Abstract
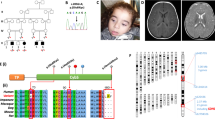
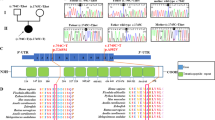
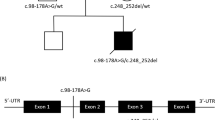
We now report a mutation in the nuclear–encoded flavoprotein (Fp) subunit gene of the succinate dehydrogenase (SDH) in two siblings with complex II deficiency presenting as Leigh syndrome. Both patients were homozygous for an Arg554Trp substitution in the Fp subunit. Their parents (first cousins) were heterozygous for the mutation that occurred in a conserved domain of the protein and was absent from 120 controls. The deleterious effect of the Arg to Trp substitution on the catalytic activity of SDH was observed in a SDH− yeast strain transformed with mutant Fp cDNA. The Fp subunit gene is duplicated in the human genome (3q29; 5p15), with only the gene on chromosome 5 expressed in human–hamster somatic cell hybrids. This is the first report of a nuclear gene mutation causing a mitochondrial respiratory chain deficiency in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wallace, D.C. Diseases of the mitochondrial DNA. A. Rev. Biochem. 61, 1175–1212 (1992).
Munnich, A. et al. Clinical aspects of mitochondrial disorders. J. inher. metab. Dis. 15, 448–455 (1992).
Walker, J.E. The NADH: ubiquinone oxidoreductase (complex I) of respiratory chain. Qu. Rev. Biophys. 25, 253–324 (1992).
Ackrell, B.A.C., Johnson, M.K., Gunsalus, R.P. & Cecchini, G. Structure and function of succinate dehydrogenase and fumarate reductase. In Chemistry and Biochemistry of Flavoproteins (ed. Muller, F.) 3, 229–297 (CRC Press, Inc., Boca Raton, FL, 1990).
Morris, A.A.M., Farnsworth, L., Ackrell, B.A.C., Turnbull, D.M. & Birch-Machin, M.A. The cDNA sequence of the flavoprotein subunit of human heart succinate dehydrogenase. Biochim. Biophys. Acta 1185, 125–128 (1994).
Kita, K., Oya, H., Gennis, R.B., Ackrell, B.A.C. & Kasahara, M. Human complex II (succinate-ubiquinone oxidoreductase): cDNA cloning of iron sulphur (Ip) subunit of liver mitochondria. Biochem. Biophys. Res. Commun. 166, 101–108 (1990).
Shoffner, J.M. & Wallace, D.C. Oxidative phosphorylation diseases and mitochondrial DNA mutations: Diagnosis and treatment. A. Rev. Nutr. 14, 535–568 (1994).
Bourgeois, M. et al. Deficiency in complex II of the respiratory chain presenting as a leukodystrophy in two sisters with Leigh syndrome. Brain Dev. 14, 404–408 (1992).
Robinson, B.H. Lacticacidemia. Biochim. Biophys. Acta 1182, 231–244 (1993).
Hall, R.E., Henriksson, K.G., Lewis, S.F., Haller, R.G. & Kennaway, N.G. Mitochondrial myopathy with succinate dehydrogenase and aconitase deficiency. Abnormalities of several iron-sulfur-proteins. J. clin. Invest. 92, 2660–2666 (1993).
Kotlyar, A.B. & Vinogradov, A.D. Interaction of the membrane-bound succinate dehydrogenase with substrate and competitive inhibitor. Biochim. Biophys. Acta 784, 24–34 (1984).
Hirawake, H., Wang, H., Kuramochi, T., Kojima, S. & Kita, K. Human complex II (succinate-ubiquinone oxidoreductase): cDNA cloning of the flavoprotein (Fp) subunit of liver mitochondria. J. Biochem. 116, 221–227 (1994).
Robinson, K.M., von Kieckebush-Guck, A. & Lemire, B.D. Isolation and characterization of a Saccharomyces cerevisiae mutant disrupted for the succinate dehydrogenase flavoprotein subunit. J. Biol. Chem. 266, 21347–21350 (1991).
Saghbini, M., Broomfield, E.P.L. & Scheffler, I.E. Studies on the assembly of complex II in yeast using chimeric human/yeast genes for the Ip subunit. Biochemistry 33, 159–165 (1994).
Mandel, J.L. & Chambon, P. DNA methylation: organ specific variations in the methylation pattern within and around ovalbumin and other chicken genes. Nucleic Acids Res. 7, 2081–2103 (1979).
