 1995).
Epstein, C.J. Mouse monosomies and trisomies as experimental systems for studying mammalian aneuploidy. Trends Genet. 1, 129–134 (1985).
Kilby, N.J., Snaith, M.R. & Murray, J.A. Site-specific recombinases: tools for genome engineering. Trends Genet. 9, 413–421 (1993).
Golic, K.G. Local transposition of P elements in Drosophila melanogaster and recombination between duplicated elements using a site-specific recombinase. Genetics 137, 551–563 (1994).
Falco, S.C., Li, Y., Broach, J.R. & Botstein, D. Genetic properties of chromosomally integrated 2 μ plasmid DNA in yeast. Cell 29, 573–584 (1982).
Lewandoski, M., Meyers, E.N. & Martin, G.R. Analysis of Fgf8 gene function in vertebrate development. Cold Spring Harbor Symp. Quant. Biol. (in the press).
Cattanach, B.M. XO mice. Genet Res. 3, 487–490 (1962).
Nagamine, C.M., Chan, K., Kozak, C.A. & Lau, Y.-F. Chromosome mapping and expression of a putative testis-determining gene in mouse. Science 243, 80–83 (1989).
Nagamine, C.M., Michot, J.-L., Roberts, C., Guénet, J.-L. & Bishop, C.E. Linkage of the murine steroid sulfatase locus, Sts, to sex reversed, Sxr. a genetic and molecular analysis. Nucleic Acids Res. 15, 9227–9238 (1987).
Andreason, G.L. & Evans, G.A. Introduction and expression of DNA molecules in eukaryotic cells by electroporation. Biotechniques 6, 650–660 (1988).
Luna-Fineman, S., Shannon, K.M. & Lange, B.J. Childhood monosomy 7: epidemiology, biology, and mechanistic implications. Blood 85, 1985–1999 (1995).
Tsien, J.Z. et al. Subregion- and cell type–restricted gene knockout in mouse brain. Cell 87, 1317–1326 (1996).
Gu, H., Marth, J.D., Orban, P.C., Mossmann, H. & Rajewsky, K. Deletion of a DNA polymerase β gene segment in T cells using cell type–specific gene targeting. Science 265, 103–106 (1994).
Ramirez-Solis, R., Liu, P. & Bradley, A. Chromosome engineering in mice. Nature 378, 720–724 (1995).
Bouffler, S.D., Morgan, W.F., Pandita, T.K. & Slijepcevic, P. The involvement of telomeric sequences in chromosomal aberrations. Mutat. Res. 366, 129–135 (1996).
Sandell, L.L. & Zakian, V.A. Loss of a yeast telomere: arrest, recovery, and chromosome loss. Cell 75, 729–739 (1993).
Lewandoski, M., Wassarman, K.M. & Martin, G.R. Zp3-cre, a transgenic mouse line for the activation or inactivation of loxP-flanked target genes specifically in the female germ line. Curr. Biol. 7, 148–151 (1997).
Hogan, B., Beddington, R., Costantini, F. & Lacy, E. Manipulating the Mouse Embryo, 2nd ed. 226–250 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, (1994).
Nagy, A. & Rossant, J. in Gene Targeting: A Practical Approach (ed. Joyner, A.L.) 147–179 (IRL Press, Oxford, UK, (1993).
Tybulewicz, V.L., Crawford, C.E., Jackson, P.K., Bronson, R.T. & Mulligan, R.C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65, 1153–1163 (1991).
Laird, P.W. et al. Simplified mammalian DNA isolation procedure. Nucleic Acids Res. 19, 4293 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lewandoski, M., Martin, G. Cre–mediated chromosome loss in mice. Nat Genet 17, 223–225 (1997). https://doi.org/10.1038/ng1097-223
Issue Date:
DOI: https://doi.org/10.1038/ng1097-223