Abstract
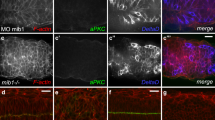
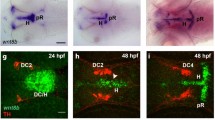
The Notch-signalling pathway is important in establishing metameric pattern during somitogenesis. In mice, the lack of either of two molecules involved in the Notch-signalling pathway, Mesp2 or presenilin-1 (Ps1), results in contrasting phenotypes: caudalized versus rostralized vertebra. Here we adopt a genetic approach to analyse the molecular mechanism underlying the establishment of rostro-caudal polarity in somites. By focusing on the fact that expression of a Notch ligand, Dll1, is important for prefiguring somite identity, we found that Mesp2 initiates establishment of rostro-caudal polarity by controlling two Notch-signalling pathways. Initially, Mesp2 activates a Ps1-independent Notch-signalling cascade to suppress Dll1 expression and specify the rostral half of the somite. Ps1-mediated Notch-signalling is required to induce Dll1 expression in the caudal half of the somite. Therefore, Mesp2- and Ps1-dependent activation of Notch-signalling pathways might differentially regulate Dll1 expression, resulting in the establishment of the rostro-caudal polarity of somites.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Tam, P.P.L., Goldman, D., Camus, A. & Schoenwolf, G.C. Early events of somitogenesis in higher vertebrates: allocation of precursor cells during gastrulation and the organization of a meristic pattern in the paraxial mesoderm . Curr. Top. Dev. Biol. 47, 1– 32 (2000).
Franco del Amo, F. et al. Expression of Motch, a mouse homolog of Drosophila Notch, suggests an important role in early postimplantation mouse development . Development 115, 737– 745 (1992).
Bettenhausen, B., Hrabe de Angelis, M., Simon, D., Guénet, J.-L. & Gossler, A. Transient and restricted expression during mouse embryogenesis of Dll1, a murine gene closely related to Drosophila Delta. Development 121, 2407–2418 (1995).
Dunwoodie, S.L., Henrique, D., Harrison, S.M. & Beddington, R.S.P. Mouse Dll3: a novel divergent Delta gene which may complement the function of other Delta homologues during early pattern formation in the mouse embryo. Development 124, 3065 –3076 (1997).
Johnston, S.H. et al. A family of mammalian Fringe genes implicated in boundary determination and the Notch pathway. Development 124 , 2245–2254 (1997).
Conlon, R.A., Reaume, A.G. & Rossant, J. Notch1 is required for the coordinate segmentation of somites. Development 121, 1533– 1545 (1995).
Swiatek, P.J., Lindsell, C.E., Franco del Amo, F., Weinmaster, G. & Gridley, T. Notch1 is essential for postimplantation development in mice. Genes Dev. 8, 707–719 (1994).
Hrabe de Angelis, M., McIntyre, J. II & Gossler, A. Maintenance of somite borders in mice requires the Delta homologue Dll1. Nature 386, 717–721 (1997).
Kusumi, K. et al. The mouse pudgy mutation disrupts Delta homologue Dll3 and initiation of early somite boundaries. Nature Genet. 19, 274–278 ( 1998).
Zhang, N. & Gridley, T. Defects in somite formation in lunatic fringe-deficient mice. Nature 394 , 374–377 (1998).
Evrard, Y.A., Lun, Y., Aulehla, A., Gan, L. & Johnson, R.L. lunatic fringe is an essential mediator of somite segmentation and patterning. Nature 394, 377–381 (1998).
Oka, C. et al. Disruption of the mouse RBP-Jk gene results in early embryonic death. Development 121, 3291– 3301 (1995).
Saga, Y., Hata, N., Koseki, H. & Taketo, M.M. Mesp2: a novel mouse gene expressed in the presegmented mesoderm and essential for segmentation initiation. Genes Dev. 11, 1827–1839 (1997).
del Barco Barrantes, I. et al. Interaction between Notch signaling and Lunatic fringe during somite boundary formation in the mouse. Curr. Biol. 9, 470–480 (1999).
De Strooper, B. et al. A presenilin-1-dependent γ-secretase-like protease mediates release of Notch intracellular domain. Nature 398, 518–522 (1999).
