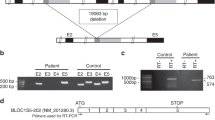
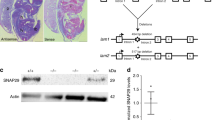
Abstract
Hermansky-Pudlak syndrome (HPS) is a genetically heterogeneous disease involving abnormalities of melanosomes, platelet dense granules and lysosomes. Here we have used positional candidate and transgenic rescue approaches to identify the genes mutated in ruby-eye 2 and ruby-eye mice (ru2 and ru, respectively), two 'mimic' mouse models of HPS. We also show that these genes are orthologs of the genes mutated in individuals with HPS types 5 and 6, respectively, and that their protein products directly interact. Both genes are previously unknown and are found only in higher eukaryotes, and together represent a new class of genes that have evolved in higher organisms to govern the synthesis of highly specialized lysosome-related organelles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Huizing, M., Anikster, Y. & Gahl, W.A. Hermansky-Pudlak syndrome and Chediak-Higashi syndrome: Disorders of vesicle formation and trafficking. Thromb. Haemost. 86, 233–245 (2001).
Spritz, R.A. Multi-organellar disorders of pigmentation: Tied up in traffic. Clin. Genet. 55, 309–317 (1999).
Gardner, J.M. et al. The mouse pale ear (ep) mutation is the homologue of human Hermansky-Pudlak syndrome (HPS). Proc. Natl. Acad. Sci. USA 94, 9238–9243 (1997).
Feng, G.H., Bailin, T., Oh, J. & Spritz, R.A. Mouse pale ear (ep) is homologous to human Hermansky-Pudlak syndrome and contains a rare 'AT-AC' intron. Hum. Molec. Genet. 6, 793–797 (1997).
Oh, J. et al. Positional cloning of a gene for Hermansky-Pudlak syndrome, a disorder of cytoplasmic organelles. Nat. Genet. 14, 300–306 (1996).
Feng, L. et al. The β3A subunit gene (Ap3b1) of the AP-3 adaptor complex is altered in the mouse hypopigmentation mutant pearl, a model for Hermansky-Pudlak Syndrome and night blindness. Hum. Molec. Genet. 8, 323–330 (1999).
Dell'Angelica, E.C., Shotelersuk, V., Aguilar, R.C., Gahl, W.A. & Bonifacino, J.S. Altered trafficking of lysosomal proteins in Hermansky-Pudlak syndrome due to mutations in the β3A subunit of the AP-3 adaptor. Mol. Cell 3, 11–21 (1999).
Suzuki, T. et al. The gene mutated in cocoa mice, carrying a defect of organelle biogenesis, is a homologue of the human Hermansky-Pudlak syndrome-3 gene. Genomics 78, 30–37 (2001).
Anikster, Y. et al. Mutation of a new gene causes a unique form of Hermansky-Pudlak syndrome in a genetic isolate of central Puerto Rico. Nat. Genet. 28, 376–380 (2001).
Suzuki, T. et al. Hermansky-Pudlak syndrome is caused by mutations in HPS4, the human homolog of the mouse light-ear gene. Nat. Genet. 30, 321–324 (2002).
Swank, R.T., Novak, E.K., McGarry, M.P., Rusiniak, M.E. & Feng, L. Mouse models of Hermansky-Pudlak syndrome: A review. Pig. Cell Res. 11, 60–80 (1998).
Swank, R.T., Novak, E.K., McGarry, M.P., Zhang, Q. & Feng, L. Abnormal vesicular trafficking in mouse models of Hermansky-Pudlak syndrome. Pig. Cell Res. 13, 59–67 (2000).
Silvers, W.K. The Coat Colors of Mice: A Model for Mammalian Gene Action and Interaction 103–104 (Springer-Verlag, New York, 1979).
Dunn, L.C. A new eye color mutant in the mouse with asymmetrical expression. Proc. Natl. Acad. Sci. 31, 343–346 (1945).
Eicher, E.M. The position of ru2 and qv with respect to the flecked translocation in the mouse. Genetics 64, 495–510 (1969).
