Abstract
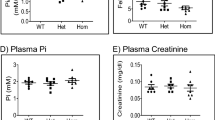
Analysis of patients with inherited hypokalaemic alkalosis resulting from salt–wasting has proved fertile ground for identification of essential elements of renal salt homeostasis and blood–pressure regulation. We now demonstrate linkage of this phenotype to a segment of chromosome 1 containing the gene encoding a renal chloride channel, CLCNKB. Examination of this gene reveals loss–of–function mutations that impair renal chloride reabsorption in the thick ascending limb of Henle's loop. Mutations in seventeen kindreds have been identified, and they include large deletions and nonsense and missense mutations. Some of the deletions are shown to have arisen by unequal crossing over between CLCNKB and the nearby related gene, CLCNKA. Patients who harbour CLCNKB mutations are characterized by hypokalaemic alkalosis with salt–wasting, low blood pressure, normal magnesium and hyper– or normocalciuria; they define a distinct subset of patients with Bartter's syndrome in whom nephrocalcinosis is absent. These findings demonstrate the critical role of CLCNKB in renal salt reabsorption and blood–pressure homeostasis, and demonstrate the potential role of specific CLCNKB antagonists as diuretic antihypertensive agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lifton, R.P. Molecular genetics of human blood pressure variation. Science 272, 676–680 (1996).
Bettinelli, A. et al. Use of calcium excretion values to distinguish two forms of primary renal tubular hypokalemic alkalosis: Bartter and Gitelman syndromes. J. Pediatr. 120, 38–43 (1992).
Matsumoto, J. et al. Hypercalciuric Bartter syndrome: resolution of nephrocalcinosis with indomethacin. Am. J. Roentgenol. 152, 1251–1253 (1989).
Simon, D.B. et al. Bartter's syndrome, hypokalaemic alkalosis with hypercalciuria, is caused by mutations in the Na-K-2CI cotransporter NKCC2. Nature Genet. 13, 183–188 (1996).
Simon, D.B. et al. Genetic heterogeneity of Bartter's syndrome revealed by mutations in the K+ channel, ROMK. Nature Genet. 14, 152–156 (1996).
Reeves, W.B., Winters, C.J., Zimniak, L. & Andreoli, T.E. Epithelial chloride channels, from kidney to airway cells. Adv. Nephol. 23, 177–190 (1994).
Jorgensen, P.L. Sodium and potassium ion pump in kidney tubules. Physiol. Rev. 60, 864–917 (1980).
Kieferle, S. et al. Two highly homologous members of the CIC chloride channel family in both rat and human kidney. Proc. Natl. Acad. Sci. USA 91, 6943–6947 (1994).
Vandewalle, A. et al. Localization and induction by dehydration of CIC-K chloride channels in the rat kidney. Am. J. Physiol. 272, F678–F688 (1995).
Adachi, S. et al. Two isoforms of a chloride channel predominantly expressed in thick ascending limb of Henle's loop and collecting tubules. J. Biol. Chem. 269, 17677–17683 (1994).
Jentsch, T.J. & Günther, W. Chloride channels: an emerging molecular picture. Bioessays 19, 117–126 (1996).
Orita, M. et al. Detection of polymorphisms of human DNA by gel electrophoresis as single-strand conformation polymorphisms. Proc. Natl. Acad. Sci. USA 86, 2766–2770 (1989).
Lander, E.S. & Botstein, D. Homozygosity mapping: a way to map human recessive traits with the DNA of inbred children. Science 236, 1567–1570 (1987).
Saito-Ohara, F. et al. Assignment of the genes encoding the human chloride channels, CLCNKA and CLCNKB, to 1p36 and of CLCN3 to 4q32–q33 by in situ hybridization. Genomics 36, 372–374 (1996).
Simon, D.B. et al. Gitelman's variant of Bartter's syndrome, inherited hypokalaemic alkalosis, is caused by mutations in the thiazide-sensitive Na–CI cotransporter. Nature Genet. 12, 24–30 (1996).
Greger, R. & Schlatter, E. Properties of the cortical thick ascending limb of Henle's loop of rabbit kidney: a model of secondary active chloride transport. Pflüngers Arch. 396, 325–334 (1983).
Lauer, J., Shen, C.K. & Maniatis, T. The chromosomal arrangement of human alpha-like globin genes: sequence homology and alpha-globin gene deletions. Cell 20, 119–130 (1980).
Nathans, J. et al. Molecular genetics of inherited variation in human color vision. Science 232, 203–210 (1986).
White, P.C., Vitek, A., Dupont, B. & New, M.I. Characterization of frequent deletions causing steroid 21-hydroxylase deficiency. Proc. Natl. Acad. Sci. USA 85, 4436–4440 (1988).
Lifton, R.P. et al. A chimaeric 11β-hydroxylase/aldosterone synthase gene causes glucocorticoid-remediable aldosteronism and human hypertension. Nature 355, 262–265 (1992).
Shimkets, R.A. et al. Liddle's syndrome: heritable human hypertension caused by mutations in the beta subunit of the epithelial sodium channel. Cell 79, 407–414 (1994).
Hansson, J.H. et al. Hypertension caused by a truncated epithelial sodium channel subunit: genetic heterogeneity of Liddle syndrome. Nature Genet. 11, 76–82 (1995).
Chang, S.S. et al. Mutations in subunits of the epithelial sodium channel cause salt wasting with hyperkalaemic acidosis, pseudohypoaldosteronism type 1. Nature Genet. 12, 248–253 (1996).
Lloyd, S.E. et al. A common molecular basis for three inherited kidney stone diseases. Nature 379, 445–449 (1996).
Bell, G., Karam, J. & Rutter, W. Polymorphic DNA region adjacent to the 5′ end of the human insulin gene. Proc. Natl. Acad. Sci. USA 78, 5759–5763 (1981).
Gyapay, G. et al. The 1993–94 Généthon human genetic linkage map. Nature Genet. 7, 246–339 (1994).
Ott, J. Analysis of Human Genetic Linkage (Johns Hopkins University Press, Baltimore, (1985).
Gill, J.R. & Bartter, F.C. Evidence for a prostaglandin-independent defect in chloride reabsorption in the loop of Henle as a proximate cause of Bartter's syndrome. Am. J. Med. 65, 766–772 (1978).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simon, D., Bindra, R., Mansfield, T. et al. Mutations in the chloride channel gene, CLCNKB, cause Bartter's syndrome type III. Nat Genet 17, 171–178 (1997). https://doi.org/10.1038/ng1097-171
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1097-171
This article is cited by
-
The dominant findings of a recessive man: from Mendel’s kid pea to kidney
Pediatric Nephrology (2023)
-
The genetics of kidney stone disease and nephrocalcinosis
Nature Reviews Nephrology (2022)
-
Genetic diagnosis and treatment of hereditary renal tubular disease with hypokalemia and alkalosis
Journal of Nephrology (2022)
-
Clues and new evidences in arterial hypertension: unmasking the role of the chloride anion
Pflügers Archiv - European Journal of Physiology (2022)
-
Pathophysiological aspects of the thick ascending limb and novel genetic defects: HELIX syndrome and transient antenatal Bartter syndrome
Pediatric Nephrology (2022)