Struhl, G. & Greenwald, I. Presenilin is required for activity and nuclear access of Notch in Drosophila. Nature 398, 522–525 ( 1999).
Ye, Y., Lukinova, N. & Fortini, M.E. Neurogenic phenotypes and altered Notch processing in Drosophila Presenilin mutants. Nature 398 , 525–529 (1999).
Wong, P.C. et al. Presenilin 1 is required for Notch1 and Dll1 expression in the paraxial mesoderm. Nature 387, 288–292 (1997).
Palmeirim, I., Henrique, D., Ish-Horowicz, D. & Pourquie, O. Avian hairy gene expression identifies a molecular clock linked to vertebrate segmentation and somitogenesis. Cell 91, 639–648 (1997).
Neidhardt, L.M., Kispert, A. & Herrmann, B.G. A mouse gene of the paired-related homeobox class expressed in the caudal somite compartment and in the developing vertebral column, kidney and nervous system. Dev. Genes Evol. 207, 330–339 (1997).
Goldstein, R.S. & Kalcheim, C. Determination of epithelial half-somites in skeletal morphogenesis. Development 116, 441–445 ( 1992).
Takebayashi, K., Akazawa, C., Nakanishi, S. & Kageyama, R. Structure and promoter analysis of the gene encoding the mouse helix-loop-helix factor HES-5. Identification of the neural precursor cell-specific promoter element. J. Biol. Chem. 270, 1342– 1349 (1995).
de la Pompa, J.L. et al. Conservation of the Notch signaling pathway in mammalian neurogenesis . Development 124, 1139– 1148 (1997).
Donoviel, D.B. et al. Mice lacking both presenilin genes exhibit early embryonic patterning defects. Genes Dev. 13, 2801– 2810 (1999).
Jen, W.-C., Wettstein, D., Turner, D., Chitnis, A. & Kintner, C. The Notch ligand, X-Delta-2, mediates segmentation of the paraxial mesoderm in Xenopus embryos. Development 124, 1169–1178 ( 1997).
Jen, W.-C., Gawantka, V., Pollet, N., Niehrs, C. & Kintner, C. Periodic repression of Notch pathway genes governs the segmentation of Xenopus embryos. Genes Dev. 13, 1486–1499 ( 1999).
Sparrow, D.B. et al. Thylacine 1 is expressed segmentally within the paraxial mesoderm of the Xenopus embryo and interacts with the Notch pathway . Development 125, 2041– 2051 (1998).
Yagi, T. et al. A novel ES cell line, TT2, with high germline-differentiating potency. Anal. Biochem. 214, 70– 76 (1993).
Ishii, Y., Fukuda, K., Saiga, H., Matsushita, S. & Yasugi, S. Early specification of intestinal epithelium in the chicken embryo: a study on the localization and regulation of CdxA expression. Dev. Growth Differ. 39, 643– 653 (1997).
Acknowledgements
We thank R. Conlon (Notch1 cDNA), A. Gossler (Dll1 cDNA), P. Gruss (Uncx4.1 cDNA), R. Kageyama (Hes5 cDNA) and E.M. De Robertis (Cer1 cDNA) for providing reagents; and W. Murai, M. Ikumi, S. Sinzawa, M. Uchida and S. Takeda for technical assistance. This work was supported in part by Grants in Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture in Japan and Special Coordination Funds for Promoting Science and Technology to Y.S., and grants from the Mochida Memorial Foundation for Medical and Pharmaceutical Research, Uehara Memorial Foundation and Ichiro Kanehara Foundation to H.K.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Takahashi, Y., Koizumi, Ki., Takagi, A. et al. Mesp2 initiates somite segmentation through the Notch signalling pathway . Nat Genet 25, 390–396 (2000). https://doi.org/10.1038/78062
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/78062
This article is cited by
-
Reconstituting human somitogenesis in vitro
Nature (2023)
-
Reconstruction and deconstruction of human somitogenesis in vitro
Nature (2023)
-
Earlier and broader roles of Mesp1 in cardiovascular development
Cellular and Molecular Life Sciences (2017)
-
The roles of Mesp family proteins: functional diversity and redundancy in differentiation of pluripotent stem cells and mammalian mesodermal development
Protein & Cell (2015)
-
What can vertebrates tell us about segmentation?
EvoDevo (2014)