Eicher, E.M. & Fox, S. Mouse News Lett. 56, 42 (1977).
Novak, E.K., Wieland, F., Jahreis, G.P. & Swank, R.T. Altered secretion of kidney lysosomal enzymes in the mouse pigment mutants ruby-eye, ruby-eye-2-J and maroon. Biochem. Genet. 18, 549–561 (1980).
Novak, E.K., Hui, S.-W. & Swank, R.T. Platelet storage pool deficiency in mouse pigment mutations associated with seven distinct genetic loci. Blood 63, 536–544 (1984).
Oberhauser, A.F. & Fernandez, J.M. A fusion pore phenotype in mast cells of the ruby-eye mouse. Proc. Natl. Acad. Sci. 93, 14349–14354 (1996).
Russell, L.B., Montgomery, C.S., Cacheiro, N.L. & Johnson, D.K. Complementation analyses for 45 mutations encompassing the pink-eyed dilution (p) locus of the mouse. Genetics 141, 1547–1563 (1995).
O'Brien, E.P. et al. Molecular map of chromosome 19 including three genes affecting bleeding time: ep, ru and bm. Mammal. Genome 5, 356–360 (1994).
Lee, S.T., Park, S.K., Lee, K.H., Holmes, S.M. & Spritz, R.A. A non-radioactive method or simultaneous detection of single-strand conformation polymorphisms (SSCPs) and heteroduplexes. Mol. Cells 5, 668–672 (1995).
Zhang, Q. et al. The gene for the muted (mu) mouse, a model for Hermansky-Pudlak Syndrome, defines a novel protein which regulates vesicle trafficking. Hum. Mol. Genet. 11, 697–706 (2002).
Kantheti, P. et al. Mutation in AP-3 delta in the mocha mouse links endosomal transport to storage deficiency in platelets, melanosomes and synaptic vesicles. Neuron 21, 111–122 (1998).
Robinson, M.S. & Bonifacino, J.S. Adaptor-related proteins. Curr. Opin. Cell. Biol. 13, 444–453 (2001).
Detter, J.C. et al. Rab geranylgeranyl transferase-α mutation in the gunmetal mouse reduces Rab prenylation and platelet synthesis. Proc. Natl. Acad. Sci. USA 97, 4144–4149 (2000).
Wilson, S.M. et al. A mutation in Rab27a causes the vesicle transport defects observed in ashen mice. Proc. Natl. Acad. Sci. USA 97, 7933–7938 (2000).
Wu, X., Wang, F., Rao, K., Sellers, J.R. & Hammer, J.A.R. Rab27a is an essential component of melanosome receptor for myosin Va. Mol. Biol. Cell 13, 1735–1749 (2002).
Strom, M., Hume, A.N., A.K., T., Barkagianni, E. & Seabra, M.C. A family of Rab27-binding proteins. Melanophilin links Rab27a and myosin Va function in melanosome transport. J. Biol. Chem. 277, 25423–25430 (2002).
Kuroda, T.S., Fukuda, M. & Mikoshiba, K. The Slp homology domain of synaptotagmin-like proteins 1–4 and Slac2 functions as a novel Rab27a binding domain. J. Biol. Chem. 277, 9212–9218 (2002).
Novak, E.K. et al. The regulation of platelet-dense granules by Rab27a in the ashen mouse, a model of Hermansky-Pudlak and Griscelli syndromes, is granule-specific and dependent on genetic background. Blood 100, 128–135 (2002).
Suzuki, T. et al. The mouse organellar biogenesis mutant buff (bf) results from a mutation in Vps33a, a homologue of yeast vps33 and Drosophila carnation (car). Proc. Natl. Acad. Sci. USA (in the press).
Huang, L., Kuo, Y.-M. & Gitschier, J. The pallid gene encodes a novel, syntaxin 13-interacting protein involved in platelet storage pool deficiency. Nat. Genet. 23, 329–332 (1999).
Falcon-Perez, J.M., Starcevic, M., Gautam, R. & Dell'Angelica, E.C. BLOC-1, a novel complex containing the pallidin and muted proteins involved in the biogenesis of melanosomes and platelet-dense granules. J. Biol. Chem. 277, 28191–28199 (2002).