Gutman, M. Modulation of mitochondrial succinate dehydrogenase activity, mechanism and function. Molec. cell. Biochem. 20, 41–60 (1978).
Dyer, M.R. & Walker, J.E. Sequences of members of the human gene family for the c subunit of mitochondrial ATP synthase. Biochem. J. 293, 51–64 (1993).
Saccone, C., Pesole, G. & Kadenbach, B. Evolutionary analysis of the nucleus-encoded subunits of mammalian cytochrome c oxidase. Eur. J. Biochem. 195, 151–156 (1991).
Desnuelle, C. et al. Multiple defect of the respiratory chain including complex II in a family with myopathy and encephalopathy. Biochem. Biophys. Res. Commun. 163, 695–700 (1989).
Spei, W. et al. Mitochondrial myopathy with lactic acidaemia, Fanconi-De Toni-Debré syndrome and a disturbed succinate: cytochrome c oxidoreductase activity. Eur. J. Pediatr. 147, 418–421 (1988).
Riggs, J.E. et al. Mitochondrial encephalopathy with decreased succinate-cytochrome c reductase activity. Neurology 34, 48–53 (1984).
Rivner, M.D. et al. Kearns-Sayre syndrome and complex II deficiency. Neurology 39, 693–696 (1989).
Rustin, P. et al. The investigation of respiratory chain disorders in heart using endomyocardial biopsies. J. Inher. metab. Dis. 16, 541–544 (1993).
Reichman, H. & Angelini, C. Single muscle fibre analyses in 2 brothers with succinate dehydrogenase deficiency. Eur. Neurol. 34, 95–98 (1993).
Rustin, P. et al. Biochemical and molecular investigations in respiratory chain deficiencies. Clin. Chim. Acta 228, 35–51 (1994).
Rötig, A. et al. Pearson's marrow-pancreas syndrome. A multisystem mitochondrial disorder in infancy. J. din. Invest. 86, 1601–1608 (1990).
Van Cong, N. et al. Assignment of the genes for human lysosomal acid lipases A and B to chromosomes 10 and 16. Hum. Genet. 55, 375–381 (1980).
Bourgeron, T. et al. Expression of respiratory chain deficiencies in human cultured cells. Neuromusc. Dis. 3, 605–608 (1993).
Chomczynski, P. & Sacchi, N. Single-step method of RNA isolation by acid guanidium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162, 156–159 (1987).
Viegas-Péquignot, E. In situ hybridization to chromosomes with biotinylated probes. In In situ hybridization: a practical approach (ed. Wilkinson, D.G.) 137–158 (IRL Press, Oxford,1992).
Robinson, K.M. & Lemire, B.D. Isolation and nucleotide sequence of the Saccharomyces cerevisiae gene for the succinate dehydrogenase flavoprotein subunit. J. Biol. Chem. 267, 10101–10107 (1992).
Chen, D.C., Yang, B.C. & Kuo, T.T. One-step transformation of yeast in stationary phase. Curr. Genet. 21, 83–84 (1992).
Birch-Machin, M.A. et al. The sequence of the flavoprotein subunit of bovine heart succinate dehydrogenase. J. biol. Chem. 267, 11553–11558 (1992).
Kita, K. cDNA cloning and comparative study on the active site of the flavoprotein subunit in mitochondrial fumarate reductase (complex II) from adult Ascaris suum. GenBank accession ♯D30650.
Aliabadi, Z., Winkler, H.H. & Wood, D.O. Isolation and characterization of the Rickettsia prowazekii gene encoding the flavoprotein subunit of succinate dehydrogenase. Gene 133, 135–140 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bourgeron, T., Rustin, P., Chretien, D. et al. Mutation of a nuclear succinate dehydrogenase gene results in mitochondrial respiratory chain deficiency. Nat Genet 11, 144–149 (1995). https://doi.org/10.1038/ng1095-144
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1095-144
This article is cited by
-
Mitochondrial dysfunction abrogates dietary lipid processing in enterocytes
Nature (2024)
-
SDHA: a key player in T cell-mediated intestinal disease severity
Cellular & Molecular Immunology (2022)
-
Cardio-onco-metabolism: metabolic remodelling in cardiovascular disease and cancer
Nature Reviews Cardiology (2022)
-
Complex II subunit SDHD is critical for cell growth and metabolism, which can be partially restored with a synthetic ubiquinone analog
BMC Molecular and Cell Biology (2021)
-
A recessive variant in TFAM causes mtDNA depletion associated with primary ovarian insufficiency, seizures, intellectual disability and hearing loss
Human Genetics (2021)