Abstract
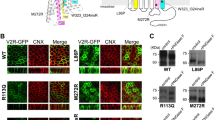
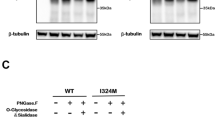
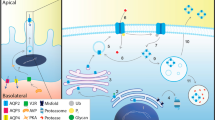
X–linked nephrogenic diabetes insipidus (NDI) is a rare disorder in which the kidney is insensitive to the antidiuretic hormone, vasopressin. It has been proposed that the kidney–specific V2 vasopressin receptor, a G protein–coupled receptor, is defective in this disorder as both the disease and the receptor map to Xq28. We report six unique mutations in the V2 receptor gene of five unrelated NDI patients, with one patient having two mutations. The most severely affected patient has a nonsense mutation which would terminate the protein in transmembrane domain III. Other mutations include three missense mutations, a frameshift and one small in–frame deletion. These results represent one of the first examples of recessive mutations affecting a G protein–coupled receptor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bimbaumer, M. et al. Molecular cloning of the receptor for human antidiuretic hormone. Nature 357, 333–335 (1992).
Lolait, S.J. et al. Cloning and characterization of a vasopressin V2 receptor and possible link to nephrogenic diabetes insipidus. Nature 357, 336–339 (1992).
Knoers, N. et al. Nephrogenic diabetes insipidus: close linkage with markers from the distal long arm of the human X chromosome. Hum. Genet. 30, 31–38 (1988).
Jans, D.A., van Oost, B.A., Ropers, H.H. & Fahrenholz, F. Derivatives of somatic cell hybrids which carry the human gene locus for nephrogenic diabetes insipidus (NDI) express functional vasopressin renal V2-type receptors. J. bioL. Chem. 265, 15379–15382 (1990).
Savarese, R.M. & Fraser, C.M. In vitro mutagenesis and the search for structure-function relationships among G protein-coupled receptors. Biochem. J. 283, 1–19 (1992).
Saiki, R.K. et al. Enzymatic amplification of β-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science 230, 1350–1354 (1985).
Orita, M., Iwahana, H., Kanazawa, H., Hayashi, K. & Sekiya, T. Detection of polymorphisms of human DNA by gel electrophoresis as single-strand conformation polymorphisms. Proc. natn. Acad. Sci. U.S.A. 86, 2766–2770 (1989).
Yap, E.P.H. & McGee, J.O. Nonisotopic SSCP detection in PCR products by ethidium bromide staining. Trends Genet. 8, 49 (1992).
Wood, W.I., Gitschier, J., Lasky, L.A. & Lawn, R.M. Base composition-independent hybridization in tetramethylammonium chloride: A method for oligonucleotide screening of highly complex gene libraries. Proc. natn. Acad. Sci. U.S.A. 82, 1585–1588 (1985).
Coulondre, C., Miller, J.H., Farabaugh, P.J. & Gilbert, W. Molecular basis of base substitution hotspots in Escherichia coli. Nature 274, 775–780 (1978).
Dohlman, H.G., Caron, M.R. & Lefkowitz, R.J. A family of receptors coupled to guanine nucleotide regulatory proteins. Biochemistry 26, 2657–2664 (1987).
Karnik, S.S., Sakmar, T.P., Chen, H.B. & Khorana, H.G. Cysteine residues 110 and 187 are essential for the formation of correct structure in bovine rhodopsin. Proc. natn. Acad. Sci. U.S.A. 85, 8459–8463 (1988).
Farabaugh, P.J., Schmeissner, U., Hofer, M. & Miller, J.H. Genetic studies of the lac represser VII. On the molecular nature of spontaneous hotspots in the lacl gene of Escherichia coli. J. molec. Biol. 126, 847–863 (1978).
Nakano, K.K. Familial nephrogenic diabetes insipidus. Hawaii med. J. 28, 205–208 (1969).
Langley, J.M. et al. Autosomal recessive inheritance of vasopressiri-resistant diabetes insipidus. Am. J. med. Gen. 38, 90–94 (1991).
Naik, D.V. & Valtin, H. Hereditary vasopressin-resistant urinary concentrating defects in mice. Am. J. Phys. 217, 1183–1190 (1969).
Bode, H.H. & Crawford, J.D. Nephrogenic diabetes insipidus in North America - the Hopewell hypothesis. New Engl. J. Med. 280, 750–754 (1969).
Dryja, T.P., Hahn, L.B., Cowley, G.S., McGee, T.L. & Berson, E.L. Mutation spectrum of the rhodopsin gene among patients with autosomal dominant retinitis pigmentosa. Proc. natn. Acad. Sci. U.S.A. 88, 9370–9374 (1991).
Rosenfeld, P.S. et al. A Null mutation in the rhodopsin gene causes rod photoreceptor dysfunction and autosomal recessive retinitis pigmentosa. Nature Genet. 1, 209–213 (1992).
Kovalic, D., Kwak, J. & Weisblum, B. General method for direct cloning of DNA fragments generated by primed enzymatic amplification. Nucl. Acid Res. 19, 4560–4563 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pan, Y., Metzenberg, A., Das, S. et al. Mutations in the V2 vasopressin receptor gene are associated with X–linked nephrogenic diabetes insipidus. Nat Genet 2, 103–106 (1992). https://doi.org/10.1038/ng1092-103
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1092-103
This article is cited by
-
Engineered transfer RNAs for suppression of premature termination codons
Nature Communications (2019)
-
Pathophysiology, diagnosis and management of nephrogenic diabetes insipidus
Nature Reviews Nephrology (2015)
-
Novel mutations associated with nephrogenic diabetes insipidus. A clinical-genetic study
European Journal of Pediatrics (2015)
-
Urinary concentration: different ways to open and close the tap
Pediatric Nephrology (2014)
-
A complex selection signature at the human AVPR1B gene
BMC Evolutionary Biology (2009)