Moriyama, K. & Bonifacino, J.S. Pallidin is a component of a multi-protein complex involved in the biogenesis of lysosome-related organelles. Traffic 3, 666–677 (2002).
Meisler, M.H., Wanner, L., & Strahler, J. Pigmentation and lysosomal phenotypes in mice doubly homozygous for both light-ear and pale-ear mutant alleles. J. Hered. 75, 103–106 (1984).
Oh, J. et al. The Hermansky-Pudlak syndrome (HPS) protein is part of a high molecular weight complex involved in biogenesis of early melanosomes. Hum. Mol. Genet. 9, 375–385 (2000).
Wixler, V. et al. Identification of novel interaction partners for the conserved membrane proximal region of α-integrin cytosplasmic domains. FEBS Lett. 445, 351–355 (1999).
Moore, K.J., Swing, D.A., Copeland, N.G. & Jenkins, N.A. Interaction of the murine dilute suppressor gene (dsu) with fourteen coat color mutations. Genetics 125, 421–430 (1990).
Feng, L. et al. The Hermansky-Pudlak syndrome 1 (HPS1) and HPS2 genes independently contribute to the production and function of platelet dense granules, melanosomes, and lysosomes. Blood 99, 1651–1658 (2002).
Bodenteich, A., Chissoe, S., Wang, Y.F. & Roe, B.A. Shotgun cloning as the strategy of choice to generate templates for high throughput dideoxynucleotide sequencing. in Automated DNA Sequencing and Analysis Techniques eds. Adams, M.D., Fields, C. and Venter, C. 42–50 (Academic Press, London, 1994).
Chissoe, S.L. et al. Strategies for rapid and accurate DNA sequencing. in Methods: A Companion to Methods in Enzymology, Vol. 3 ed. Roe. B.A. (Academic Press, New York 1991).
Acknowledgements
We thank M. Reddington, L. Zhen, Y. Jiang, D. Poslinski, D. Tabaczynski, M. Ellsworth, J. Pazik, and J. Tan for technical assistance; D. Swing for the microinjection; L. Gagnon for helping with the JE/Le cross; Y. Ni, X. Jiang and L. Song for sequencing BACs; and H. Chen for assistance in 5′ RACE analysis of ru2. P. Graf, C. McGregor and D. Ako-Adjei contributed to the construction of the ru genetic map. We thank H. Baumann for the gift of the STAT3 construct. This work was supported in part by the National Institutes of Health (R.T.S., E.M.E., B.A.R. and R.A.S.) and by the National Cancer Institute, Department of Health and Human Services (N.G.C. and N.A.J.). This research used core facilities supported in part by Roswell Park Cancer Institute's National Cancer Institute–funded Cancer Center Support Grant and by the Cancer Core Grant of The Jackson Laboratory.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, Q., Zhao, B., Li, W. et al. Ru2 and Ru encode mouse orthologs of the genes mutated in human Hermansky-Pudlak syndrome types 5 and 6. Nat Genet 33, 145–153 (2003). https://doi.org/10.1038/ng1087
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1087
This article is cited by
-
Novel HPS6 mutations identified by whole-exome sequencing in two Japanese sisters with suspected ocular albinism
Journal of Human Genetics (2016)
-
The construction of transgenic and gene knockout/knockin mouse models of human disease
Transgenic Research (2012)
-
A homolog of the human Hermansky–Pudluck syndrome-5 (HPS5) gene is responsible for the oa larval translucent mutants in the silkworm, Bombyx mori
Genetica (2012)
-
Clinical, Molecular, and Cellular Features of Non-Puerto Rican Hermansky–Pudlak Syndrome Patients of Hispanic Descent
Journal of Investigative Dermatology (2011)
-
Homozygosity Mapping and Whole-Exome Sequencing to Detect SLC45A2 and G6PC3 Mutations in a Single Patient with Oculocutaneous Albinism and Neutropenia
Journal of Investigative Dermatology (2011)